Team:Freiburg Bioware/NoteBook/Labjournal/August2
From 2010.igem.org
(→Picking clones from the ViralBricks) |
(→P282/B234 mini-prep) |
||
| Line 1,556: | Line 1,556: | ||
===<p style="font-size:17px; background-color:#00dd77;">104. labday 29.08.2010</p>=== | ===<p style="font-size:17px; background-color:#00dd77;">104. labday 29.08.2010</p>=== | ||
====<p style="font-size:15px; background-color:#66bbff;"><b>P282/B234 mini-prep</b></p>==== | ====<p style="font-size:15px; background-color:#66bbff;"><b>P282/B234 mini-prep</b></p>==== | ||
| + | '''Investigator: Patrick''' | ||
... was performed according to the standard protocol. <br> | ... was performed according to the standard protocol. <br> | ||
Labelling: | Labelling: | ||
* P323, pSB1C3_NLS clone 2.1, 129 ng/µl | * P323, pSB1C3_NLS clone 2.1, 129 ng/µl | ||
* P324, pSB1C3_NLS clone 2.2, 141 ng/µl | * P324, pSB1C3_NLS clone 2.2, 141 ng/µl | ||
| - | |||
====<p style="font-size:15px; background-color:#66bbff;"><b>Digestion of pSB1C3_NLS (P324)</b></p>==== | ====<p style="font-size:15px; background-color:#66bbff;"><b>Digestion of pSB1C3_NLS (P324)</b></p>==== | ||
Revision as of 10:49, 20 September 2010

99. labday 24.08.2010
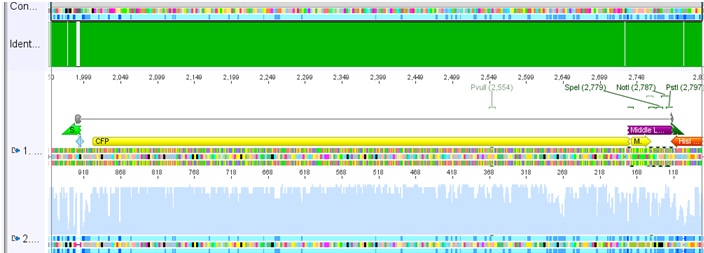
Sequence analysis of pSB1C3_CFP_middlelinker
Investigator: Jessica
- pSB1C3_CFP_middlelinker is ready
Harvest viral particles, Transduction of HT1080
Investigator: Kerstin
- Harvest viral particles following the standard protocol
- Transduction of three 6-well plates:
Plate 1 YFP: 150.000 cells per well
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 500µl virus (1) | 500µl virus (2) |
| B | control, no virus | 500µl virus (1) | 500µl virus (2) |
Plate 2 YFP: 150.000 cells per well
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 500µl virus (3) | 500µl virus (4) |
| B | control, no virus | 500µl virus (3) | 500µl virus (4) |
Plate 3 YFP: 200.000 cells per well
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 500µl virus (5) | 500µl virus (9) |
| B | control, no virus | 500µl virus (9) | 500µl virus (5) |
- (1)= 10µg RC, 10µg pHelper, 10µg GOI (YFP); pH(2xHBS)=7,06
- (2)= 10µg RC, 10µg pHelper, 10µg GOI (YFP); pH(2xHBS)=7,08
- (3)= 10µg RC, 10µg pHelper, 10µg GOI (YFP); pH(2xHBS)=7,10
- (4)= 10µg RC, 10µg pHelper, 10µg GOI (YFP); pH(2xHBS)=7,12
- (5)= 10µg RC, 10µg pHelper, 10µg GOI (YFP); pH(2xHBS)=7,14
- (9)= 10µg RC, 10µg pHelper, 3,3µg GOI (eGFP)
Mini-prep of SDM NgoMIV CD
Investigator: Kira
Motivation: Harvest DNA for further analysis, e.g. test digestion
Mini-prep was performed on 3 colonies.
c(clone1) = 358, 05 ng/ul c(clone2) = 352, 10 ng/ul c(clone 3) = 376, 53 ng/ul
Sent for sequencing: pSB1C3_6xHis
Investigator: Jessica
- Plasmid: P84 c= 107,8 ng/µl
- Primer: VR2
- tube number: JG84
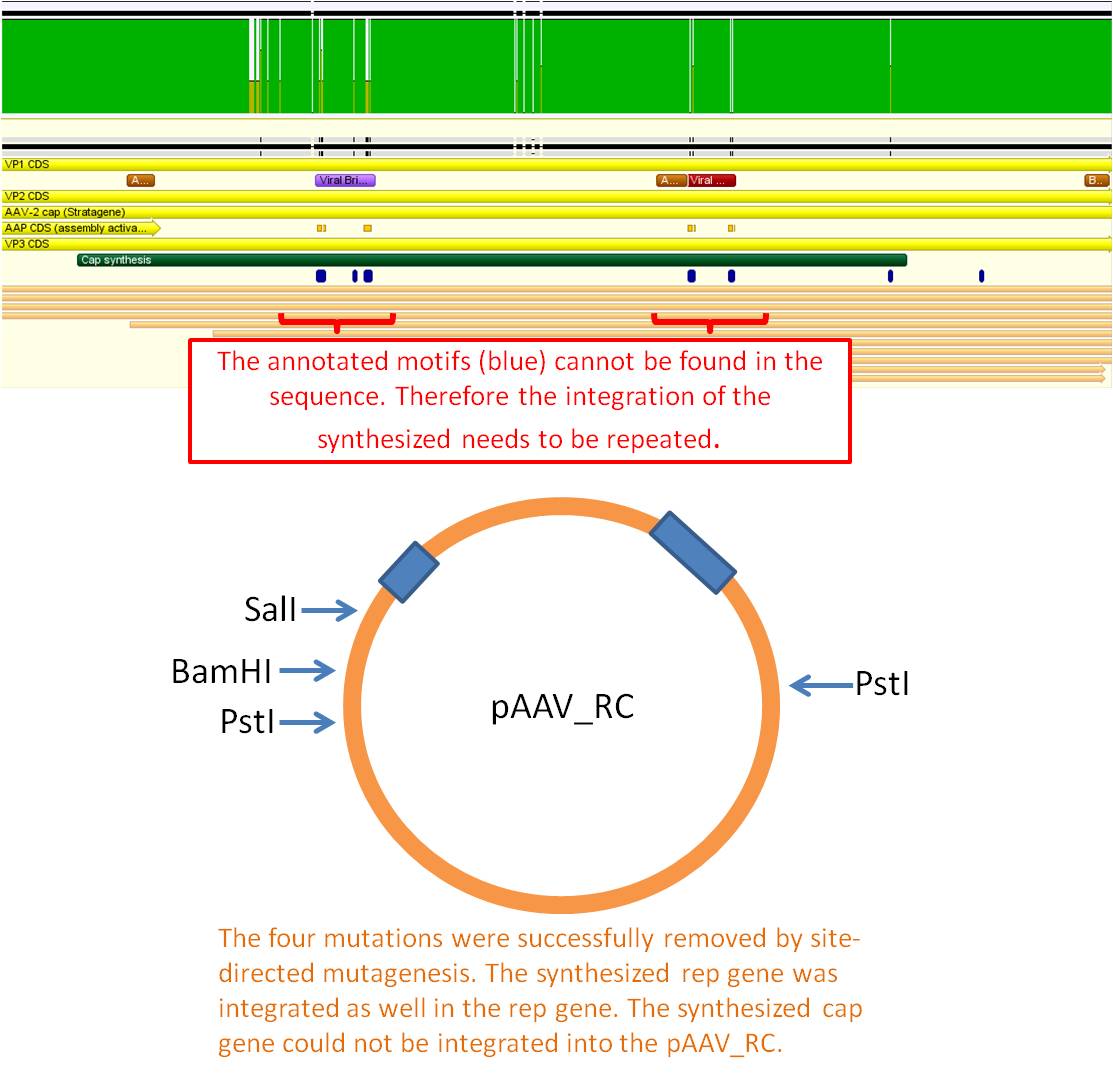
Subcloning Cap into pAAV_RC_ins-rep
Investigator: Stefan
Comment: Sequencing of pAAV_RC_final showed that Cap was not inserted succsessfully. Therefore it has to be repeated.
Digestion of vector and insert:
- Insert: pMA_RepCap Vector_SDM_InsPvuII clone 1 (P211)
- Vector: pAAV_RC_ins-rep clone1 (P250)
A two-step digestion was performed:
1st step
| P211 / µl | P250 / µl | |
| DNA | 5,8 | 5,2 |
| buffer 3 | 2 | 2 |
| Enzyme BsiWI | 1 | 1 |
| H2O | 11,2 | 11,8 |
| total volume | 20 | 20 |
2nd step
| P211 / µl | P250 / µl | |
| Mix obtained from step 1 | 20 | 20 |
| Enzyme BspMI | 1 | 1 |
| H2O | 1,25 | 1,25 |
| total volume | 22,25 | 22,25 |
Samples were loaded on a 0,8% agarose gel an run at 115V for about 60 minutes.
Gelextraction:
Gelex was performed according to protocol exept for elution was done with 60 µl instead of 20 µl.
- c(P211)= 1,14 ng/µl
- c(P250)= 5,60 ng/µl
T4 Ligation:
T4 ligation was performed according to protocol.
used DNA amounts:
- v(P211)= 2,56 µl
- v(P250)= 5,44 µl
Transformation:
Trafo was performed according to protocol using XL1b cells.
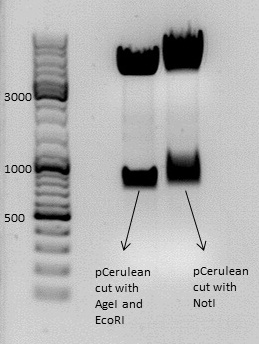
Test digestion of pCerulean (p273)
Investigator: Anissa
Comments:Sequencing of pCerulean showed strange results, because the qualtity of sequenzing was not good. For working further with pCerulean, we started with a test digestion, to be sure, everything works.
Because the sequencing showed no AgeI, we cut one time with AgeI and EcoRI, the other time with NotI.
| Components | p273/µL | p273/µL |
| DNA | 2 | 2 |
| BSA (10x) | 1,5 | 1,5 |
| Buffer 4 (10x) | 1,5 | 1,5 |
| Enzyme 1 | 0,5 NotI | 0,5 EcoRI |
| Enzyme 2 | 0,5 NotI | 0,5 AgeI |
| H2O | 9 | 9 |
| Total volume | 15 | 15 |
Comments: Results of digestion seem to be all right
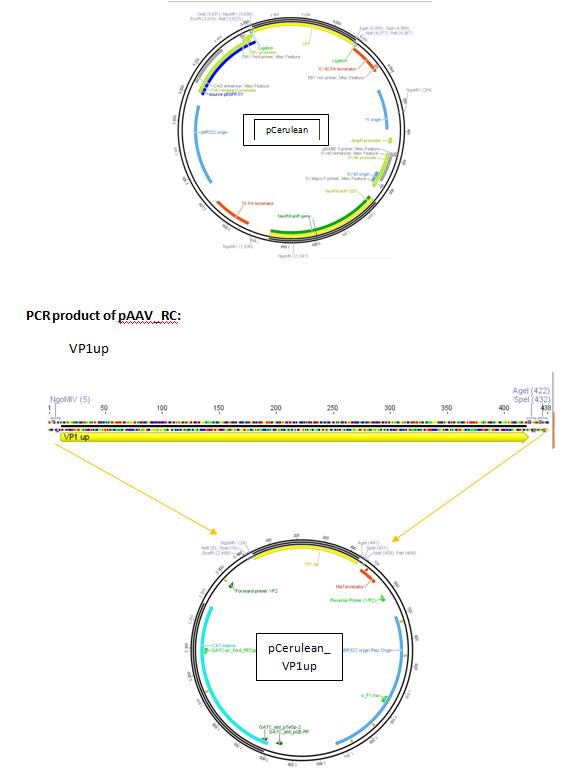
Cloning of VP1up into PCerulean
Investigator: Anissa
VP1up was cut out of pSB1C3 and into pCerulean
- Digestion:
Components Vector/µL Insert/µL DNA 3,7 8,4 BSA (10x) 1,5 2 Buffer 4 (10x) 1,5 2 Enzyme 1 EcoRI 1 1 Enzyme 2 PstI 1 1 H2O 6,3 5,6 Total volume 15 20
- Gel:
A 1% agarose-gel was made, after 30 minutes samples were cut

- Gelextraction was performed according the standardprotocol
- Ligation:
Components used volume for T4 ligation/µL concentration /ng/µl Vector 5,15 27,7 Insert 2,85 15,4 In addition 1µl T4-ligase and 1µl T4-ligase-buffer were added
- Transformation: was performes into Xl1 blue according the standard-protocol
Sequence analysis of sequencing reads prepared yesterday (23.08.2010)
Investigator: Bea
GENERAL COMMENT: pSB1C3 {NLS and VP1up} were sent for sequencing. Aswell as the pAAV_RC_final which contains all four mutations in the Rep-Cap gene and the integrated and synthesiezd rep and cap gene and the plasmid pCerulean whith the CMV promoter and sv40 polyadenylation site.
Comments: Sequence analysis of pAAV_RC_final containing the subcloned "rep" (ordered) and "Cap (ordered) plus the four mutations to delete the restriction sites PstI, BamHi and SalI.
- Primer used:
- VP1 primer for pKex
- GATC_std_SK
- Primer Cap 2800 rev
- Primer Cap 2800 for
- Primer Cap 3500 for
- Plasmid sequenced: P283
- Sequence sample: iGEM4
- Stored in Geneious-folder: RepCap insert ordered
Sequencing of the mutation ok. Rep integration worked. Cap integration needs to be repeated.
Next steps: Repetition of the integration of the synthesized cap gene performed by Stefan (see topic).
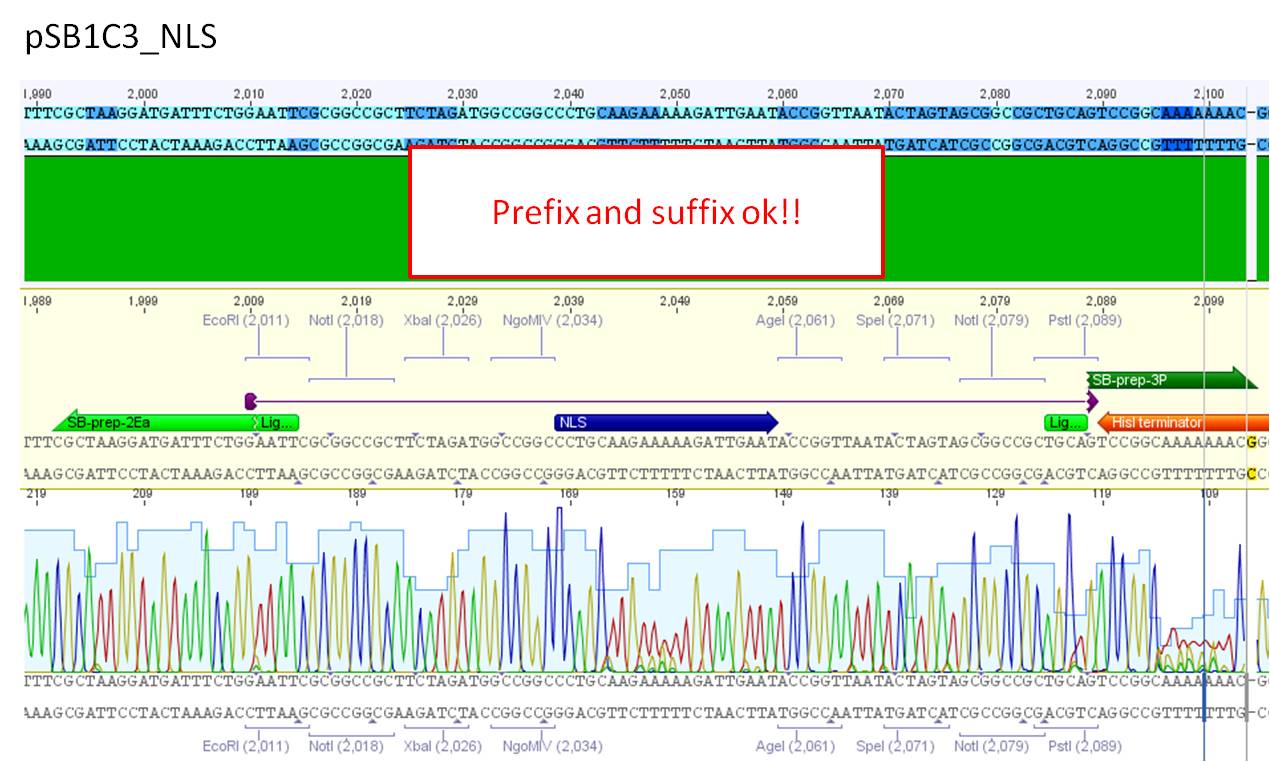
Comments: Sequence analysis of pSB1C3_NLS
- Primer used: VR-2
- Plasmid sequenced: P282
- Sequence sample: iGEM3_O51_VR-2
- Stored in folder: pSB1C3_NLS
Insertion of the nuclear localisation signal (NLS) into the pSB1C3_NLS as one step in the N-terminal insertion of targeting molecules. Sequencing results look good. prefix and suffix ok!
Next steps: Fuse NLS to targeting molecule.
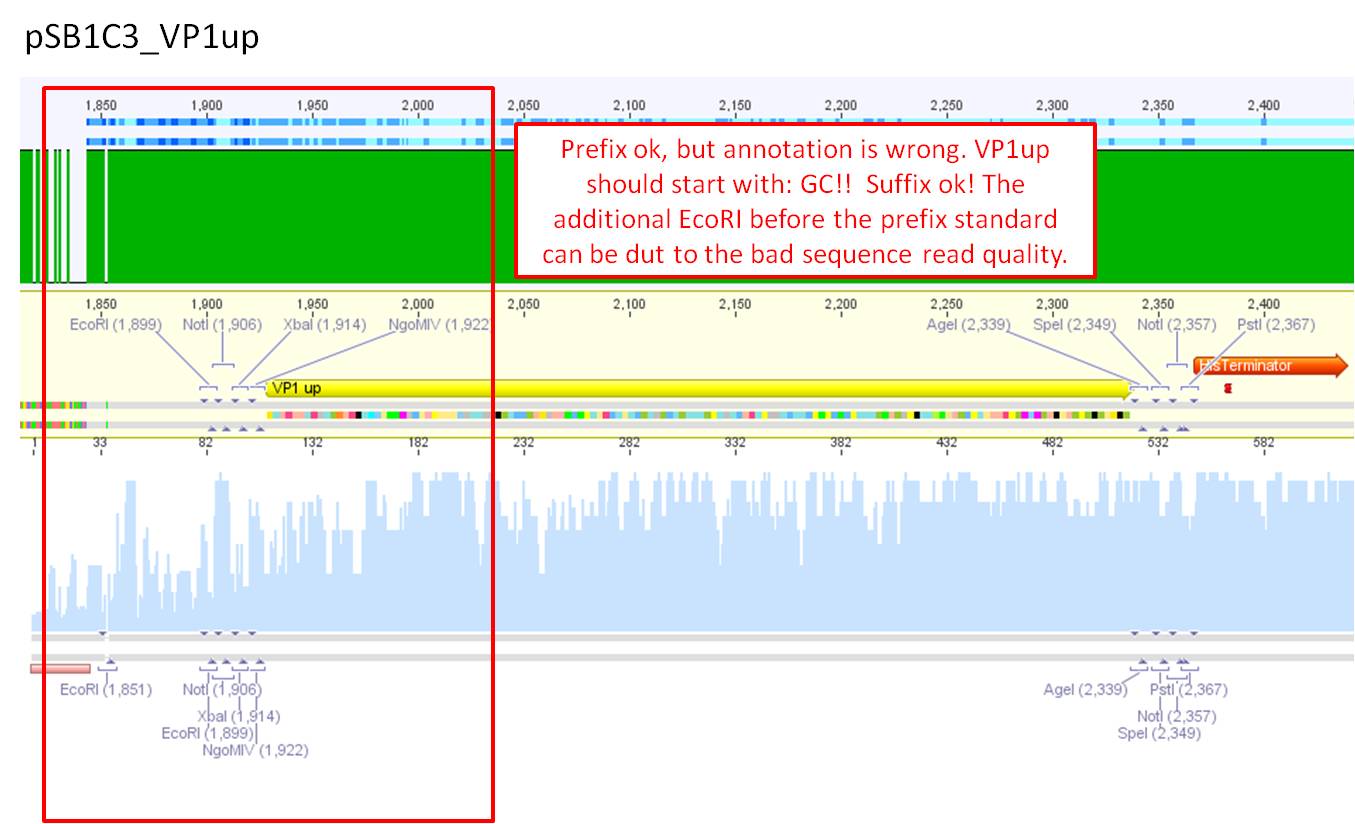
Comments: Sequence analysis of pSB1C3_VP1up
- Primer used: VF-2
- Plasmid sequenced: P280
- Sequence sample: iGEM1_VF-2
- Stored in folder: N-Terminal targeting --> pSB1C3_VP1up
Prefix ok, but annotation is wrong. VP1up should start with: GC!! Suffix ok! The additional EcoRI before the prefix standard can be due to the bad sequence read quality.
Next steps: Clone VP1up into pCerulean in order to obtain the plasmid with a CMV promoter and the sv40 polyadenylation site.
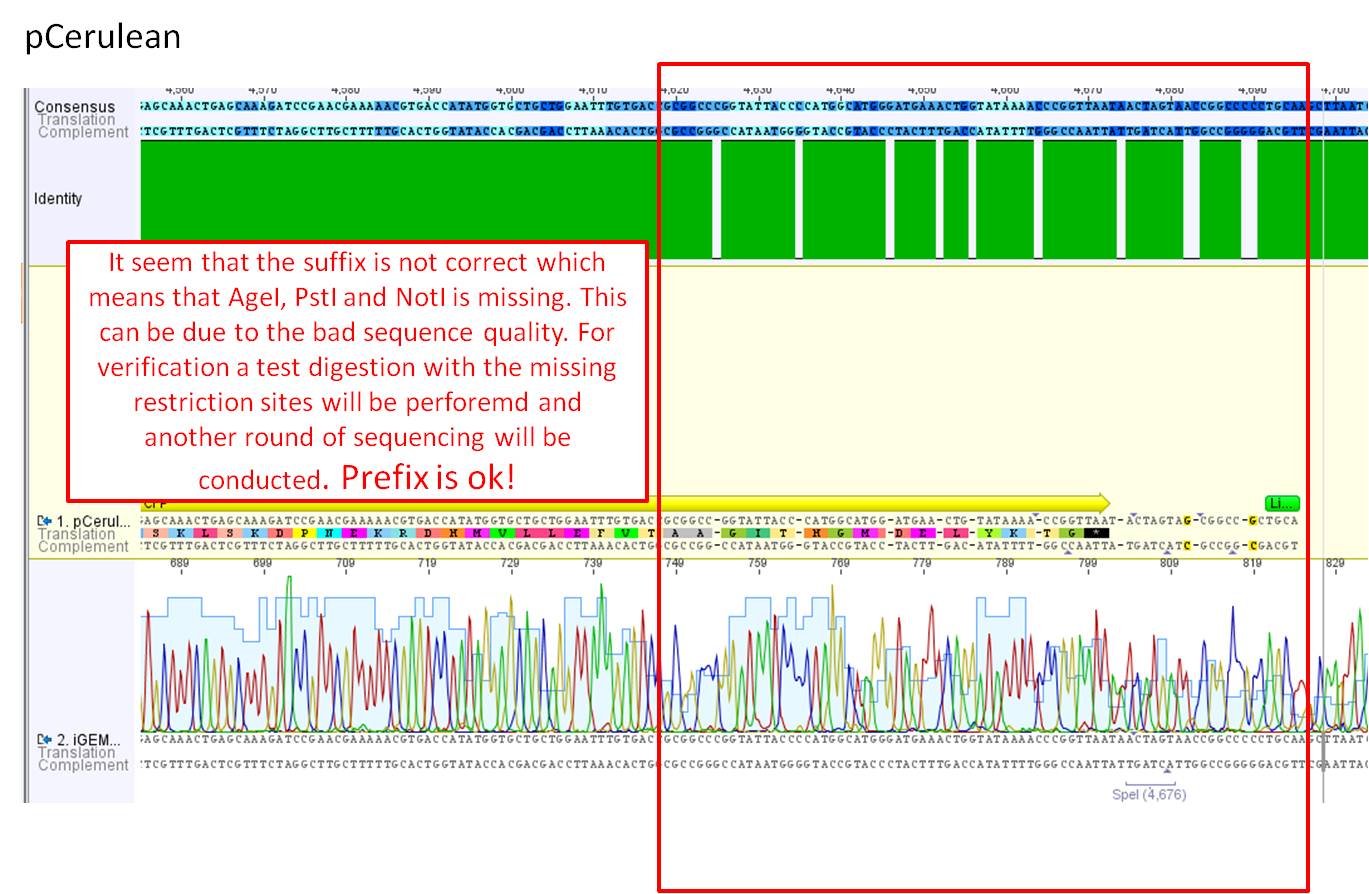
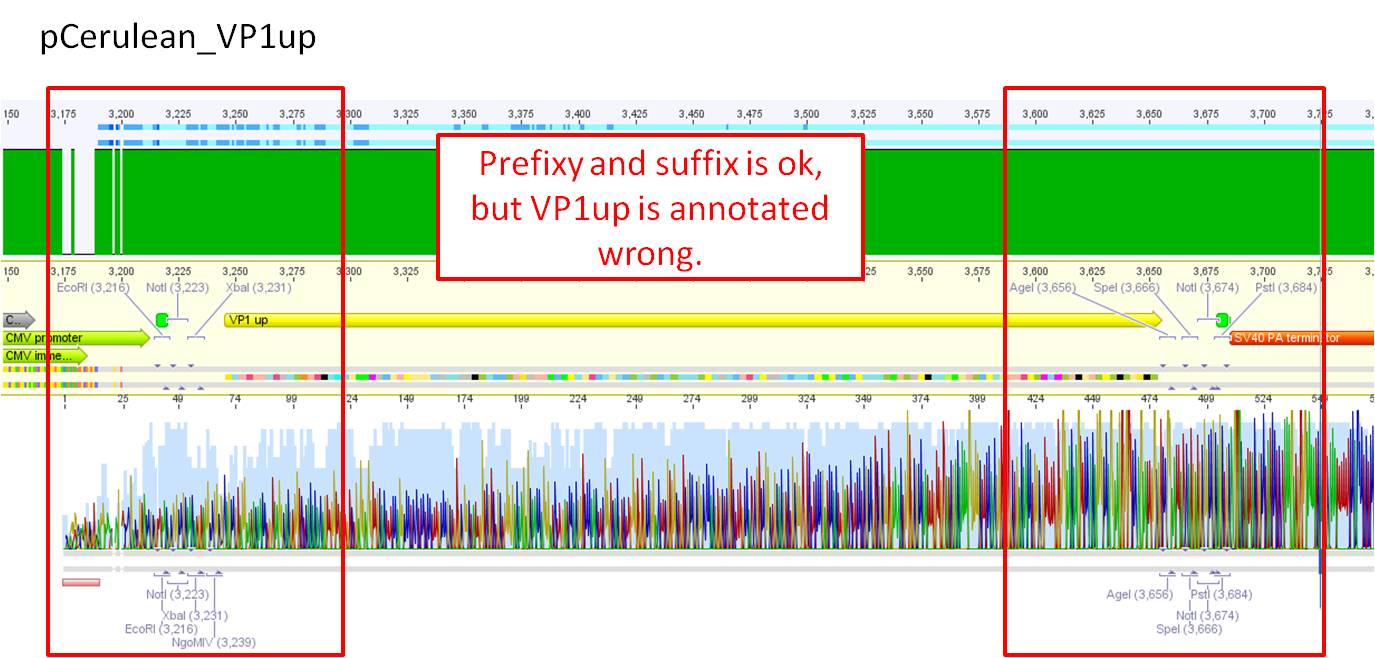
Comments: Sequence analysis of pCerulean
- Primer used: GATC_CMV-F
- Plasmid sequenced: P273
- Sequence sample: iGEM2_CMV-F
- Stored in folder: N-Terminal targeting --> pCerulean
It seem that the suffix is not correct which means that AgeI, PstI and NotI is missing. This can be due to the bad sequence quality. Prefix is ok!
Next steps: For verification a test digestion with the missing restriction sites will be perforemd and another round of sequencing will be conducted.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
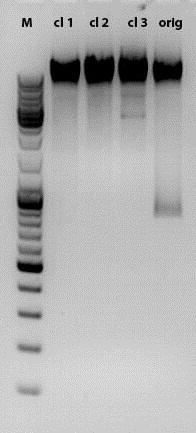
Test digestion of SDM NgoMIV CD
Investigator: Kira
Motivation: In order to check if site directed mutagenesis was successfull, test digestion was performed with 3 clones, which were picked yesterday as well as with 'original' DNA, which still contains all the restriction sites
| Components | clone 1/µL | clone 2/µL | clone 3/µL | original/µL |
| DNA (500ng) | 1,4 | 1,4 | 1,4 | 1,4 |
| BSA (10x) | 1 | 1 | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 | 1 | 1 |
| Enzyme 1 NgoMIV | 0,5 | 0,5 | 0,5 | 0,5 |
| H2O | 6,1 | 6,1 | 6,1 | 6,1 |
| Total volume | 10 | 10 | 10 | 10 |
According to the gel, NgoMIV SDM was successful in clones 1 and 2. Clone 3 well reveales 2 bands of unknown origin. Samples 1 and 3 were sent for sequencing.
Picking clones of pSB1C3_Affibody_linker and pSB1C3_ß-Globin_YFP_hGH_rITR
Investigator: Anna
Comments: A 1:1000 dilution of pSB1C3_Affibody_GSAT-Linker was prepared.
To do: Mini-Prep of pSB1C3_Affibody_(Short/Middle/Long and SEG-Linker) and pSB1C3_ß-Globin_YFP_hGH_rITR. Picking clones of pSB1C3_Affibody_GSAT-Linker.
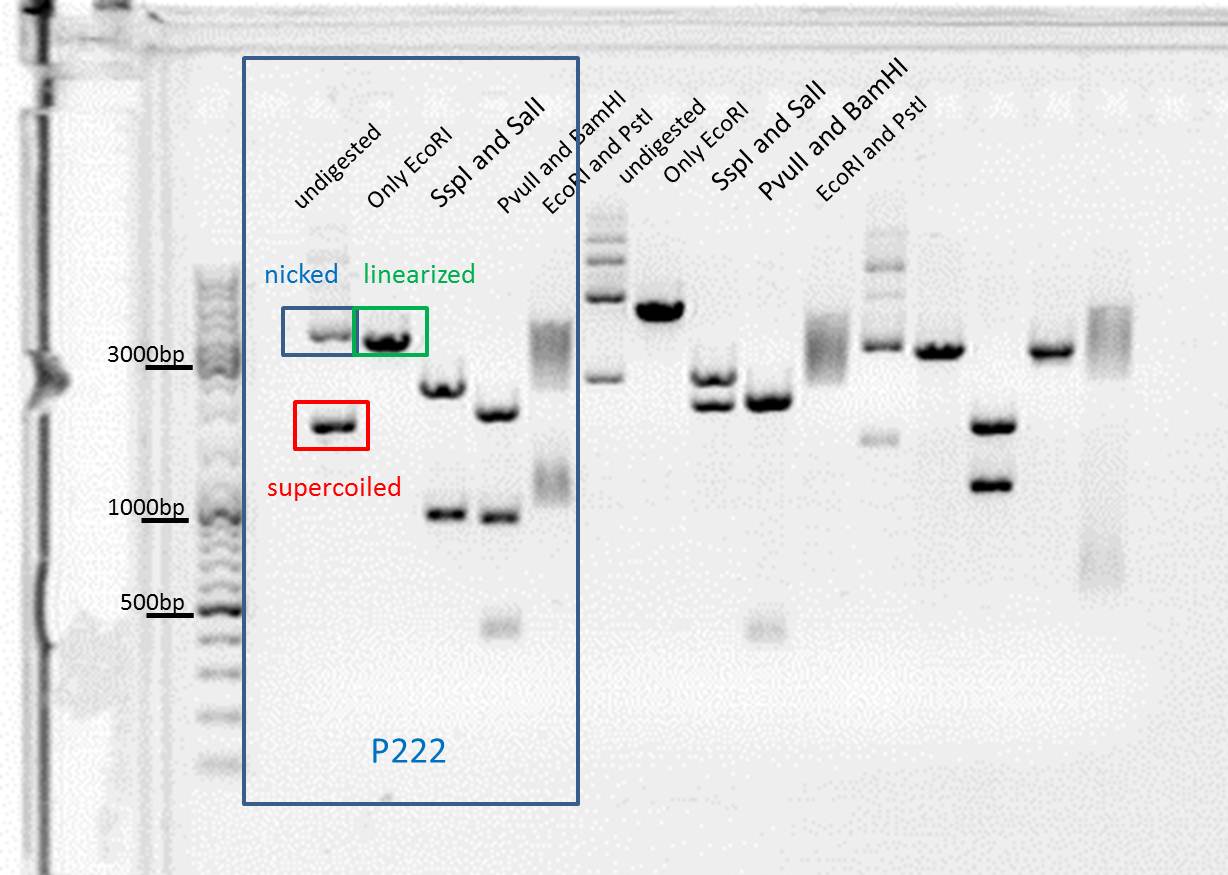
Test digestion of pSB1C3_SDM_SspI_Bla14FM (P222), pSB1C3_BLA (P286, P288)
Investigator: Achim
CommentsNew test digestion of two SDM-attempts, this time we also digested the original vector containing the PvuII restriction site to see differences.
- The original vector is being cut as expected: the uncut vector can be seen in different conformations, the linearized vector shows one distinct band and the digestions cutting out BLA show a band at ~900 bp. Our SDM attempts show inconsistent results, not only does the Pvu site still seem to be in the backbone, there are no more proper insert bands visible either. We therefore conclude that either the DpnI digestion or the dilution of the transformation must have gone wrong (differing plasmids...). Tomorow we'll try a new cloning approach using an old vector with deleted Pvu restriction site and MscI & XbaI digestion. In case this also fails we also yet have to prep and test digest the repetition of the mutagenesis which was carried out by Stefan.
Prefix ok, but annotation is wrong. VP1up should start with: GC!! Suffix ok! The additional EcoRI before the prefix standard can be due to the bad sequence read quality.
Next steps: Clone VP1up into pCerulean in order to obtain the plasmid with a CMV promoter and the sv40 polyadenylation site.
Comments: Sequence analysis of pCerulean
- Primer used: GATC_CMV-F
- Plasmid sequenced: P273
- Sequence sample: iGEM2_CMV-F
- Stored in folder: N-Terminal targeting --> pCerulean
It seem that the suffix is not correct which means that AgeI, PstI and NotI is missing. This can be due to the bad sequence quality. Prefix is ok!
Next steps: For verification a test digestion with the missing restriction sites will be perforemd and another round of sequencing will be conducted.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
Test digestion of SDM NgoMIV CD
Investigator: Kira
Motivation: In order to check if site directed mutagenesis was successfull, test digestion was performed with 3 clones, which were picked yesterday as well as with 'original' DNA, which still contains all the restriction sites
| Components | clone 1/µL | clone 2/µL | clone 3/µL | original/µL |
| DNA (500ng) | 1,4 | 1,4 | 1,4 | 1,4 |
| BSA (10x) | 1 | 1 | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 | 1 | 1 |
| Enzyme 1 NgoMIV | 0,5 | 0,5 | 0,5 | 0,5 |
| H2O | 6,1 | 6,1 | 6,1 | 6,1 |
| Total volume | 10 | 10 | 10 | 10 |
According to the gel, NgoMIV SDM was successful in clones 1 and 2. Clone 3 well reveales 2 bands of unknown origin. Samples 1 and 3 were sent for sequencing.
Picking clones of pSB1C3_Affibody_linker and pSB1C3_ß-Globin_YFP_hGH_rITR
Investigator: Anna
Comments: A 1:1000 dilution of pSB1C3_Affibody_GSAT-Linker was prepared.
To do: Mini-Prep of pSB1C3_Affibody_(Short/Middle/Long and SEG-Linker) and pSB1C3_ß-Globin_YFP_hGH_rITR. Picking clones of pSB1C3_Affibody_GSAT-Linker.
Test digestion of pSB1C3_SDM_SspI_Bla14FM (P222), pSB1C3_BLA (P286, P288)
Investigator: Achim
CommentsNew test digestion of two SDM-attempts, this time we also digested the original vector containing the PvuII restriction site to see differences.
- The original vector is being cut as expected: the uncut vector can be seen in different conformations, the linearized vector shows one distinct band and the digestions cutting out BLA show a band at ~900 bp. Our SDM attempts show inconsistent results, not only does the Pvu site still seem to be in the backbone, there are no more proper insert bands visible either. We therefore conclude that either the DpnI digestion or the dilution of the transformation must have gone wrong (differing plasmids...). Tomorow we'll try a new cloning approach using an old vector with deleted Pvu restriction site and MscI & XbaI digestion. In case this also fails we also yet have to prep and test digest the repetition of the mutagenesis which was carried out by Stefan.
100. labday 25.08.2010
Cloning of pSB1C3_Rep40, pSB1C3_Rep68 and pMA_RC_insert
Investigator: Chris L.
- buffer used: 2; Restriction-enzymes used: Enzyme 1: HindIII ; Enzyme 2: BstEII
Plasmids:
- pSB1C3_Rep40 P231
- pSB1C3_Rep68 P266
- pMA_RC_insert P190
Insert:
| Components | P190/µL | P231/µL | P266/µL |
| DNA | 5.9 | 6.4 | 5 |
| BSA (10x) | 2 | 2 | 2 |
| Buffer 2 (10x) | 2 | 2 | 2 |
| Enzyme 1 HindIII | 1 | 1 | 1 |
| Enzyme 2 BstEII | 1 | 1 | 1 |
| H2O | 8.1 | 7.6 | 9 |
| Total volume | 20 | 20 | 20 |
- Incubation: 90 minutes; 37°C with HindIII
- Incubation: 90 minutes; 60°C with BstEII
Agarosegel
0.5 g Agarose, 50 ml TAE (1 %), 3 µL GELRED, 5 min at 90 Volt, 40 min at 115 Volt
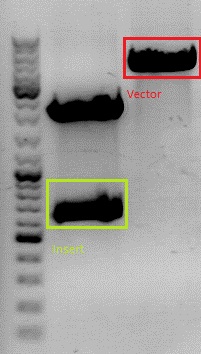
Concentrations measured via NanoDrop:
- pSB1C3_Rep40: 81.97 ng/µl
- pSB1C3_Rep68: 48,68 ng/µl
- RC_Insert: 3,60 ng/µl
T4 Ligation of pSB1C3_Rep40 with RC_Insert
Volume insert: 6,54 µl
Volume vector: 1,46 µl
T4 Ligation of pSB1C3_Rep68 with RC_Insert
Volume insert: 5,46 µl
Volume vector: 2,54 µl
Trafo was prepared with XL1blue and Cm
Picking clones of pAAV_RC-ins_rep_cap and pCeruleanVP1up
Investigator: Chris L.
To do: Mini-Prep of pAAV_RC-ins_rep_cap and pCeruleanVP1up.
Cloning of pSB1C3_6xHis with middlelinker and pSB1C3_6xHis with pGA14_mVenus_YFP
Investigator: Jessica
- buffer used: 4; Restriction-enzymes used: Enzyme 1: AgeI ; Enzyme 2: PstI ; Enzyme3: NgoMIV
- Plasmids:
- pSB1C3_6xHis P84
- pGA14_middle linker P65
- GA14_mVenus_YFP P60
| Components | Mastermix | P84/µL | P65/µL | P60/µL |
| DNA | - | 13,91 | 13,52 | 8,93 |
| BSA (10x) | - | 2 | 2 | 2 |
| Buffer 4 (10x) | - | 2 | 2 | 2 |
| Enzyme 1 AgeI | - | 1 | - | - |
| Enzyme 2 PstI | - | 1 | 1 | 1 |
| Enzyme 3 NgoMIV | - | - | 1 | 1 |
| H2O | - | 0,09 | 0,48 | 5,07 |
| Total volume | - | 20 | 20 | 20 |
- Incubation:110 minutes; 37°C
Agarosegel
0.5 g Agarose, 50 ml TAE (1 %), 3 µL GELRED, at 120 Volt
concentrations measured via NanoDrop:
- SB1C3_6xHis: 1,7 ng/µl
- middle linker: 3,8 ng/µl
- mVenus_YFP: 3,2 ng/µl
T4 Ligation pSB1C3_6xHis_middlelinker
Volume insert: 0,25 µl
Volume vector: 7,75 µl
T4 Ligation pSB1C3_6xHis_mVenus_YFP
Volume insert: 2,64 µl
Volume vector: 5,36 µl
Trafo was prepared with XL1blue and Cm
Sequence analysis of pSB1C3_6xHis
Investigator: Jessica
P84 was sequenced for continuation of working with it
- pSB1C3_6xHis P84 is ready to use, but is empty. a new tube will be made tomorrow
Inoculation of pGA14_middlelinker and pSB1C3_6xHis clone1
Investigator: Jessica
- both tubes (P65 and P84)are empty --> inoculation with glycerolstocks
- B48 pGA14_middlelinker was inoculated with 10ml DYT and 10µl Amp
- B64 pSB1C3_6xHis clone1 was inoculated with 10ml DYT and 10µl Cm
Cloning of pSB1C3_SspI_BLA and pSB1C3_CFP_PvuII
Investigator: Achim
Comment:I ligated a sequence from pSB1C3_CFP_PvuII missing the PvuII restriction site into pSB1C3_SspI_BLA to obtain a pSB1C3 vector without PvuII in the cat marker.
Vector:
- pSB1C3_SspI_BLA (p222): c= 308,32 ng/µl
Insert:
- pSB1C3_CFP_PvuII (p129): c= 264,86 ng/µl
| components | Vector | Insert |
| DNA | 3,8 | 4,9 |
| BSA (10x) | 2 | 2 |
| Buffer 4 (10x) | 2 | 2 |
| Enzyme MscI | 1 | 1 |
| Enzyme XbaI | 1 | 1 |
| H2O | 10,2 | 9,1 |
| Total volume | 20 | 20 |
- Gelextraction:
0,5 g Agarose,50 ml TAE (1%), 3 µl GELRED , at 110 Volt, running time:45
Marker: GeneRuler ladder mix (Fermentas)
- c(Insert)= 15,66 ng/µl; size: 730 bp
- c(Vector)= 14,42 ng/µl; size: 2200 bp
- Ligation of PCR products and vector:
For the Ligation 1µl T4 buffer (2x) and 1µl T4 ligase were used. Incubation time: 60 min due to blunt end ligation
| vector /µl | insert /µl | |
| pSB1C3_BLA | 0,86 | 7,14 |
- Transformation:
The transformation was done following the standard protocol using XL1 blue cells.
Comment: Two clones were picked, but because we already obtained our plasmid via SDM in the meantime, no prep was performed. Glycerol stocks of the two clones were created just in case, B264 and B265.
Mini-Preps and test digestion of pSB1C3_Affibody_Middle-Linker, pSB1C3_Affibody_Short-Linker, pSB1C3_Affibody_SEG-Linker, pSB1C3_Affibody_Long-Linker and pSB1C3_betaglobin_mVenus_hGH_rITR
Investigator: Stefan
-
Glycerol stocks were prepared:
- B240 = pSB1C3_Affibody_Middle-Linker clone1
- B241 = pSB1C3_Affibody_Middle-Linker clone2
- B242 = pSB1C3_Affibody_Short-Linker clone 1
- B243 = pSB1C3_Affibody_Short-Linker clone 2
- B246 = pSB1C3_Affibody_SEG-Linker clone1
- B247 = pSB1C3_Affibody_SEG-Linker clone2
- B248 = pSB1C3_Affibody_Long-Linker clone 1
- B249 = pSB1C3_Affibody_Long-Linker clone 2
- B244 = pSB1C3_betaglobin_mVenus_hGH_rITR clone 1
- B245 = pSB1C3_betaglobin_mVenus_hGH_rITR clone 2
- P290 = pSB1C3_Affibody_Middle-Linker clone1 c= 227,36 ng/µl
- P291 = pSB1C3_Affibody_Middle-Linker clone2 c= 219,05 ng/µl
- P292 = pSB1C3_Affibody_Short-Linker clone 1 c= 196,63 ng/µl
- P293 = pSB1C3_Affibody_Short-Linker clone 2 c= 224,76 ng/µl
- P296 = pSB1C3_Affibody_SEG-Linker clone1 c= 218,68 ng/µl
- P297 = pSB1C3_Affibody_SEG-Linker clone2 c= 229,66 ng/µl
- P298 = pSB1C3_Affibody_Long-Linker clone 1 c= 229,73 ng/µl
- P299 = pSB1C3_Affibody_Long-Linker clone 2 c= 202,05 ng/µl
- P294 = pSB1C3_betaglobin_mVenus_hGH_rITR clone 1 c= 425,14 ng/µl
- P295 = pSB1C3_betaglobin_mVenus_hGH_rITR clone 2 c= 328,28 ng/µl
Mini-Prep following the standard protocol
Test digestion
- Restriction-enzymes used: for P290-P293 and P296-P299: NotI ; for P294-P295: NgoMIV and PstI
comment: The same amount of ingredients were used for P290-P293 and P296-P299, therefore these approaches will be merged into the chart. The same goes for P294-P295.
| Components | P290-P293 and P296-P299 | P294-P295 |
| DNA | 1 | 1 |
| BSA (10x) | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 |
| NotI | 0,5 | - |
| NgoMIV | - | 0,5 |
| PstI | - | 0,5 |
| H2O | 6,5 | 6 |
| Total volume | 10 | 10 |
Incubation time: 70 minutes; Incubation temperature: 37°
0,5 g Agarose,50 ml TAE (1%), 3 µl GELRED , running time:5 minutes at 90 Volt and 50 minutes at 115 Volt
2µl loading dye (6x) for the sample, Marker: GeneRuler ladder mix (Fermentas)

Comment: Sizes of fragments look like expected. Clone 1 of each plasmid will be sent for sequencing tomorrow.
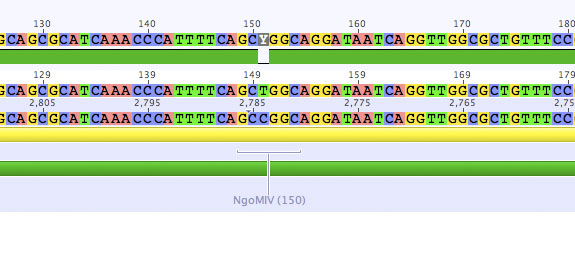
Sequenzing evaluation of SDM NgoMIV
Investigator: Kira
2 samples have been sent for sequencing yesterday. According to the data, both samples do not contain any NgoMIV restriction site anymore.
101. labday 26.08.2010
Mini-Preps of pGA14_middle linker and pSB1C3_6xHis clone 1
Investigator: Jessica
-
Glycerol stocks were not prepared because I have inoculated from glycerol stocks
- P301 = pGA14_middle linker (= P65)
c=305,7 - P300 = pSB1C3_6xHis clone 1 (= P84)
c=177,5
Mini-Prep following the standard protocol
Mini-Preps and test digestion of pAAV_RC-ins_rep_cap, pCeruleanVP1up and pAAV_RC_ins-rep-cap.
Investigator: Chris L.
-
Glycerol stocks were prepared:
- B250 = pCerulean_VP1up clone1
- B251 = pCerulean_VP1up clone2
- B252 = pCerulean_VP1up clone3
- B253 = pCerulean_VP1up clone4
- B254 = pSB1C3_VCK_Bla clone1
- B255 = pSB1C3_VCK_Bla clone2
- B256 = pSB1C3_VCK_Bla clone3
- B257 = pSB1C3_VCK_Bla clone4
- B262 = pAAV_RC_ins-rep-cap clone1
- B263 = pAAV_RC_ins-rep-cap clone2
- P302 = pCerulean_VP1up clone1
c=410,70 ng/µl - P303 = pCerulean_VP1up clone2
c=387,78 ng/µl - P304 = pCerulean_VP1up clone3
c=321,04 ng/µl - P305 = pCerulean_VP1up clone4
c=350,34 ng/µl - P306 = pSB1C3_VCK_Bla clone1
c=82,53 ng/µl - P307 = pSB1C3_VCK_Bla clone2
c=92,70 ng/µl - P308 = pSB1C3_VCK_Bla clone3
c=82,22 ng/µl - P309 = pSB1C3_VCK_Bla clone4
c=105,34 ng/µl - P314 = pAAV_RC_ins-rep-cap clone1
c=517,04 ng/µl - P315 = pAAV_RC_ins-rep-cap clone2
c=444,33 ng/µl
Mini-Prep following the standard protocol
Test digestion:
| components | volume of P302/µl | volume of P303/µl | volume of P304/µl | volume of P305/µl | volume of P306/µl | volume of P306/µl | volume of P306µl | volume of P307/µl | volume of P307µl | volume of P307/µl | volume of P308/µl | volume of P308/µl | volume of P308/µl | volume of P309/µl | volume of P309/µl | volume of P309/µl | volume of P314/µl | volume of P315/µl |
| DNA | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 1 |
| BSA (10x) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | - | - |
| Buffer 2 (10x) | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 1 | 1 |
| Enzyme NotI | - | - | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - | - | - |
| Enzyme SspI | - | - | - | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - | - |
| Enzyme SalI | - | - | - | - | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - | 0,5 | - | - |
| Enzyme BamHI | - | - | - | - | - | - | 0,5 | - | - | - | - | - | - | - | - | - | - | - |
| Enzyme PvuII | - | - | - | - | - | - | 0,5 | - | - | - | - | - | - | - | - | - | - | - |
| Enzyme Acc65I | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0,75 | 0,75 |
| Enzyme XcmI | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0,5 | 0,5 |
| Enzyme PstI | 0,5 | 0,5 | 0,5 | 0,5 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Enzyme EcoRI | 0,5 | 0,5 | 0,5 | 0,5 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| H2O | 6 | 6 | 6 | 6 | 5,5 | 5 | 5 | 5,5 | 5 | 5 | 5,5 | 5 | 5 | 5,5 | 5 | 5 | 5,75 | 5,75 |
| Total volume /µl | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
Incubation time: 1 h, Incubation temperature: 37°
Preparation of gel:
1 g Agarose, 100 ml TAE (1%), 6 µl GELRED , at 115 Volt, running time: 50 minutes
pSB1C3_VCK_Bla: Test digestion looks like expected. Clone 4 was sent for sequencing.
pCerulean_VP1up: Gel looks well as well. clone 2 was sent for sequencing.
pAAV_RC_ins-rep-cap: Test digestion looks strange. Just one big band with more than 10000 bp and one very small with 150 bp. Maybe the restriction enzymes didn´t cut. (@Christian: please insert picture)
102. labday 27.08.2010
Sequence analysis of pSB1C3_Bla_final (P309)and pCerulean_VP1up (P303)
Investigator: Bea
GENERAL COMMENT: pSB1C3_Bba_final (P309, SB_9) and pCerulean_Vp1up (P303; SB_10) were sent for sequencing. In the case of SB1C3_Bba_final, which means that the whole vector was assembled from the buttom,the purpose was to test the designed primers. In the other case we wanted to verify the insertion of Vp1up inot pCerulean.
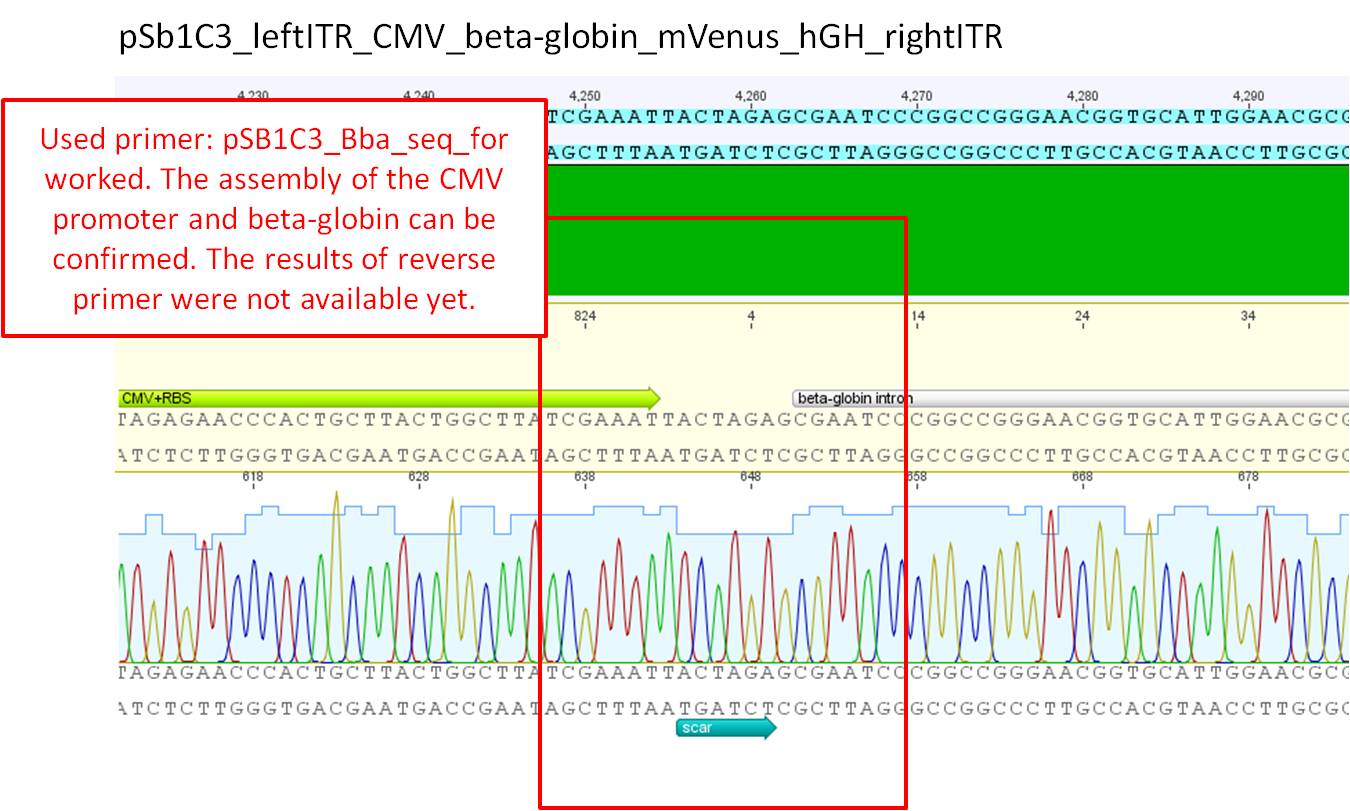
Comments: Sequence analysis of pSB1C3_leftITR_CMV_betaglobin_mVenus_hGH_rightITR.
- Primer used:
- pSB1C3_Bba_seq_for
- pSB1C3_Bba_seq_rev
- Plasmid sequenced: P??
- Sequence sample: ??
- Stored in Geneious-folder: BioBricks --> final parts
Results look good. The forward primer worked, still waiting for the results of the reverse primer. The scar inbetween the CMV promoter and the beat globin intron corresponds to the expected result.
Next steps: Wwait until sequencing results of the reverse primer can be confirmed aswell. If that will not be the case, order new sequensing primer.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
Comments: Sequence analysis of pCerulean_Vp1up.
- Primer used: CMV-F
- Plasmid sequenced: P303
- Sequence sample: ??
- Stored in Geneious-folder: N-terminal Targeting --> pCerulean_Vp1up
Results are good. Insertion of the Vp1up region can be confirmed.
Next steps: Fuse pCerulean_Vp1up to the NLS.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
Hybridisation and cloning of loop insertions
Investigator: Achim, Volker
We hybridized the oligos for the different loop insertions and cloned them into the pSB1C3_BLA backbone.
Hybridisation
- 4 Inserts contained overlapping ends and had to be filled up using Klenov fragments. We therefore added Klenow buffer and dNTPs to those samples.
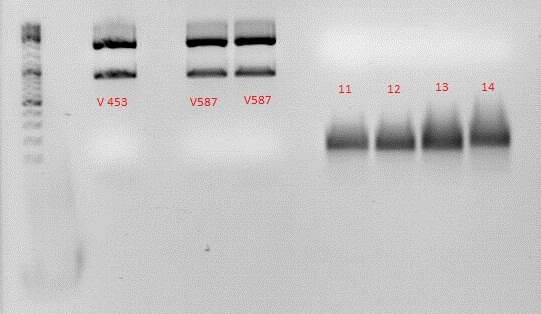
| Components | 453 BAP | 587 BAP | 587 KO BAP | 453 RGD | 587 RGD | 587 KO RGD | 453 HIS | 587 HIS | 587 KO HIS | 587 KO EMPTY | 453 Z34C | 587 Z34C | 587 KO Z34C | 587 KO Z34C SPACER |
| Oligo 1 | O124: 10 | O126: 10 | O128: 10 | O130: 10 | O132: 10 | O134: 10 | O135: 10 | O137: 10 | O139: 10 | O141: 10 | O143: 10 | O145: 10 | O147: 10 | O149: 10 |
| Oligo 2 | O125: 10 | O127: 10 | O129: 10 | O131: 10 | O133: 10 | O151: 10 | O136: 10 | O138: 10 | O140: 10 | O142: 10 | O144: 10 | O146: 10 | O148: 10 | O150: 10 |
| TrisHCl pH8 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | - | - | - | - |
| 5mM MgCl2 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | - | - | - | - |
| Klenow Buffer | - | - | - | - | - | - | - | - | - | - | 4 | 4 | 4 | 4 |
| dNTP Mix | - | - | - | - | - | - | - | - | - | - | 1 | 1 | 1 | 1 |
| H2O | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 8 | 15 | 15 | 15 | 15 |
| Total volume | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 | 40 |
- Hybridisation was carried out according to standard protocoll
- Klenov fill-in reaction:
- Added 1 µl of NEB Klenow fragment to samples 11,12,13,14
- incubated for 1h @ 37°C
Digestion of pSB1C3_BLA vector and Samples 11-14
- We digested the standard vector with the 453 (Ssp/Sal) and the 587 (Bam/Pvu) standard. The samples that were filled in were also digested to create sticky ends.
| Components | V453 | V587 | 11 | 12 | 13 | 14 |
| DNA | 2,5 | 2,5 | 3,5 | 3,5 | 3,5 | 3,5 |
| BSA (10x) | - | - | - | - | - | - |
| Buffer 4 (10x) | 2 | 2 | 2 | 2 | 2 | 2 |
| Enzyme 1 | Ssp: 1 | Bam: 1 | Ssp: 1 | Bam: 1 | Bam: 1 | Bam: 1 |
| Enzyme 2 | Sal:1 | Pvu: 1 | Sal: 1 | Pvu: 1 | Pvu: 1 | Pvu: 1 |
| H2O | 13,5 | 13,5 | 12,5 | 12,5 | 12,5 | 12,5 |
| Total volume | 20 | 20 | 20 | 20 | 20 | 20 |
Gel Extraction
Ligation
Cloning of pCerulean_VP1up_NLS
Investigator: Anissa
Comment:Cloning did not work, because no NLS-band could be seen in the gel... Will be repeated on monday in two new approaches. One time VP1up will be cloned into pSB1C3_NLS and recloned as a fusion-product into pCerulean. Another time the oligos of NLS will be hybridisized, cut with rescriction enzymes and purificated with the Qiaex II kit.Then the NLS will be ligated into the pCerulean_VP1up
- Digestion:
| components | Vector | Insert |
| DNA | 2,6 | 32 |
| BSA (10x) | 1,5 | - |
| Buffer 4 (10x) | 1,5 | 4 |
| Enzyme | 1 PstI | 2 PstI |
| Enzyme | 1 AgeI | 2 NgoMIV |
| H2O | 7,4 | - |
| Total volume | 15 | 40 |
- Gel: that's only the picture of the 2% agarose gel for seperating the NLS. But no band could be seen.
DARPin E_01
Investigator: Bea
General overview of DARPins:
Natural protein ankyrin repeat molecules are motifs which can be found in proteins. These motifs are mediating protein-protein interactions. This suggests that ankyrin repeat (AR) proteins can be used for designing binding molecules. In Kohl et al. (2003) and Binz et al. (2003) the authors designed a structural framework with fixed consensus regions and randomized positions of interacting residues.
The repetitive nature of the ankyrin proteins allows modifications in their variable and modular binding surface. Therefore consensus sequences of natural ankyrin proteins were used to design novel and stable scaffolds for binding proteins.
Designed Ankyrin Proteins (DARPins) are well expressed, monomeric in solution, thermodynamically stable and have the ability to fold fast. In the publication of Steiner et al. (2008) screening libraries were created by useing the signal recognition particle (SRP) translocation pathway for phage display. The proteins containing the appropriate translocation signal sequence are efficiently displayed on filamentous phage particles. Screening for DARPins binding to specific target proteins like EGF-R (Erb1) were performed by phage ELISAs. The extracellular domain I-III of the receptor ErbB1 was fused to the Fc part of human IgG1 and used for selection. The previously described combinatorial N3C library was used as a template for SRP selection. After phage ELISA ans sequencing have been performed only one clone (E_01) could be selected with high-affinity binding characteristics. For broaden the diversity of ErbB1 DARPins three more clones were selected by epitope masking (E_67, E_68 and E_69). Binding and epitope localization experiments showed that all selected clones recognize a competing epitope as the dominant binder E_01, except of E_69 which recognizes an epitope not competing with E_01. The low diversity obtained for DARPins against ErbB1 suggests that the binding interfaces are well suited for dominant selection. Additionally the selected clones showed no cross-reactivity with other ErbB-family receptors (Figure 2C).
The dominant DARPpin E_01 has very high affinities to the target protein ErbB1 (Table 3) and can be used as a potential targeting molecule four our approach in fusing the DARPin to the N-terminal VP proteins.
Overview of DARPinE_01 used in our approach:
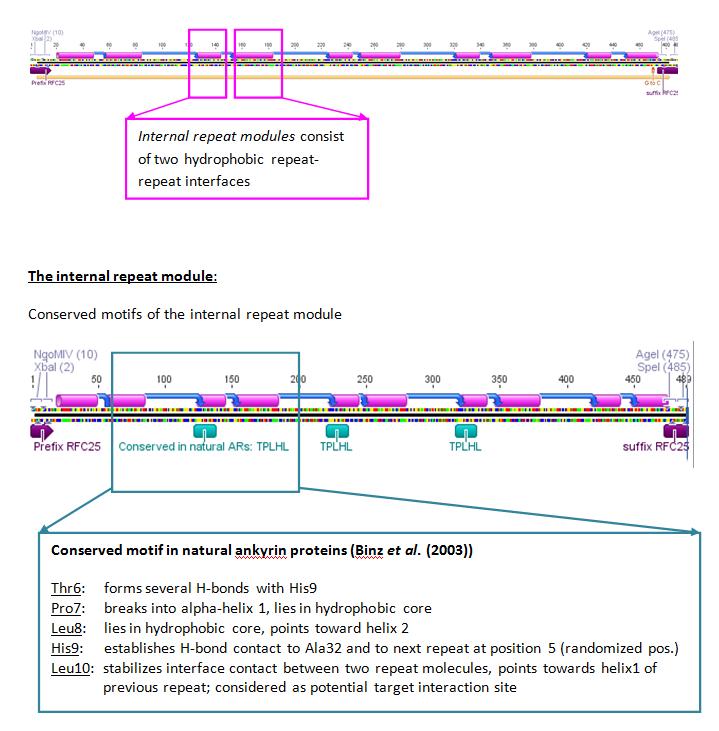
The designed ankyrin repeat protein used as a potential targeting molecule consists of three internal capping repeats and the C-and N-terminal capping repeats. Each internal repeat module comprises of one beta-turn and two hydrophobic alpha-helices. The potential interaction residues are located in the beta-turn the first alpha-helices of the AR-proteins. The complete nucleotide and corresponding amino acid sequence can be found in the appendix.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
Update VP1 insertion
Investigator: Anissa, Bea
Done:
- pSB1C3_VP1up was succesfully created by Achim. For details see labday from 2010-22-08.
- pCerulean_VP1up was suceesfully created by Anissa. pCerulean was sequenced and the VP1up which contains the upstream 137 aa´s of the VP1 protein could be successfully cloned into the plasmid backbone pCerulean (obtained by performeing a PCR).
- pSB1C3_NLSHybridization of the nuclear localization sequence (NLS) was successfully incorporated into the pSB1C3_CFP.
- Today, Anissa tried to fuse the NLS to the VP1up sequence. Cutting the pSB1C3_NLS with NgomIV and PstI leads to a 45 nt fragment which normally can be resoluted in a 2% agarose gel. But no fragment could be detected in the gel (for further details see: Cloning of pCerulean_VP1up_NLS).
On monday another approach will be performed: The NLS oligos will be hybridized and digested with NgomIV and SpeI. With the Qiaex II Kit (protocol for desalting and concentrating DNA solutions),the hybrifized oligos will be purified. Parallel, another digestion approach will be conducted.
Next steps:
- We obtain pSB1C3_VP23_HSPG_ko by conducting a PCR with the pAAV_RC_final (which contains the integrated "cap".
- Fuse the obtained construct pSB1C3_Targeting Molcule to the pSB1C3_VP23_HSPG_ko.
- Affibody ZEGFR:1907
- DARPin E_01
- 6xHis Tag
- CFP
- Fuse pSB1C3_VP23_HSPG_ko_Targeting molecule to the construct pCerulean_VP1up_NLS.
- Finally, for providing the possibilty to express obtained construct in trans: perform a site-directed mutagenesis with the pAAV_RC_final to remove the startcodon of VP1.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
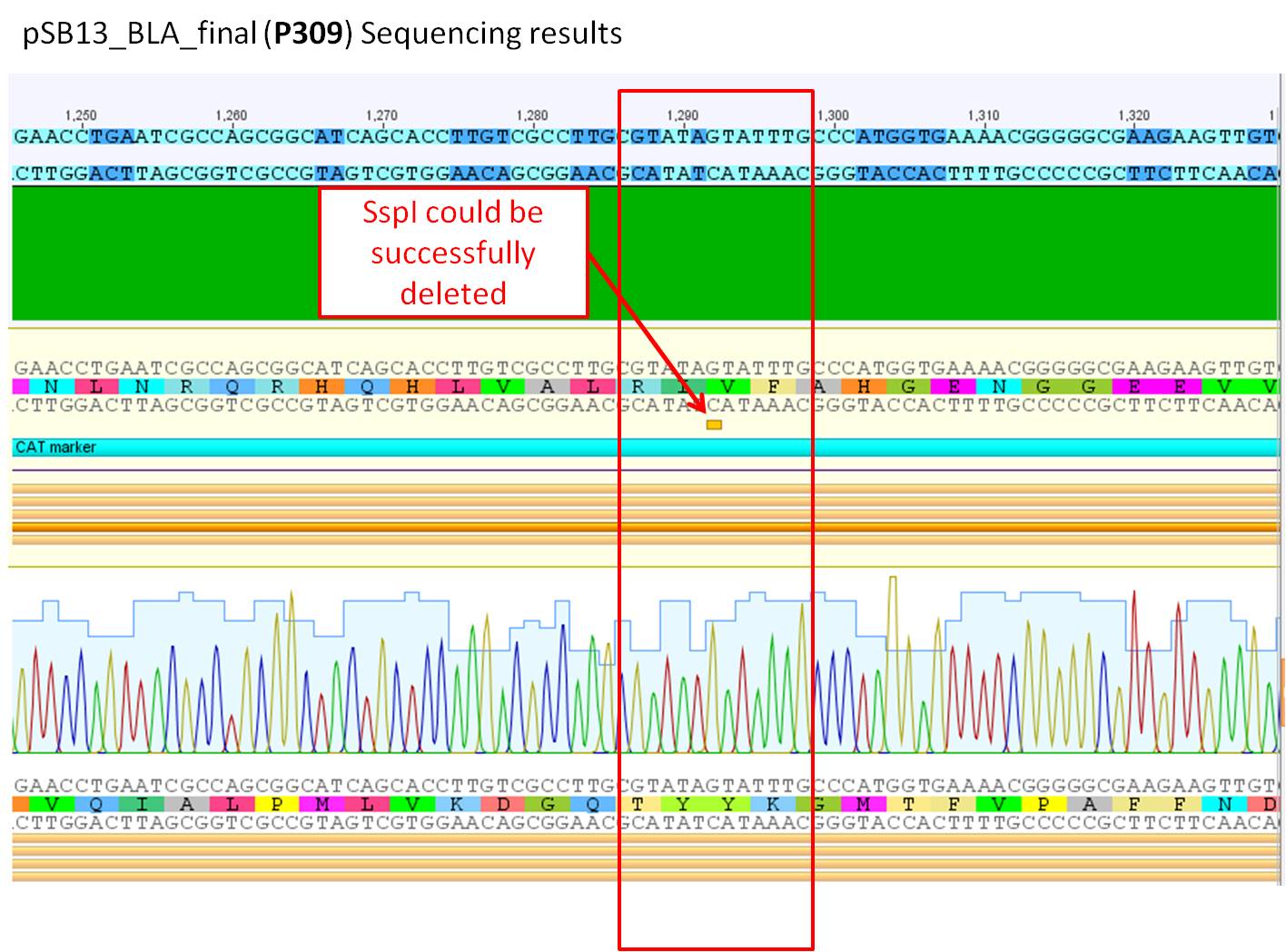
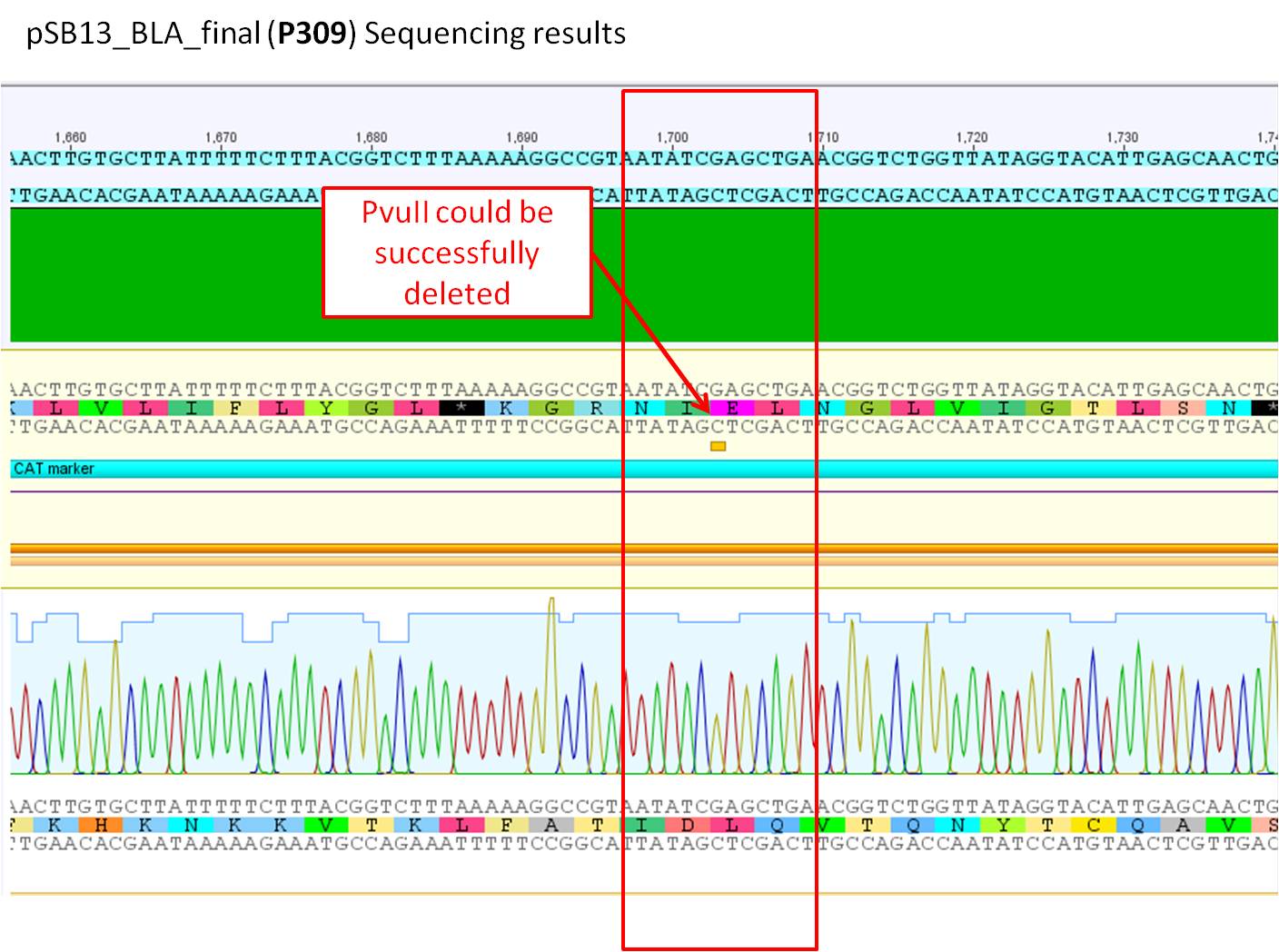
Sequence analysis of pSB1C3_BLA_final (P309)
Investigator: Bea
GENERAL COMMENT: pSB1C3_BLA_final (P309) was sent for sequencing. the beta lactamse was inserted and two site-directed mutagenesis have been performed in order to delete the loop insertion restriction enzymes in the CAT marker.
Comments: Sequence analysis of pSB1C3_BLA_final containing the two deleted restriction sites (SspI and PvuII)in the CAT marker.
- Primer used:
- pQE-RP
- VF-2
- Plasmid sequenced: P309
- Sequence sample: SB_9
- Stored in Geneious-folder: pSB1C3_BLA
The two performed site-directed mutagenesis performed at the backbone of the pSB1C3 containing the beta-lactamase can be confrimed as it can be seen in the two pictures above. Additionally the BLA sequence was sequenced aswell as the insertion of the BLA which can be confirmed with no mutations.
Next steps: This new plasmid can be used for further experiments. Clone the PCR prdouct of the pAAV_RC into the pSB1C3 and subclone the loop insertion motifs into the vector.
[http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/Laborjournal top of page]
Mini-Preps and test digestion of pSB1C3_6xHis_middlelinker and pSB1C3_6xHis_mVenus
Investigator: Stefan
-
Glycerol stocks were prepared:
- B258 = pSB1C3_6xHis_middlelinker clone1
- B259 = pSB1C3_6xHis_middlelinker clone2
- B260 = pSB1C3_6xHis_mVenus clone 1
- B261 = pSB1C3_6xHis_mVenus clone 2
- P310 = pSB1C3_6xHis_middlelinker clone1
c= 185,31 ng/µl - P311 = pSB1C3_6xHis_middlelinker clone2
c= 178,87 ng/µl - P312 = pSB1C3_6xHis_mVenus clone 1
c= 247,62 ng/µl - P313 = pSB1C3_6xHis_mVenus clone 2
c= 268,98 ng/µl
Mini-Prep following the standard protocol
Test digestion
- Restriction-enzymes used: EcoRI and PstI
| Components | P310+311 | P312+313 |
| DNA | 2 | 2 |
| BSA (10x) | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 |
| EcoRI | 0,5 | 0,5 |
| PstI | 0,5 | 0,5 |
| H2O | 5 | 5 |
| Total volume | 10 | 10 |
Incubation time: 90 minutes; Incubation temperature: 37°
1 g Agarose, 100 ml TAE (1%), 6 µl GELRED , running time: 55 minutes at 120 Volt

Comment: Test digestion looks good. Clones 1 of each plasmid (P310 and p312) were sent in for sequencing.
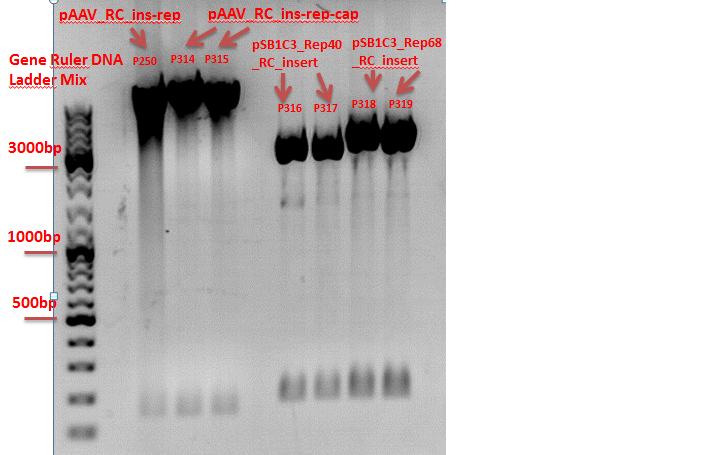
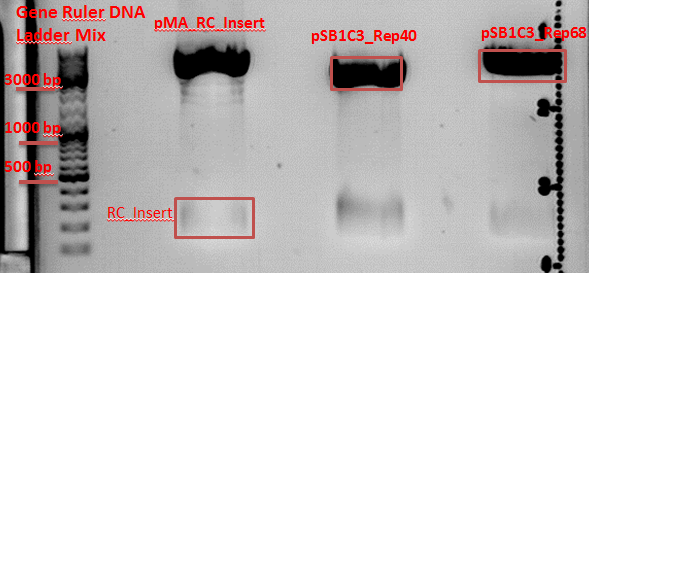
Repetition of test digestion of pAAV_RC_ins-rep-cap, pAAV_RC_ins-rep, pSB1C3_Rep40_RC_Insert and pSB1C3_Rep68_RC_Insert.
Investigator: Chris L.
- P250 = pAAV_RC_ins-rep clone1
c=467,50 ng/µl - P314 = pAAV_RC_ins-rep-cap clone1
c=517,04 ng/µl - P315 = pAAV_RC_ins-rep-cap clone2
c=444,33 ng/µl - P316 = pSB1C3_Rep40_ins clone 1
c=302,22 ng/µl - P317 = pSB1C3_Rep40_ins clone 2
c=274,37 ng/µl - P318 = pSB1C3_Rep68_ins clone 1
c=491,69 ng/µl - P319 = pSB1C3_Rep68_ins clone 2
c=498,71 ng/µl
Test digestion:
| components | volume of P250/µl | volume of P314/µl | volume of P315/µl | volume of P316/µl | volume of P317/µl | volume of P318/µl | volume of P319/µl |
| DNA | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| BSA (10x) | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Buffer 2 (10x) | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Enzyme Acc65I | 0,75 | 0,75 | 0,75 | - | - | - | - |
| Enzyme XcmI | 0,5 | 0,5 | 0,5 | - | - | - | - |
| Enzyme HindIII | - | - | - | 0,5 | 0,5 | 0,5 | 0,5 |
| Enzyme BstEII | - | - | - | 0,5 | 0,5 | 0,5 | 0,5 |
| H2O | 5,75 | 5,75 | 5,75 | 6 | 6 | 6 | 6 |
| Total volume /µl | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
Incubation time: 1 h, Incubation temperature: 37° for P250, P314 and P315
Incubation time: 45 min, Incubation temperature: 37° with HindIII for P316, P317, P318 and P319 ; 45 min, Incubation temperature: 60° with BstEII for P316, P317, P318 and P319
>br />
Preparation of gel:
1 g Agarose, 100 ml TAE (1%), 6 µl GELRED , at 120 Volt, running time: 45 minutes
- Marker: GeneRuler ladder mix
| Marker | Sample P250 10 /µl | Sample P314 10 /µl | Sample P315 10 /µl | Sample P316 10 /µl | Sample P317 10 /µl | Sample P318 10 /µl | Sample P319 10 /µl | |
|---|---|---|---|---|---|---|---|---|
| Lane | 1 | 3 | 4 | 5 | 7 | 8 | 9 | 10 |
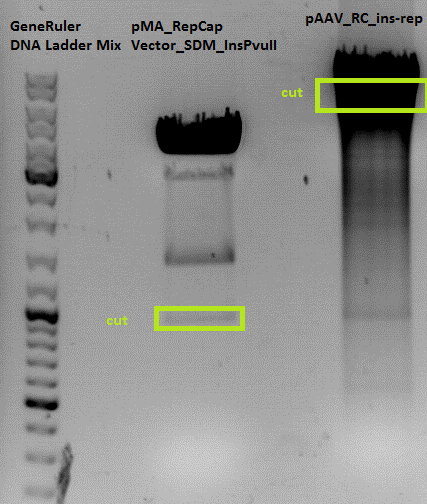
Comment: The P314 and P315 looks like the control vector. The RepCap Vector_SDM_InsPvuII insertion didn´t work. The P316, P317, P318 and P318 was cut with the "wrong" enzymes. The same fragment size as the original vector appeared. Next time cut with EcoRI.
Midi-Prep of pSB1C3_VCK_Bla
Investigators: Chris W.
Comment: Midi-Preps of P 320, B257
The Midi-Preps were performed according to the standard protocol yielding the following concentrations:
| plasmid-no. | P320 |
| concentration (ng/µl) | 408 |
103. labday 28.08.2010
Mini-Prep of pSB1C3_NLS
Investigator: Stefan
comments: No glycerol stock and test digestion was performed because bacteria culture inocculated from glyerol stock (B233).
-
Mini-Prep following the standard protocol
- P321 = pSB1C3_NLS (1)
c= 180,45 ng/µl - P322 = pSB1C3_NLS (2)
c= 187,26 ng/µl
COMMENT (Patrick): These mini-preps will be thrown away because B233 was used for inoculation due to a wrong entry in our plasmid excel sheet. B233 this is NOT the sequenced clone ! We sequenced B234/P282. Therefore 2x10 ml DYT + Cm were inoculated with B234.
Cell culture
Harvest viral Stock and Transductiot of HT and A431 cells
- P169: 100.000 cells
- P169: 200.000 cells
- P169: 300.000 cells
- P169: 400.000 cells
- P169: 500.000 cells
- P169: 600.000 cells
- P169: 700.000 cells
- P262: 100.000 cells
- P262: 200.000 cells
- P262: 300.000 cells
- P262: 400.000 cells
- P262: 500.000 cells
- P262: 600.000 cells
- P262: 700.000 cells
200.000 cells per well
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 1 | 2 |
| B | control, no virus | 1 | 2 |
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 3 | 4 |
| B | control, no virus | 3 | 4 |
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 5 | 6 |
| B | control, no virus | 5 | 6 |
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 7 | 8 |
| B | control, no virus | 7 | 9 |
| 1 | 2 | 3 | |
|---|---|---|---|
| A | control, no cells | 11 | 13 |
| B | 10 | 12 | 14 |
Seeding AAV293 for Transfection at 30.8
8 plates with 200.000 cells each were seeded
Picking clones from the ViralBricks
Investigator: Volker
Clones from all 14 transformations of the ViralBricks were picked. Because the probability that the ligation worked and that the synthesized oligos do not conatin mutations is quite bad I picked 8 to 10 clones from each ligation, resulting in approximately 150 clones at all. The clones were used to inoculate 5ml of a mixture of 60%LB-media nd 40%DYP-media because our stocks were not sufficient for this experiment.
104. labday 29.08.2010
P282/B234 mini-prep
Investigator: Patrick
... was performed according to the standard protocol.
Labelling:
- P323, pSB1C3_NLS clone 2.1, 129 ng/µl
- P324, pSB1C3_NLS clone 2.2, 141 ng/µl
Digestion of pSB1C3_NLS (P324)
The NLS was cut out with NgoMIV and EcoRI, therefore the expected size of the hoped-for fragment is 55 bp. About 30 ng DNA can be detected in a 1% gel. According to my calculation there will be maximal 80 ng insert.
55bp/2019bp = 0,027
0,027x140 ng/µl = 3,81 ng/µl
80 ng/3,81 ng/µl = 21 µl
Digestion: 21 µl pSB1C3_NLS (P324), 2,5 µl EcoRI, 2,5 µl NgoMIV, 3 µl Buffer 4, 1 µl H2O.
Digestion Time: 2 h 20 minutes.
The 2% agarose gel was loaded with 36 µl digesteion product including 6 µl 30% glycerol solution. I did not use loading dye because i to prevent that the dye overlay the mutual 55 bp insert.
The gelextraction was performed according to the the standard protocl. The resolution of the picture ist not sufficient to be able to see the band clearly as the gel was put onto the UV-light table a very thin band with the expected size could be seen.
DNA concentration measurement:
3,28 & 7,7 ng/µl (two measurements). Especially the second value is doubtable because according to my calculation above the insert conccentration cant be higher than 3,81 ng/µl. Labeled: NLS, put into 4°C room.
New digestion over night following a gelrun tomorrow: 23 µl pSB1C3_NLS (P324), 2 µl EcoRI, 2 µl NgoMIV, 3 µl Buffer 4.
Investigator: Patrick
Harvesting cultures from the ViralBricks
Investigator: Volker
All approximately 150 cultures were pelletted (twice 2 ml) and the supernatant thrown away. The pellets were freezed and the rest of the culture stored in the 4°C room. These pellets will be preped, test digested and sequenced subsequently until right clones are identified.
105. labday 30.08.2010
BioBrick assembly of pSB1C3_leftITR_pTERT & pSB1C3_ß-globin_YFP_hGH_rITR
Investigator: Achim
comment: final assembly of a vector containing all aav elements & the tert promoter
Digestion:
| components: | pSB1C3_ß-globin_YFP_hGH_rITR(Vector): P294 | pSB1C3_leftITR_pTERT (Insert): P256 |
| DNA | 2,4 | 8,4 |
| BSA (10x) | 2 | 2 |
| Buffer 4 (10x) | 2 | 2 |
| Enzyme1 | 1 (EcoRI) | 1 (EcoRI) |
| Enzyme2 | 1 (XbaI) | 1 (SpeI) |
| H2O | 11,6 | 5,6 |
| Total volume | 20 | 20 |
Preparation of gel:
0,5 g Agarose,50 ml TAE (1%), 3 µl GELRED , at 110 Volt, running time: 45 minutes
Expected sizes of constructs:
- pSB1C3_ß-globin_YFP_hGH_rITR: ~4000 bp
- pSB1C3_leftITR_pTERT: 2896 bp, ~640 bp
The corresponding bands were cut out and Gel-Extraction was performed according to protocol.
concentrations measured via NanoDrop:
- pSB1C3_ß-globin_YFP_hGH_rITR: 34.73 ng/µl
- pSB1C3_leftITR_pTERT: 9.48 ng/µl
Ligation:
T4 ligation was performed according to protocol.
Volumes used:
- vector: 2,9 µl
- insert: 5,1 µl
Transformation:
bacterial strain used: XL1bB
antibiotic used for plate: chlorampenicol
Transformation was performed according to protocol.
Plate was prepared and put in 37°C room.
Continuation: Digestion of pSB1C3_NLS (P324)
Investigator: Patrick
The gelextraction was performed with QIAEX II Gel Extraction Kit which should also allow to extract fragments smaller than 70 bp (at least 40 bp). According to the QIAGEN Gel Extraction Kit the lenght of the DNA fragment should be about 70 bp. Unfortunately the DNA fragment was blurred so a quite large piece had to be cut out.
The extraction yielded 4,26 and 3,77 ng/µl (two measurements) but this sample also quite polluted. ... put into freezer
CD biobrick production
Investigator: Kira
NA samples were diluted 1:100
| Ingredients | CD sample |
| 5X Phusion HF buffer | 10 µl |
| 10 mM dNTP mix | 1µl |
| forward primer: O158 | 2,5µl |
| reverse primer: O159 | 2,5 µl |
| DNA Template | 0,5 µl |
| DMSO | 0 µl |
| Phusion Polymerase | 0,5 µl |
| H2O | 33µl |
| Total volume | 50 µl |
PCR program:
| Cycles | Temperature | Time |
| 98°C | 1 | |
| 8x | 98°C | 15" |
| 60°C | 25" | |
| 72°C | 60" | |
| 17x | 98°C | 15" |
| 65°C | 25" | |
| 72°C | 60" | |
| 1x | 72°C | 5' |
| Hold 4°C |
Digestion of plasmid backbone:
c (pSB1C3) = 151, 1 ng/ µl
| Components | vector Volume/µL |
| DNA 1 µg | 6,0 µl |
| BSA (100x) | 0,2 µl |
| Buffer no. 4 (10x) | 2,0 µl |
| Enzyme 1 XbaI | 0,5 µl |
| Enzyme 2 AgeI HF | 0,5 µl |
| H2O | 10,8 µl |
| Total volume | 20 |
incubation @ 37 C for approx. 2 h
Digestion of PCR product:
| Components | PCR product Volume/µL |
| DNA | 30,0 µl |
| BSA (100x) | 0,4 µl |
| Buffer no. 4 (10x) | 4,0 µl |
| Enzyme 1 XbaI | 1,5 µl |
| Enzyme 2 AgeI HF | 1,0 µl |
| H2O | 3,1 µl |
| Total volume | 40 |
incubation @ 37 C for approx. 2 h
1% agarose gel
Ligation
T4 ligase was used
1 ul T4 Buffer
1 ul T4 Ligase
9 ul (6,9 ul vector+ 1,1 ul insert) DNA-mix
incubation over-night @ 18C
Repetition of test digestion of pSB1C3_Rep40_RC_Insert and pSB1C3_Rep68_RC_Insert.
Investigator: Chris L.
- P316 = pSB1C3_Rep40_ins clone 1
c=302,22 ng/µl - P317 = pSB1C3_Rep40_ins clone 2
c=274,37 ng/µl - P318 = pSB1C3_Rep68_ins clone 1
c=491,69 ng/µl - P319 = pSB1C3_Rep68_ins clone 2
c=498,71 ng/µl
Test digestion:
| components | volume of P316/µl | volume of P317/µl | volume of P318/µl | volume of P319/µl |
| DNA | 1 | 1 | 1 | 1 |
| BSA (10x) | 1 | 1 | 1 | 1 |
| Buffer 4 (10x) | 1 | 1 | 1 | 1 |
| Enzyme XcmI | 0,5 | 0,5 | 0,5 | 0,5 |
| H2O | 6,5 | 6,5 | 6,5 | 6,5 |
| Total volume /µl | 10 | 10 | 10 | 10 |
Incubation time: 1 h, Incubation temperature: 37°
>br />
Preparation of gel:
0,5 g Agarose, 50 ml TAE (1%), 3 µl GELRED , at 120 Volt, running time: 45 minutes
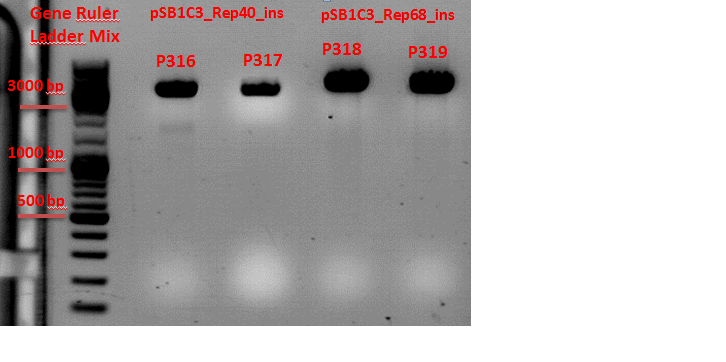
Comment: Test digestion looks good. Clones 2 of each plasmid (P317 and P319) were sent in for sequencing with VR2 Primer.
Midi-Prep of pAAV_RC_insertparts clone1
Investigators: Chris W.
Comment: Midi-Preps of B208
The Midi-Preps were performed according to the standard protocol yielding the following concentrations:
| plasmid-no. | P325 | p326 |
| concentration (ng/µl) | 2481,04 | 2050,66 |
FACS analysis, Seeding HT1080 and AAV293
Investigators: Kerstin, Adrian
- FACS analysis of Transduction from 28.08.2010
- Seeding HT1080 cells for Transduction (31.08.2010): 5x 6-well plates, 270.000 cells per well
- Seeding AAV293 cells for Transfection (01.09.2010): 10 plates, 300.000 cells per well
Mini-Preps and test digestion of Loop insertion BioBricks
Investigator: Anna
Comment: Two clones of each ViralBrick were prepared and test digested.
Mini-Preps:
| p331 | p335 | p339 | |
| Construct | pSB1C3_587_KO_BAP_clone3.1 | pSB1C3_587_RGD_clone5.1 | pSB1C3_453_His_clone7.1 |
| DNA-Concentration / µl | 65,08 | 94,29 | 73,49 |
| p342 | p345 | |
| Construct | pSB1C3_587_His_clone8.3 | pSB1C3_587_KO_empty_clone10.1 |
| DNA-Concentration / µl | 69,05 | 66,73 |
Test digestion:
1% Agarose gel: 1 g Agarose, 100 ml TAE, 6 µl Gelred
Marker: Gene ruler ladder DNA mix, 5 µl
Used enzymes: SalI, BamHI (in general, SDS has to be added because of the strong binding of enzymes to DNA!)
Comment: Test digestion had no significant results. In addition, one sample was forgotten, it was not clear which one (description makes no sense). Test digestion has to be repeated.
Repetition of subcloning Cap-insert into pAAV_RC
Investigator: Stefan
Comments: There are still problems with BspMI. It was assumed that longer digestion could improve the results, therefore digestion with BspMI will be performed overnight.
P.S: Welcome back Hanna!
Digestion of vector and insert:
- Insert: pMA_RepCap Vector_SDM_InsPvuII clone 1 (P211)
- Vector: pAAV_RC_ins-rep clone1 (P250)
A two-step digestion was performed:
1st step
| P211 / µl | P250 / µl | |
| DNA | 8,5 | 2 |
| buffer 3 | 4,6 | 5 |
| Enzyme BsiWI | 1 | 1 |
| H2O | 32,4 | 38 |
| total volume | 46 | 46 |
2nd step
| P211 / µl | P250 / µl | |
| Mix obtained from step 1 | 46 | 46 |
| Enzyme BspMI | 4 | 4 |
| total volume | 50 | 50 |
Digestion will be performed overnight and continued tomorrow.
106. labday 31.08.2010
Cloning ZEGFR:1907 and 6xHis_middlelinker into pCerulean
Investigator: Hanna
Comment: In order to fuse the His-Tag to VP2, the His-Tag-Middlelinker construct will be cloned into the pCerulean plasmid (contains CMV promoter and SV40 terminator) today. In the next step, VP2 will be cloned behind the His-Tag-Middlelinker.
In addition to that the Affibody (ZEGFR:1907) will be also cloned into pCerulean (without linker). Also here, the next step will be fusing VP2 behind the this targeting molecule.
Practical Cloning:
- plasmid:
- Vector: name: pCerulean; number: P273
- Insert: name: pSB1C3_6xHis_middlelinker (P310) + pSB1C3_Zegfr:1907 (because P267 was empty, P285 was used! See lab day 23.08.!)
- new vector name: pCerulean_6xHis_middlelinker + pCerulean_Zegfr:1907
- buffer used: 4; Restriction-enzymes used: XbaI and PstI
Comments: Because the P267 tube was empty (!), P285 was used which was prepared by Jessy - I don't know whether this is a trafo of the glycerol stock? Sequenced?
Digestion
| components | volume of pCerulean/µl | volume of pSB1C3_6xHis_middlelinker/µl | volume of pSB1C3_ZEGFR:1907 |
| DNA | 3.6 | 16.2 | 11.5 |
| BSA (10x) | 3 | 2.5 | 2 |
| Buffer 4 (10x) | 3 | 2.5 | 2 |
| Enzyme 1 XbaI | 1.5 | 1 | 1 |
| Enzyme 2 PstI | 1.5 | 1 | 1 |
| H2O | 17.4 | 1.8 | 2.5 |
| Total volume | 30 | 25 | 20 |
- Incubation: 1.5 h
Agarose-Gel:
0.8 g Agarose, 55 ml TAE (1.45 %), 3 µL GELRED, at 115 Volt, running time: 35 minutes
| Sample | Sample/µl] | Loading dye (6x)/µl | Expected size 1 (Geneious) |
|---|---|---|---|
| P285 | 20 µl | 4 µl | 210 bp |
| P273 | 30 µl | 6 µl | ~ 3900 bp |
| P310 | 25 µl | 5 µl | 86 bp |
- Marker: GeneRuler ladder mix
| Marker /µL | Sample P285 /µl | Sample P273 /µl | Sample P310 /µl | |
|---|---|---|---|---|
| Lane | 5.5 | 24 | 25 | 25 |
Gel extraction
Gel measurement:
| Sample | Weight | Volume | Concentration |
| P285 | 240 mg | 20 µL | 5.4 ng/µL |
| P273 | 130 mg | 20 µL | 14.5 ng/µL |
| P310 | 220 mg | 20 µL | 2.4 ng/µL |
Comment: The DNA concentrations were very bad, which could be due to the fact, that the 6xHis_middlelinker construct's size is just 86 bp and the affibody's size is just 210 bp...
Ligation
1. pCerulean_6xHis_middlelinker
| 6xHis_middlelinker | pCerulean | |
| Volume/µl | 2.57 | 6.43 |
2. pCerulean_Zegfr:1907
| Zegfr:1907 | pCerulean | |
| Volume/µl | 2.72 | 6.28 |
Trafo
Trafo was performed following the standard protocol. Cells: XL1b (60 µL), DNA amount per sample: 3 µL; cells were plated onto Kanamycin-plates.
PCR of SV 40 terminator
Investigator: Bea
Comment: In order to obtain the SV40 poly adenylation site and clone it into the iGEM standard plasmid pSB1C3 a PCR needs to be performed with the pEGFP-C1 which contains the SV40 terminator.
- Plasmid used for PCR: pEGFP-C1 (P230)
- 1:1000 dilution of P230 has been prepared
- Plasmid used as vector: pSB1C3_CFP1 (P51.2)
PCR (was performed following the standard protocol)
| Ingredients | Volume / µl |
| 5X Phusion HF buffer | 10 |
| 10 mM dNTP mix | 1 |
| forward primer: O160 | 2,5 |
| reverse primer: O161 | 2,5 |
| DNA Template | 3 |
| Phusion Polymerase | 0,5 |
| H2O | 30,5 |
| Total volume | 50 |
PCR program:
| PCR Program | temperature/ °C | Time |
| 1 | 98 | 1min |
| 2 | 98 | 15s |
| 8x | 59 | 25s |
| 3 | 72 | 7s |
| 4 | 98 | 15s |
| 17x | 67 | 25s |
| 5 | 72 | 7s |
| 6x | 72 | 5min |
| Hold | 4 |
After the PCR was performed, the sample was loaded on a 1.5% agarose gel and run for 30 minutes. As it can be seen in the picture below the expected size of the PCR product (260bp) can be confirmed. The lower band corresponds to the added primers and does not seem to be an additional band.
For cloning the obtained fragment into the pSB1C3 standard vector, the construct need to be digested with the same restriction enzymes as the PCR product will be cut with. In this case the primers were designed with XbaI overhang for the prefix, and with SpeI-NotI-PstI overhang for the suffix. Therefore the vector will be cut with XbaI and PstI. The sample were incubated at 37 C for approx. 1.5 h
- Plasmid used: pSB1C3_CFP (P51.2) c=150ng/µL
- Add BSA
- XbaI and PstI were used
Digestion of vector:
| Components | vpSB1C3_CFP/µL |
| DNA | 8 |
| BSA (100x) | 2 |
| Buffer no. 4 (10x) | 2 |
| Enzyme 1 XbaI | 1 |
| Enzyme 2 AgeI HF | 1 |
| H2O | 6 |
| Total volume | 20 |
- Incubation of the sample at 37°C
- Incubation for 120 minutes
After the PCR has been cut out of the gel and purified with the Qiagen Gel Extraction Kit obtaining a concentration of 45 ng/µl measured with the Nanodrop, the complete PCR product volume of 29 µL was digested with XbaI and PstI for 2 hours.
Digestion of the PCR product SV40
| Components | vPCR product/µL |
| DNA | 29,0 µl |
| BSA (10x) | 4,0 µl |
| Buffer no. 4 (10x) | 4,0 µl |
| Enzyme 1 XbaI | 1,0 µl |
| Enzyme 2 AgeI HF | 1,0 µl |
| H2O | 1,0 µl |
| Total volume | 40 |
- Incubation of the sample at 37°C
- Incubation for 120 minutes
The digested PCR product SV40 was purifed with the PC Purification Kit and concentration was measured.
- cSV40= 31,5 ng/µl
- cpSB1C3= 7,74 ng/µL
For T4 ligation of the two fragments, the needed volumes of insert (SV40) and vector (pSB1C3) were calculated with the labtools program. The total volume DNA (vector/insert) mix will be 8 µL.
- vSV40= 0,67 µL
- vpSB1C3= 7,33 µL
- vT4 buffer= 1 µL
- vt4 ligase= 1 µL
The ligation reaction was carried out for 40 minutes at room temperature. The ligated plasmid was transformed into XL1-Blue cells and plated on agar plates containing the appropriate amount of chloramphenicol.
Cloning of NLS into pCerulean_VP1up
Investigator: Anissa
Comment: NLS was already 2 times cut of the pSB1C3_NLS and purified by Patrick (NLS 1: of the 29.08.10, NLS 2: of the 30.08.10) . Now, pCerulean_VP1up will be digested and NLS will be ligated into it.
- Digestion of pCerulean_VP1up (p303)
components Vector DNA 2,6 BSA (10x) 1,5 Buffer 4 (10x) 1,5 Enzyme PstI 1 Enzyme AgeI 1 H2O 7,4 Total volume 15
- Gel:
A 1% gel was made and the sample was running 30 minutes.

Afterwards a gelextraction according the qiagen-protocol was performed.
- Ligation:
components Volume /µl concentration /ng/µl NLS 2 insert of 30.08.10 1,51 3 NLS 1 insert of 29.08.10 1,51 3 pCerulean_VP1up 6,49 29
- Transfomation: was performed into XL1 blue on a kanamycin plate according the standard protocol.
Cloning CFP_middlelinker (from pSB1C3_CFP_middlelinker, P276) into pCerulean (P273)
Investigator: Patrick
P276 digestion (207 ng/µl): 14 µl DNA sample, 2 µl Buffer 4, 2 µl BSA, 1 µl XbaI, 1 µl PstI-HF.
P273 digestion (419 ng/µl): 4 µl DNA sample, 1 µl Buffer 4, 1 µl BSA, 1 µl XbaI, 1 µl PstI-HF.
Digestion time: 1 h 45 min.
Expected size of the fragments:
P276: 786 & 2058 bp
P273: 756 & 3938 bp
see: http://www.molbiotech.uni-freiburg.de/iGEM/wiki2010/index.php/August_2010#Strategy_for_VP2_N-terminal_fusion_and_VP1_insertion_of_targeting_moelcules
Haha, good idea! :)
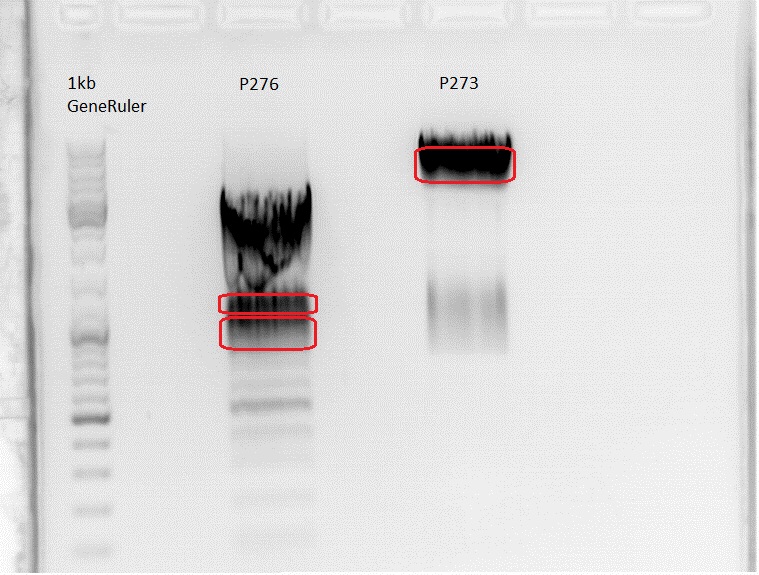
Gelextraction:
Obviously there was something wrong with the gel or the sample. The marked bands were cut out and the gelextraction was performed acording to he standard protocol, yielding the following concentrations:
P276 up: 12,6 ng/µl
P276 down: 9,9 ng/µl
P273: 37,7 ng/µl
All of them were polluted.
There were two bands at a level where no bands should be. P 276 looks also strange because accidentally 6,5 µl 1kb GeneRuler were pipetted into the sample. The expected bands are all blurred. Even the expected 756 bp fragment of P273 is not at the level it should be. To ensure i dont throw away the insert i cut out both bands from P276. Now tomorrow there will be a ligation and a transformation with two samples instead of one
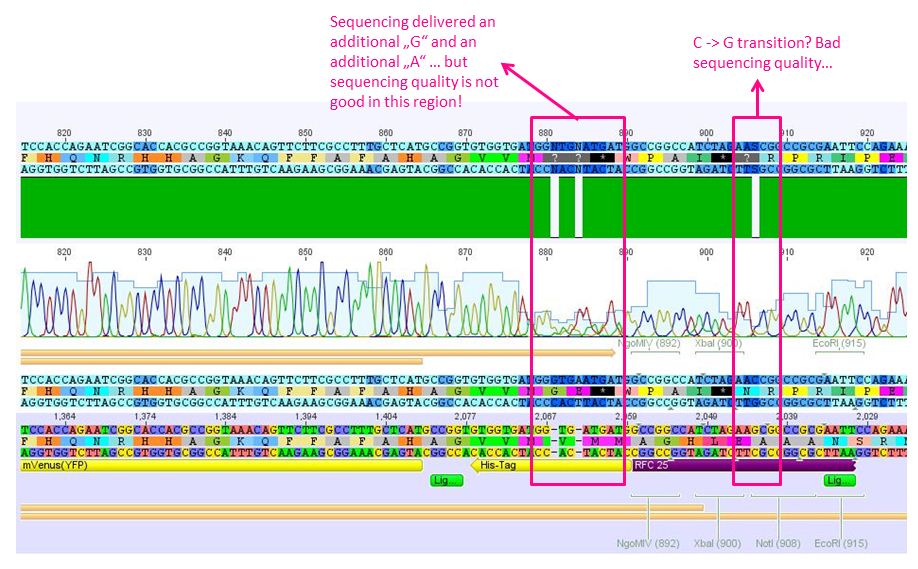
Sequencing results of pSB1C3_6xHis_middlelinker and pSB1C3_6xHis_mVenus
Investigator: Hanna
1st Comment: Sequencing of pSB1C3_6xHis_middlelinker looked good:
2nd Comment: Sequencing of mVenus looked good. The His-Tag delivered 2 additional bp and the suffix 1 transition (see picture). This could be due to bad sequencing qualities at this region... but needs to be verified!
Contiuation: Repetition of subcloning Cap-insert into pAAV_RC
Investigator: Stefan
Comments: Overnight digestion does not look good, therefore a new approach will be performed today using different concentrations of spermidine (0,5mM, 2,5mM and 10mM) because in Oller et al (Biochemistry, 1991) it was suggested that this improves digestion with BspMI.
A two-step digestion was performed:
1st step
| P211 / µl | P250 / µl | |
| DNA | 8,5 | 2 |
| buffer 3 | 4,6 | 5 |
| Enzyme BsiWI | 1 | 1 |
| H2O | 32,4 | 38 |
| total volume | 46 | 46 |
- Incubation: 70 minutes; 55°C
2nd step
| P211 / µl | P250 / µl | |
| Mix obtained from step 1 | 46 | 46 |
| Enzyme BspMI | 4 | 4 |
| total volume | 50 | 50 |
- Incubation: 2,5h ; 37°C
Agarosegel
0.4 g Agarose, 50 ml TAE (0,8 %), 3 µL GELRED, at 115 Volt
Gel extraction
Gel-Ex was performed according to protocol.
NanoDrop:
- pMA_RepCap-insert: c= 7,42 ng/µl
- pAAV_RC_ins-rep: c=12,9 ng/µl
T4 Ligation
Volume insert: 3,43 µl
Volume vector: 4,57 µl
Ligation incubated for 50 minutes at room temperature.
Transformation
Trafo was preformed according to standard protocol using XL1blue cells on plates containing Ampicilin.
2nd Repetition of test digestion of Loop insertion BioBricks
Investigator: Achim, Anna
Comment: First test digestion didn't work (see 31.08.10), this time a new approach was done with NotI.
Test digestion:
| components | Volume for each sample /µl |
| DNA | 10 |
| BSA (10x) | 1,5 |
| Buffer 4 (10x) | 1,5 |
| Enzyme NotI | 0,5 |
| H2O | 1,5 |
| Total volume /µl | 15 |
| Sample-No. | 1+2 | 3+4 | 5+6 | 7+8 | 9+10 | 11+12 | 13+14 | 15+16 | 17+18 | 19+20 | 21-23 | 24+25 | 26+27 | 28+29 |
| Components | 453 BAP | 587 BAP | 587 KO BAP | 453 RGD | 587 RGD | 587 KO RGD | 453 HIS | 587 HIS | 587 KO HIS | 587 KO EMPTY | 453 Z34C | 587 Z34C | 587 KO Z34C | 587 KO Z34C SPACER |
| Oligo 1 | O124: 10 | O126: 10 | O128: 10 | O130: 10 | O132: 10 | O134: 10 | O135: 10 | O137: 10 | O139: 10 | O141: 10 | O143: 10 | O145: 10 | O147: 10 | O149: 10 |
| Oligo 2 | O125: 10 | O127: 10 | O129: 10 | O131: 10 | O133: 10 | O151: 10 | O136: 10 | O138: 10 | O140: 10 | O142: 10 | O144: 10 | O146: 10 | O148: 10 | O150: 10 |
30: pSb1C3_Bla (cut version) 31: pSb1C3_Bla (uncut)
Incubation time: 1 h, Incubation temperature: 37°
Preparation of gel:
1 g Agarose, 100 ml TAE (1%), 6 µl GELRED , at 115 Volt, running time: 45 minutes
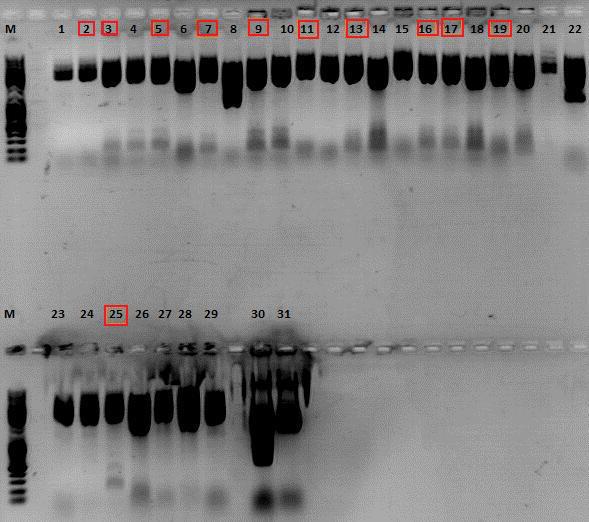
Expected fragment sizes /bp:
pSB1C3: 2051 bp
Bla: 905
BAP: ~150
RGD: ~ 130
Z34C: ~ 200
His: ~ 125
The following clones were sent for sequencing: 2,3,5,7,9,11,13,16,17,19 and 25.
Comment: Clones (see above) were sent for sequencing with VR2 Primer. In addition, 8 mL DYT medium was inoculated with each of the clones for preparation of glycerol stocks. For sequencing results go to labday 01.09.10.
Midi-Prep of pAAV_RC & pHelper
Investigators: Chris W.
Comment: Midi-Preps of pHelper=P356 and pAAV_RC=P357
The Midi-Preps were performed according to the standard protocol yielding the following concentrations:
| plasmid-no. | P356 | P357 |
| concentration (ng/µl) | 1068,0 | 1274.80 |
Transformation of Biobrick CD
Investigator: Kira
Transformation was performed according to the standard protocol.
 "
"