Team:Tokyo Tech/Project/Apple Reporter2
From 2010.igem.org
(→Abstract) |
|||
| Line 43: | Line 43: | ||
=Abstract= | =Abstract= | ||
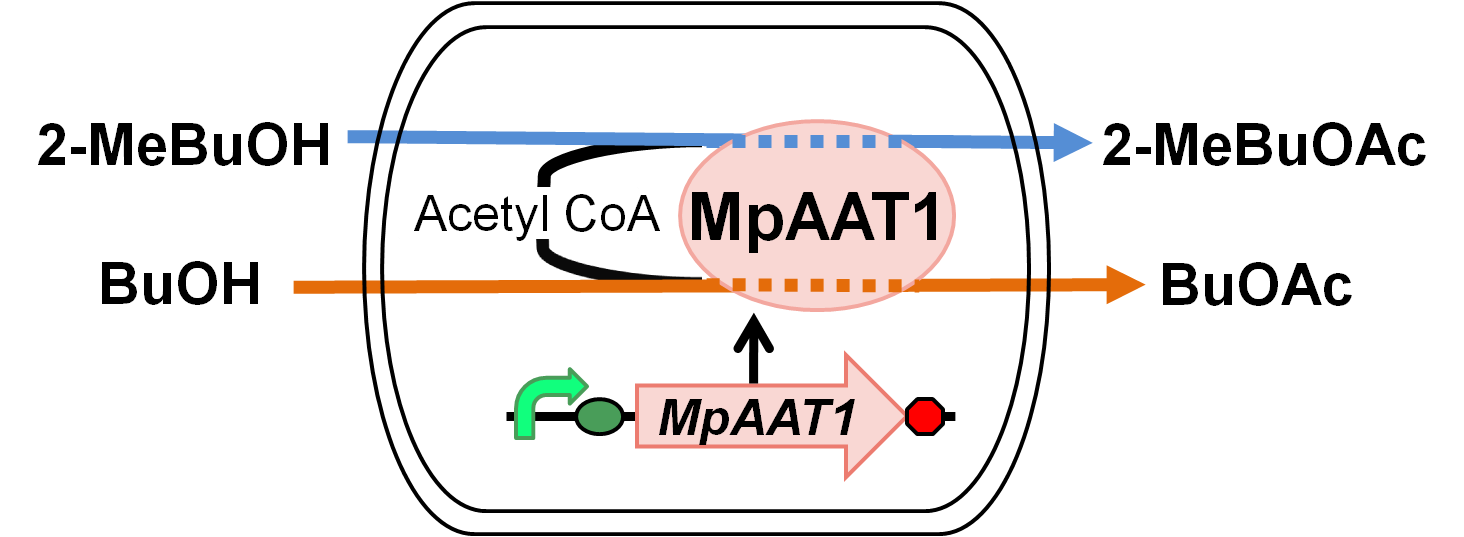
| - | We designed apple fragrance expression device with MpAAT1. Fig. 2-2-1 shows the outline of the device. MpAAT1 converts substrate and Acetyl CoA into apple fragrance. Acetyl CoA exists originally in E.coli cell. | + | We designed apple fragrance expression device with MpAAT1[1]. Fig. 2-2-1 shows the outline of the device. MpAAT1 converts substrate and Acetyl CoA into apple fragrance molecules. Acetyl CoA exists originally in E.coli cell. MpAAT1 converts 2-methyl butanol and butanol into 2-methylbutyl acetate and butyl acetate respectively. |
| + | [[image:K395602.png|630px|center|thumb|Fig. 2-2-1. Apple fragrance expression device]] | ||
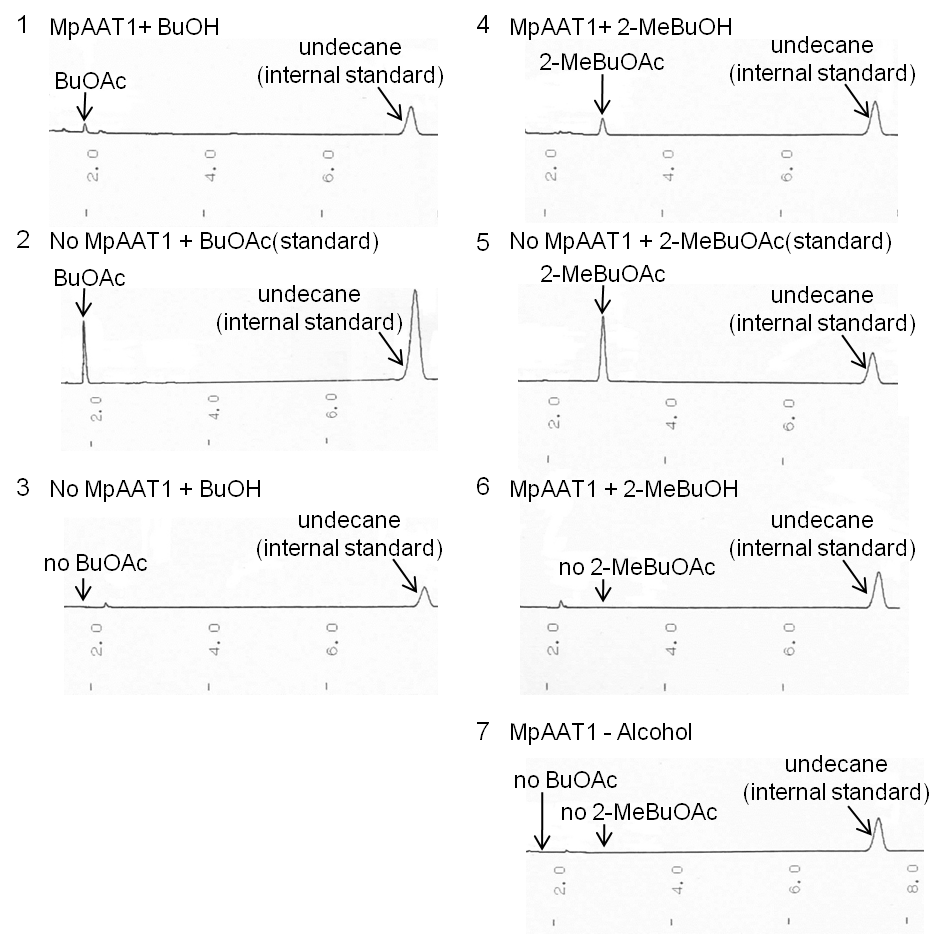
| - | [[image: | + | We performed gas chromatography to confirm the production of the esters, and the results revealed that MpAAT1 successfully converted alcohol and Acetyl CoA into our target esters (Fig. 2-2-2). |
| + | |||
| + | [[image:Tokyotech apple fragrance GC.png|630px|center|thumb| Fig. 2-2-2. Gas chromatography analysis of apple fragrance expression device. <br>This work is done by Toshitaka Matsubara]] | ||
| + | (1) MpAAT1 + BuOH, (2) No MpAAT1 + BuOAc, (3) No MpAAT1 + BuOH, (4) MpAAT1 + 2-MeBuOH, (5) No MpAAT1 + 2-MeBuOAc, (6) No MpAAT1 + 2-MeBuOH, (7) MpAAT1 - 2-MeBuOH. | ||
| + | |||
| + | (A) In (2), the peak of BuOAc is shown. Comparing with (1), the position of the peak is in the same position. From this result, we can see MpAAT1 converted BuOH into BuOAc. | ||
| + | (B) In (3), we observed the peak of BuOH, which we are going to use as substrate.This graph doesn’t show peak in the same place with that of BuOAc in (1).This means BuOH used as substrate doesn’t contain BuOAc. | ||
| + | (C) In the same way as (A), from comparison of (4) and (5), we can see 2-MeBuOAC is synthesized from 2-MeBuOH. | ||
| + | (D) (6) shows the peak of 2-MeBuOH, though no peak of 2-MeBuOAc. Thus, 2-MeBuOH doesn’t contain 2- MeBuOAc. | ||
| + | (E) In (7), no peak of ester is observed. This sample wasn’t added with substrates, so MpAAT1 without substrates didn’t produce esters. | ||
| + | From these results, we can conclude that E.Coli with MpAAT1 is able to convert substrate alcohols into esters. | ||
| - | |||
=Introduction= | =Introduction= | ||
| Line 57: | Line 67: | ||
=Result= | =Result= | ||
| - | We | + | We transformed MpAAT1([http://partsregistry.org/Part:BBa_K395602 BBa_K395602]) on pSB6 along with pTrx6 into E.coli BL21 DE3, and cultured after addition of alcohols(2-MeBuOH or BuOH) as substrates. |
| - | After 12 hours of incubation, we extracted organic soution layer from the culture and analyzed by gas chromatography (Fig. 2-2-2). Peaks of the esters producing apple fragrance(2-MeBuOAc or BuOAc) were detected. | + | After 12 hours of incubation, we extracted organic soution layer from the culture and analyzed by gas chromatography (Fig. 2-2-2). Peaks of the esters producing apple fragrance (2-MeBuOAc or BuOAc) were detected. |
| + | [[image:Tokyotech apple fragrance GC.png|630px|left|thumb|Fig. 2-2-2. Gas chromatography analysis of apple fragrance expression device. <br>This work is done by Toshitaka Matsubara]] | ||
| + | (1) MpAAT1 + BuOH, (2) No MpAAT1 + BuOAc, (3) No MpAAT1 + BuOH, (4) MpAAT1 + 2-MeBuOH, (5) No MpAAT1 + 2-MeBuOAc, (6) No MpAAT1 + 2-MeBuOH, (7) MpAAT1 - 2-MeBuOH. | ||
| - | + | (A) In (2), the peak of BuOAc is shown. Comparing with (1), the position of the peak is in the same position. From this result, we can see MpAAT1 converted BuOH into BuOAc. | |
| + | (B) In (3), we observed the peak of BuOH, which we are going to use as substrate.This graph doesn’t show peak in the same place with that of BuOAc in (1).This means BuOH used as substrate doesn’t contain BuOAc. | ||
| + | (C) In the same way as (A), from comparison of (4) and (5), we can see 2-MeBuOAC is synthesized from 2-MeBuOH. | ||
| + | (D) (6) shows the peak of 2-MeBuOH, though no peak of 2-MeBuOAc. Thus, 2-MeBuOH doesn’t contain 2- MeBuOAc. | ||
| + | (E) In (7), no peak of ester is observed. This sample wasn’t added with substrates, so MpAAT1 without substrates didn’t produce esters. | ||
| + | From these results, we can conclude that E.Coli with MpAAT1 is able to convert substrate alcohols into esters. | ||
| - | |||
=Discussion= | =Discussion= | ||
| - | |||
| - | |||
From the result of the experiment above, we can conclude that engineered E.coli successfully converted alcohols added as substrates into esters. | From the result of the experiment above, we can conclude that engineered E.coli successfully converted alcohols added as substrates into esters. | ||
| - | Moreover, we synthesized MpAAT1 and esters using E.coli BL21 DE3 as a chassis, which was reported to be implausible in former report(s) | + | Moreover, we synthesized MpAAT1 and esters using E.coli BL21 DE3 as a chassis, which was reported to be implausible in former report(s)[1]. |
| - | In 2008, C.R. Shen and J.C. Liao succeeded in synthesizing butanol from E.coli. If we take advantage of this engineered E.coli, we could produce apple fragrance ester without the addition of substrate. | + | In 2008, C.R. Shen and J.C. Liao[2] succeeded in synthesizing butanol from E.coli. If we take advantage of this engineered E.coli, we could produce apple fragrance ester without the addition of substrate. |
| - | + | ||
<!-- ここまでね--> | <!-- ここまでね--> | ||
Revision as of 14:56, 27 October 2010
2-2 Fragrance
Contents |
Abstract
We designed apple fragrance expression device with MpAAT1[1]. Fig. 2-2-1 shows the outline of the device. MpAAT1 converts substrate and Acetyl CoA into apple fragrance molecules. Acetyl CoA exists originally in E.coli cell. MpAAT1 converts 2-methyl butanol and butanol into 2-methylbutyl acetate and butyl acetate respectively.
We performed gas chromatography to confirm the production of the esters, and the results revealed that MpAAT1 successfully converted alcohol and Acetyl CoA into our target esters (Fig. 2-2-2).
(1) MpAAT1 + BuOH, (2) No MpAAT1 + BuOAc, (3) No MpAAT1 + BuOH, (4) MpAAT1 + 2-MeBuOH, (5) No MpAAT1 + 2-MeBuOAc, (6) No MpAAT1 + 2-MeBuOH, (7) MpAAT1 - 2-MeBuOH.
(A) In (2), the peak of BuOAc is shown. Comparing with (1), the position of the peak is in the same position. From this result, we can see MpAAT1 converted BuOH into BuOAc. (B) In (3), we observed the peak of BuOH, which we are going to use as substrate.This graph doesn’t show peak in the same place with that of BuOAc in (1).This means BuOH used as substrate doesn’t contain BuOAc. (C) In the same way as (A), from comparison of (4) and (5), we can see 2-MeBuOAC is synthesized from 2-MeBuOH. (D) (6) shows the peak of 2-MeBuOH, though no peak of 2-MeBuOAc. Thus, 2-MeBuOH doesn’t contain 2- MeBuOAc. (E) In (7), no peak of ester is observed. This sample wasn’t added with substrates, so MpAAT1 without substrates didn’t produce esters. From these results, we can conclude that E.Coli with MpAAT1 is able to convert substrate alcohols into esters.
Introduction
Apple fragrance is featured by combinations of volatile compounds, including alcohols, aldehydes, ketones, sesquiterpenes and esters. Esters are associated with fruity attributes of fruit fragrance. In the commercial apple cultivar, over 30 esters have been identified.[Ref]Ester biosynthesis is catalyzed by alcohol acyl transferases (AATs) that use coenzyme A(CoA) donors together with alcohol acceptors as substrates. We learned it’s able to make E. coli produce apple fragrance by adding the gene MpAAT1 because Acetyl CoA exists originally in E.coli. MpAAT1 produces a predicted protein containing character of other plant acyl transferases. In contrast with production of pigment, production of fragrance has hardly been worked in iGEM and few parts exist.For these reasons, we introduced a new BioBrick part to synthesize components of apple fragrance.
Result
We transformed MpAAT1(BBa_K395602) on pSB6 along with pTrx6 into E.coli BL21 DE3, and cultured after addition of alcohols(2-MeBuOH or BuOH) as substrates.
After 12 hours of incubation, we extracted organic soution layer from the culture and analyzed by gas chromatography (Fig. 2-2-2). Peaks of the esters producing apple fragrance (2-MeBuOAc or BuOAc) were detected.
(1) MpAAT1 + BuOH, (2) No MpAAT1 + BuOAc, (3) No MpAAT1 + BuOH, (4) MpAAT1 + 2-MeBuOH, (5) No MpAAT1 + 2-MeBuOAc, (6) No MpAAT1 + 2-MeBuOH, (7) MpAAT1 - 2-MeBuOH.
(A) In (2), the peak of BuOAc is shown. Comparing with (1), the position of the peak is in the same position. From this result, we can see MpAAT1 converted BuOH into BuOAc. (B) In (3), we observed the peak of BuOH, which we are going to use as substrate.This graph doesn’t show peak in the same place with that of BuOAc in (1).This means BuOH used as substrate doesn’t contain BuOAc. (C) In the same way as (A), from comparison of (4) and (5), we can see 2-MeBuOAC is synthesized from 2-MeBuOH. (D) (6) shows the peak of 2-MeBuOH, though no peak of 2-MeBuOAc. Thus, 2-MeBuOH doesn’t contain 2- MeBuOAc. (E) In (7), no peak of ester is observed. This sample wasn’t added with substrates, so MpAAT1 without substrates didn’t produce esters. From these results, we can conclude that E.Coli with MpAAT1 is able to convert substrate alcohols into esters.
Discussion
From the result of the experiment above, we can conclude that engineered E.coli successfully converted alcohols added as substrates into esters. Moreover, we synthesized MpAAT1 and esters using E.coli BL21 DE3 as a chassis, which was reported to be implausible in former report(s)[1]. In 2008, C.R. Shen and J.C. Liao[2] succeeded in synthesizing butanol from E.coli. If we take advantage of this engineered E.coli, we could produce apple fragrance ester without the addition of substrate.
 "
"