Team:Calgary/31 July 2010
From 2010.igem.org

Saturday July 31, 2010
We love weekends!! Except when we come in to work...
Chris
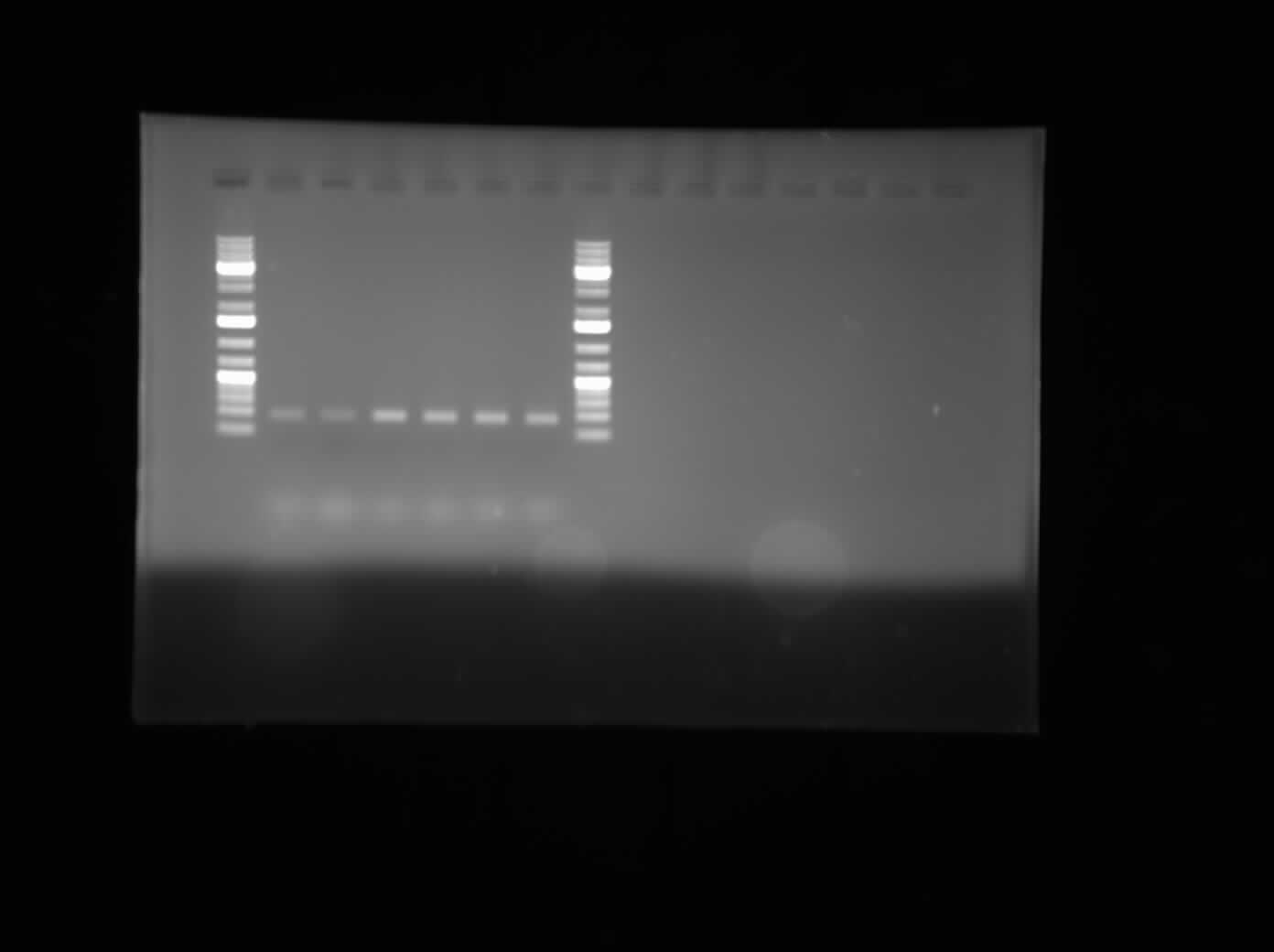
Today, I did a PCR purification of the CpxP promoter that was PCR-ed out of the E. coli genome using the Qiagen kit that we have. 100 µL of each PCR tube was purified. Emily and I took a look at the vacuum PCR purification that Henry provided but were unsure of the protocol to use. One modification to the protocol that was used was the addition of 15 minutes waiting time in between the washing and the eluting step of the protocol. This was suggested by another lab as a method to increase DNA concentration in the final product. Another modification to the procedure was that the elution was done in 35 µL of water instead of the PB Buffer. A gel was run of the purified PCR product and the gel can be seen on the right. The bands are still about the same place which they should be for the CpxP promoter.
Emily
Today I PCR Purified the malE and malE31 that Raida had previously PCR'd out of the plasmid DNA from the Betton labs. The primers that she used add the XbaI and SpeI restriction sites on either end of the genes, so on Monday I will be digesting both, as well as the psB1AC3 and psB1AK3 vectors with XbaI and SpeI in order to try to get these genes into a BBK construction vector. I also ran a gel of the malESSdel PCR that Henry, Raida and I set up yesterday. Unfortunately my overnight cultures of malE and malE31 in TOPO vectors did not have any growth. Chris is going to try setting these up again tomorrow night so that I can miniprep them Monday morning.
 "
"