Team:Newcastle/24 August 2010
From 2010.igem.org
(→Results) |
|||
| Line 64: | Line 64: | ||
Please refer to [[Team:Newcastle/Restriction_digests|restriction digests]] and [[Team:Newcastle/Gel_electrophoresis|gel electrophoresis]]. | Please refer to [[Team:Newcastle/Restriction_digests|restriction digests]] and [[Team:Newcastle/Gel_electrophoresis|gel electrophoresis]]. | ||
| - | == | + | ==Discussion== |
| - | + | ||
| - | + | ||
| - | + | ||
| + | Gel electrophoresis will be done on the 25th August, 2010. | ||
=Single digestion of pSB1C3= | =Single digestion of pSB1C3= | ||
Revision as of 03:17, 26 October 2010

| |||||||||||||
| |||||||||||||
Contents |
Miniprep for pGFPrrnB with yneA
Aims
The aim of this experiment is to produce more stocks of yneA in pGFPrrnB.
Materials and Protocol
Please refer to qiagen minipreps and nanodrop spectrophotometer protocols.
Results
| Tube 1 | Tube 2 | Tube 3 | Tube 4 | Tube 5 | Tube 6 | Tube 7 | Tube 8 | Tube 9 | Tube 10 | Tube 11 | Tube 12 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 286.1 µl/ml | 304.0 µl/ml | 316.3 µl/ml | 421.6 µl/ml | 518.7 µl/ml | 460.1 µl/ml | 370.1 µl/ml | 377.3 µl/ml | 346.0 µl/ml | 347.4 µl/ml | 202.8 µl/ml | 307.4 µl/ml |
Table 1: Nanodrop spectrophotometer results. Table represents the amount of plasmid present in µl/ml quantity.
Discussion
The concentration ranged from 286.1 µl/ml to 518.7 µl/ml. Therefore we have obtained high concentration of yneA in pGFPrrnB.
Single and Double Digestion of pGFPrrnB with yneA
Aims
The aim of this experiment is to check if the insert yneA has been inserted into vector pGFPrrnB.
Materials and Protocol
We are doing two digests for pGFPrrnB and yneA:
- Single digest with HinDIII;
- Double digest with EcoR1 and Nhe1.
Please refer to restriction digests and gel electrophoresis.
Discussion
Gel electrophoresis will be done on the 25th August, 2010.
Single digestion of pSB1C3
Aims
To linearise the vector pSB1C3. The linear vector will be used as a PCR template for Gibson cloning of the Subtilin immunity and rocF BioBricks.
Materials and Protocol
We are doing a single digest with HinDIII.
Please refer to restriction digests and gel electrophoresis.
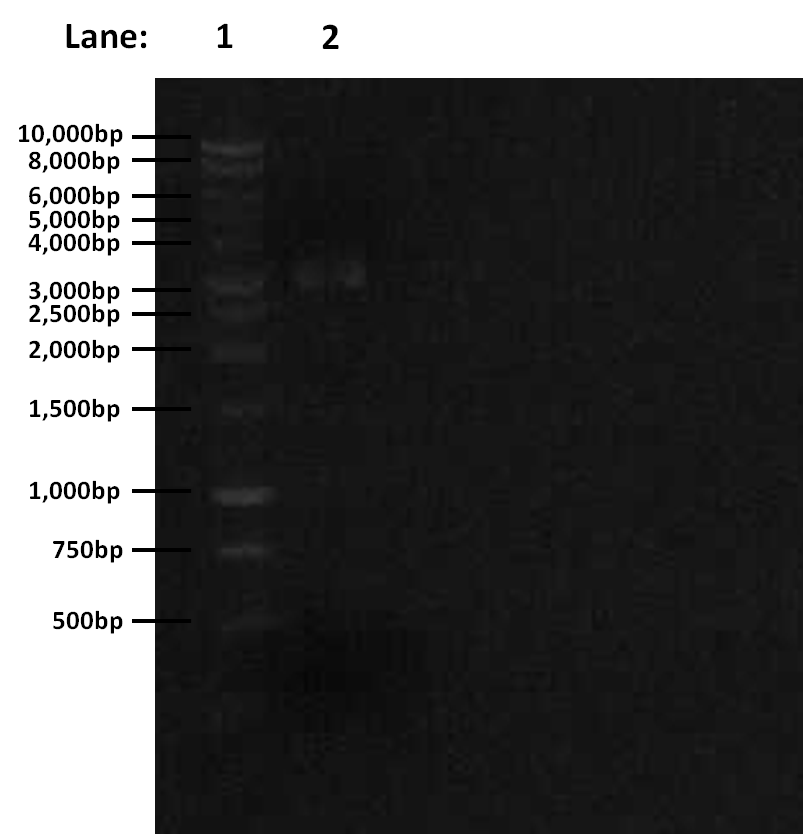
Results
Gel electrophoresis results for digestion:
Figure 1: Gel electrophoresis of the amplified PCR products and restriction digest
- Lane 1: 1 Kb ladder
- Lane 2: digested (linearised) pSB1C3
Discussion and Conclusion
The digestion was successful — the correct band was seen. We can proceed to gel extraction.
 
|
 "
"