Team:Freiburg Bioware/Project/Methods
From 2010.igem.org
Methods
Contents |
Method Development
Purification of AAV particles
Cell Culture
Imaging AAV: EM/AFM pictures
To visualise our custom AAV particles, we took electron microscopy as well as atomic force microscopy pictures from our virus samples
ITR cloning
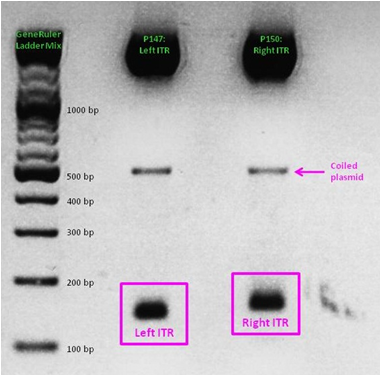
As a part of our modularization of the AAV vector plasmids, we needed to extract the sequences making up the ITRs at each end of the vector and clone them into an IGEM-compatible backbone. But due to the ITRs’ strong secondary structures, none of our PCR-based approaches worked. External companies weren’t able to synthesize or even sequence the ITRs. Taking advantage of NotI and PstI restriction sites flanking the ITRs, we worked out a complex cloning strategy that finally led to functional ITR motives in the rfc10 standard.
See also: Hannas' ITR Diary
Serum-free cell culture medium
Introduction
Serum-free mediums allow users to standardize their cell culture conditions. It contains no animal proteins or animal-origin constituents, e.g. FCS (fetal calf serum).
The AAV-293 cells are used for AAV-2 production and are usually grown in (among other chemicals, such as nutrients, antibiotics, growth factors) serum supplemented DMEM medium. Regarding Western Blots, size exclusion chromatographies and other (purification) methods, the undefined and also highly variable serum products can disturb or interfere with these methods. Therefore it is useful for many applications to grow AAV-293 cells in serum-free medium.
Because our long term goal for AAV vectors is application in human patients, we are also trying to develop new methods to produce pure, uncontaminated AAV particles. The use of FCS to supplement cell culture medium for AAV particle production is problematic because even smallest amounts of animal antigens in the administered drug could lead to a strong immune response in patients.
Testing serum free medium
Serum free medium was obtained from AAV-293 cells are not adapted to serum-free growth conditions so we had to accustom them to the new growth conditions step by step, starting with 25% serum-free medium, e.g. 15 ml DMEM (not serum-free) + 5 ml serum-free medium (FreeStyle™ 293 Expression Medium) for a T75 flask. Each step takes at least 1 passage. We raised the serum-free ratio to 100 % over 7 passages.
100% serum-free cells grow slower compared to the serum-supplemented ones and we had to check them regularly via microscopy because the medium contains no pH indicator.
Results
Even though cells grew slower and handling was more difficult due to a missing pH indicator, we successfully cultivated AAV-293 cells in serum-free medium. The cells were used for AAV production, and we produced similar amounts of virus particles compared to cells grown in FCS-supplemented medium. Production efficiency can’t be compared exactly because after seeding cells for transfection they don’t grow as fast as the AAV-293 in serum containing medium.
Established Methods
Protocols; Standard Operating Procedures
Standard Protocol: Cloning
Media:Freiburg10_Advanced_Cloning_Protocol_04_08_2010.pdf
Media:Split_cellculture.pdf
Media:Freeze_cellculture.pdf
Media:production of competent E.coli.pdf
Media:Freiburg10_Transfection_protocoll.pdf
Media:Freiburg10 Thawing cells.pdf
Media:Freiburg10_Aminoacids_vs_restrictionsites.pdf
Media:Freiburg10 Endotoxinfreie Midi.pdf
Media:Freiburg10_LB+Agar.pdf
Media:Freiburg10_Subcloning_cap_into_pAAV_RC.pdf
Media:Freiburg10_Quantitative_realtime_PCR_for_Titering_of_infectious_AAV_particles.pdf
 "
"