Team:Newcastle/13 August 2010
From 2010.igem.org
(→Results) |
(→Gel electrophoresis of subtilin immunity BioBrick fragments) |
||
| (3 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
| - | =Gel electrophoresis of subtilin immunity | + | =Gel electrophoresis of subtilin immunity part fragments= |
==Aims== | ==Aims== | ||
| Line 21: | Line 21: | ||
==Results== | ==Results== | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
{|border=1 | {|border=1 | ||
| Line 44: | Line 38: | ||
==Discussion== | ==Discussion== | ||
| - | + | The concentration of the fragments range from 21.3 µl/ml to 28.8 µl/ml. This is acceptable as during the gel extraction step, some of the DNA would be lost. | |
==Conclusion== | ==Conclusion== | ||
| - | + | We have successfully extracted all the parts required for the Subtilin Immunity BioBrick. The Gibson cloning method will be used next week. | |
Latest revision as of 23:41, 27 October 2010

| |||||||||||||
| |||||||||||||
Contents |
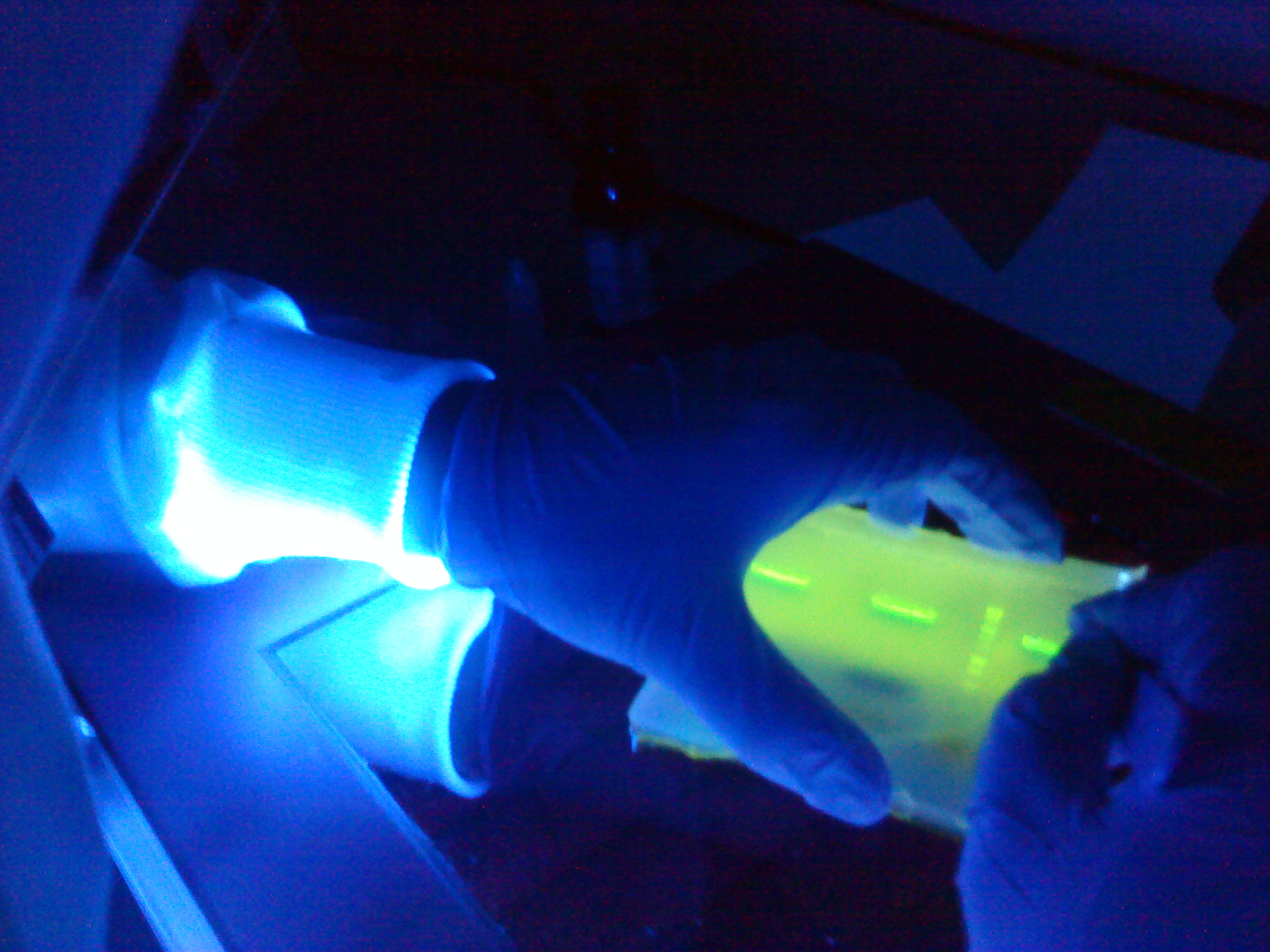
Gel electrophoresis of subtilin immunity part fragments
Aims
The aim of this experiment is to do gel extraction for the spaIFEG gene cluster amplifed on the 12th August, 2010 and the Plasmid Vector, Promoter & RBS and Double terminator amplifed on the 11th August, 2010. Finally, NanoDrop will be performed for all the fragments.
Materials and protocol
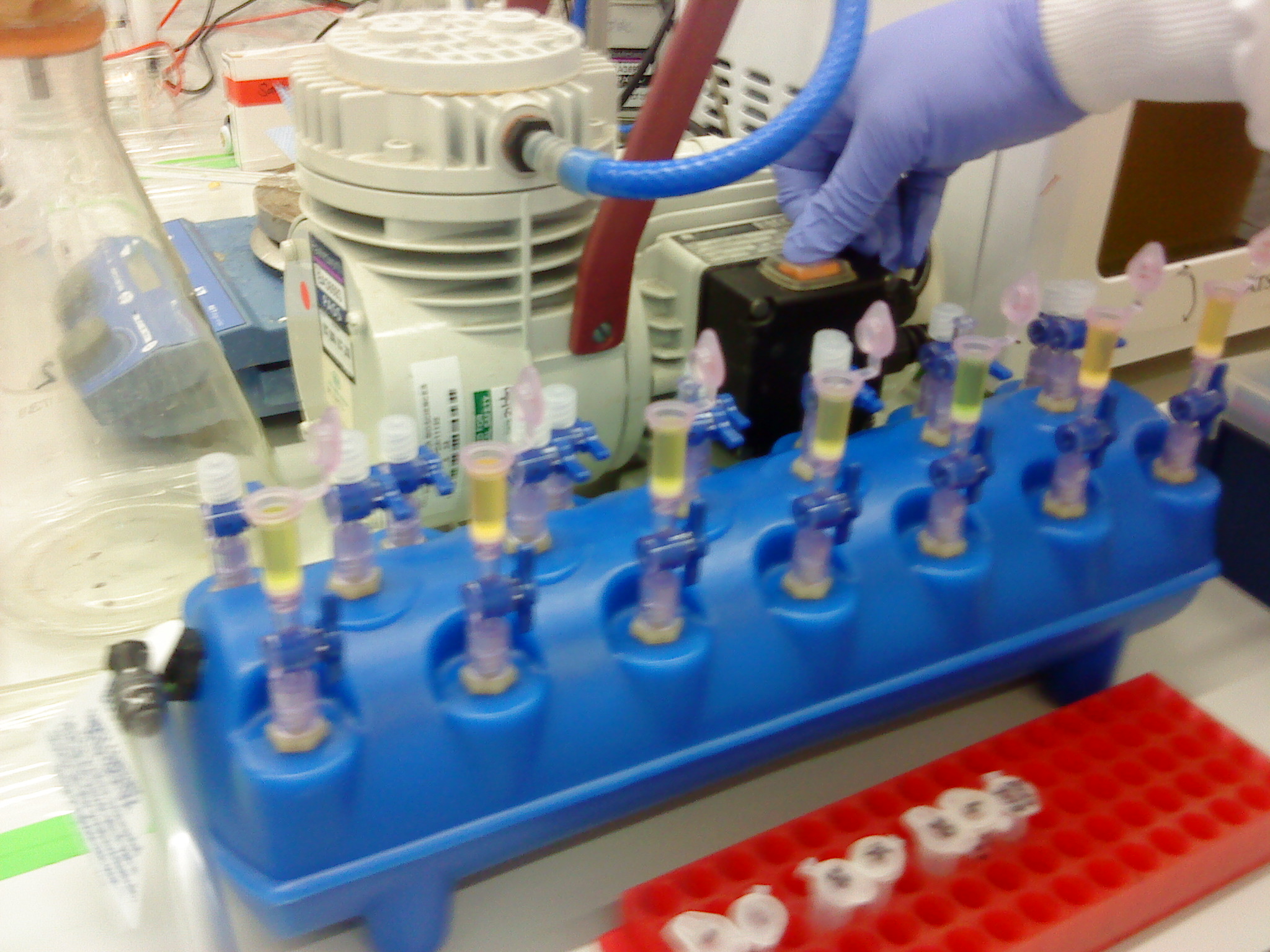
Please refer to the gel extraction and NanoDrop protocols. Instead of using the centrifuge to bind the DNA from our sample to the QIAquick column, a vacuum manifold was used instead. It is the first time it had been used. The advantage of this was that the extraction could be carried out all at the same time.
Results
| Vector | Promoter | Immunity gene cluster | Double terminator |
|---|---|---|---|
| 21.3 µl/ml | 28.8 µl/ml | 27.0 µl/ml | 27.5 µl/ml |
Table 1: Nanodrop spectrophotometer results. Table represents the amount of plasmid present in µl/ml quantity.
Discussion
The concentration of the fragments range from 21.3 µl/ml to 28.8 µl/ml. This is acceptable as during the gel extraction step, some of the DNA would be lost.
Conclusion
We have successfully extracted all the parts required for the Subtilin Immunity BioBrick. The Gibson cloning method will be used next week.
Go back to our main Lab book page
 
|
 "
"