Home
People
Project Details
Protocols
Completed Systems
Notebook
Biosafety
Human Impact
References
Support
|
Monday 7/19
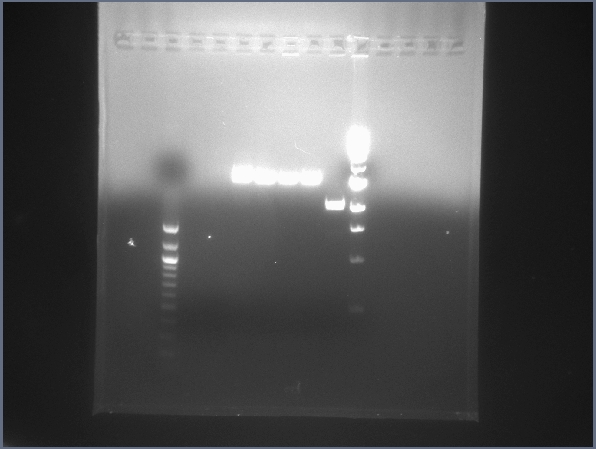 Digest test gel with US and DS digested bricks. (7/19) - Group meeting!
- Discussed issues with cloning and next week's focus.
- Performed a digest (now for 2hr) and ligations (L18-23). Transformed into DH5α.
- Turns out that the sequencing primers we ordered (G00100 & G00101) are incorrect - they appear to bind on the wrong sides of the insert. We ordered new primers designed to bind to the BioBrick prefix and suffix.
- Ran gel of digest product to verify correct bands.
Tuesday 7/20
- All ligation transformations failed. Will try 16hr ligations and purifying digest products with a PCR purification kit.
- Made new growth curves testing AIBN effect on cells in LB
- Performed another live/dead assay
- Had solutions of cells in glycerol and AIBN
- Plated cells every hour for three hours
- Combined glycerol solutions with LB and plated the solution
- New 16hr ligation procedure:
- 30μL total: 2μL T4 ligase, 3μL 10x ligase buffer, 5μL vector, 10μL of each brick. Incubate for 16hr at 16°C.
- Started more cultures to miniprep tomorrow: I13504, J04450, K274002, M30109.
Wednesday 7/21
- Transformed 3μL of each ligation product from yesterday (time constants all w/in 3.8-4.0). Plated 50μL of each on plates.
Thursday 7/22
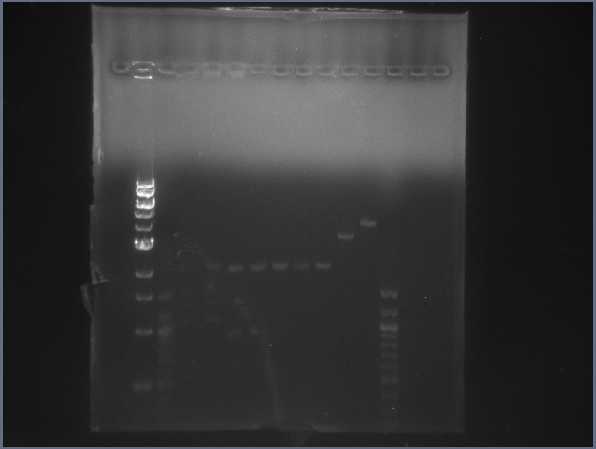 Digest test gel with upstream, downstream, and backbone digested bricks. (7/22) 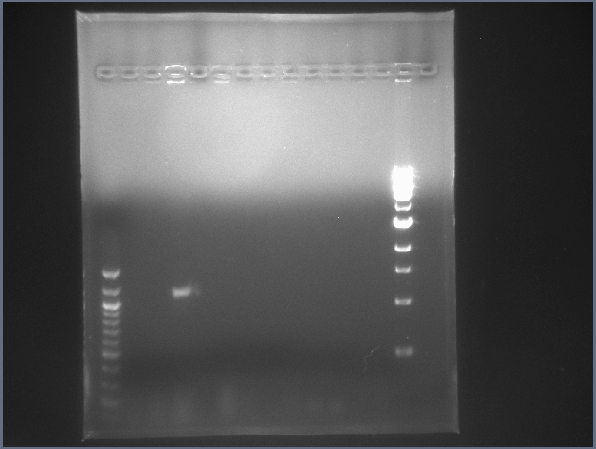 CPCR gel featuring L17, L18, L25, & L27. (7/22) - Prepared new sequencing primers in preparation for CPCR.
- Transformation plates are almost completely empty - will centrifuge and plate remaining transformation cells.
- Colony PCR mixture:
- 10μL 10x PCR buffer, 2μL dNTP mix, 1μL forward primer, 1μL reverse primer, 0.5μL Taq polymerase, fill to 100μL total. (Optional: add 20μL Qiagen Q buffer. Doesn't appear to help any of our reactions.)
- Pick colony and add to water (85.5μL). Incubate at 100°C for 10min. Add remaining ingredients and begin PCR program:
- Initial denaturation: 94°C, 4min
- 30 cycles:
- 94°C, 30s
- 54°C, 30s
- 72°C, 2min (1min/kb)
- Final extension: 72°C, 10min
- Finish: 5°C forever
- Ran colony PCR on 4 ligations, 2 had the correct insert length: L17, L27.
- Ran gel of digest products from yesterday to verify correctness of the digestion.
Friday 7/23
 CPCR gel featuring L16, L17, L18, & L21. (7/23) 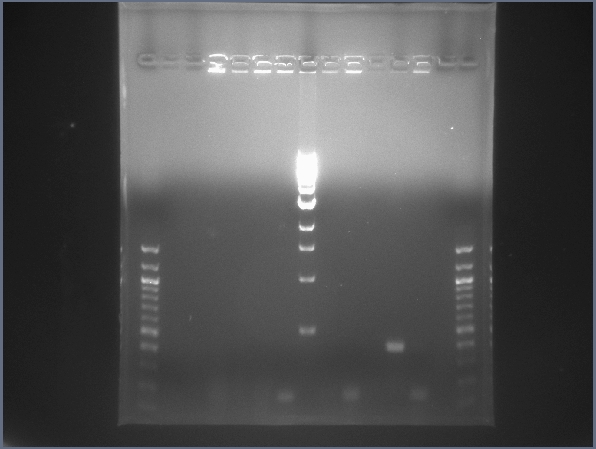 CPCR gel featuring L25, L26, L27, & L29. (7/23) - Plates containing the centrifuged transformation cells contained many colonies!
- Began 21 more CPCR reactions on ligations: L17, L18, L21, L27, L29, L16, L26, L25
- Note that because L18 is a coding sequence, it required the coding prefix primer; all others used the noncoding prefix primer.
- Received our heat shock promoter from the Registry today! Made a plate and culture of this brick (K112400).
- Purified yesterday's 4 CPCR reactions using a PCR purification kit.
- Concentrations of each are all quite low (<20ng/μL) - we may want to increase the number of PCR cycles.
- Ran 2 gels of all the CPCR results.
Weekend 7/24-25
Saturday 7/24
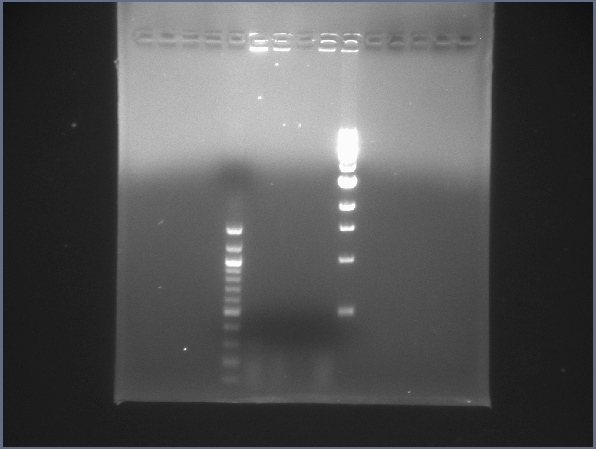 CPCR gel featuring L26 & L21. (7/24) - Started 4 new CPCR reactions on L21 and L26 to verify them more accurately.
- Disposed of all failed cultures...
- Prepped successful clones: 17-6, 18-3, 21-3, 26-1, 27-4, 29-2, and the heat shock promoter (K112400).
- Prepared digest:
- K215000 (u/d)
- K112400 (u/d)
- B0034 (u)
- R0082 (u)
- L17 (u)
- L18 (u/d)
- L26 (u)
- I13504 (d)
- L21 (d)
- Began ligations: L16, L19, L42, L43, L44, L23, L24, L31, L40, L34, L35
- Ran another gel of the CPCR products.
Sunday 7/25
- Transformed all ligations from yesterday (time constants all within 3.6-4.0). Used 4μL for all, except for L34 & L35, which sparked, so used 3μL instead.
- Made 2YT for making more electrocompetent cells.
- Plated all transformation cells (centrifuged at 1000g for 15min).

|
 "
"






