Team:Washington/Gram Negative/Build
From 2010.igem.org
(→Creating the Toxin/Antitoxin circuit) |
(→Creating the Toxin/Antitoxin circuit) |
||
| Line 29: | Line 29: | ||
=Creating the Toxin/Antitoxin circuit= | =Creating the Toxin/Antitoxin circuit= | ||
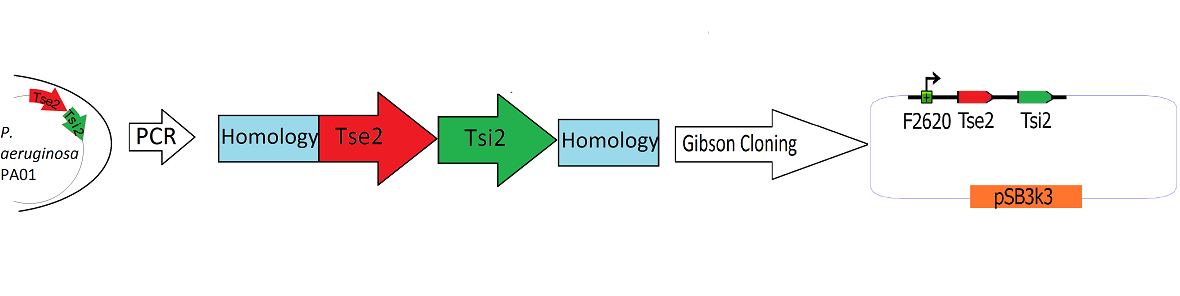
| - | [[Image:Washington_Building_Tse2_circuit.png|800px|]] | + | [[Image:Washington_Building_Tse2_circuit.png|800px|thumb|Schematic Detailing the Transfer of Tse2 and Tsi2 from ''P. Aeruginosa'' into pSB3k3]] |
The Tse2/Tsi2 locus was amplified using PCR. Primers were used that amplified the region from the start codon of Tse2 to the stop codon of Tsi2. The primers were designed to flank the Tse2/Tsi2 product with approximately 40bp regions of homology to the psb3k3-F2620 plasmid. The Tse2/Tsi2 locus was then placed downstream from F2620 via [https://2010.igem.org/Team:Washington/Project/Tools/Klavins Gibson Cloning]. Gibson cloning is a method that joins regions of homology found on the 5' and 3' ends of linearized molecules. The end result of this Gibson cloning reaction is a circularized plasmid containing the final F2620-Tse2/Tsi2 construct in psb3k3. The cicrularized plasmid is then transformed into electrocompetent ''E. coli'' Dh5a and the resulting colonies were screened for the insert of expected length via double restriction digest followed by agarose gel electrophoresis. The plasmids that showed inserts of expected length were sequenced, and it was determined that multiple plasmids had the correct F2620-Tse2/Tsi2 inserts. | The Tse2/Tsi2 locus was amplified using PCR. Primers were used that amplified the region from the start codon of Tse2 to the stop codon of Tsi2. The primers were designed to flank the Tse2/Tsi2 product with approximately 40bp regions of homology to the psb3k3-F2620 plasmid. The Tse2/Tsi2 locus was then placed downstream from F2620 via [https://2010.igem.org/Team:Washington/Project/Tools/Klavins Gibson Cloning]. Gibson cloning is a method that joins regions of homology found on the 5' and 3' ends of linearized molecules. The end result of this Gibson cloning reaction is a circularized plasmid containing the final F2620-Tse2/Tsi2 construct in psb3k3. The cicrularized plasmid is then transformed into electrocompetent ''E. coli'' Dh5a and the resulting colonies were screened for the insert of expected length via double restriction digest followed by agarose gel electrophoresis. The plasmids that showed inserts of expected length were sequenced, and it was determined that multiple plasmids had the correct F2620-Tse2/Tsi2 inserts. | ||
Revision as of 01:49, 21 October 2010
Building a Type VI Dependent Probiotic
One of the major hurdles in creating our probiotic was the high level of regulation that this system is under in its native host. Several transcriptional and post-transcriptional regulators are present in both promoter regions which prevented the system from expressing in E. coli. Another problem was the large size of the secretion system. These two issues made standard cloning very difficult if not impossible. A set of vectors were also designed which would contain the toxin/antitoxin system. These vectors were designed to activate in the presence of our signal molecule, HSL.
Creating the Toxin/Antitoxin circuit
The Tse2/Tsi2 locus was amplified using PCR. Primers were used that amplified the region from the start codon of Tse2 to the stop codon of Tsi2. The primers were designed to flank the Tse2/Tsi2 product with approximately 40bp regions of homology to the psb3k3-F2620 plasmid. The Tse2/Tsi2 locus was then placed downstream from F2620 via Gibson Cloning. Gibson cloning is a method that joins regions of homology found on the 5' and 3' ends of linearized molecules. The end result of this Gibson cloning reaction is a circularized plasmid containing the final F2620-Tse2/Tsi2 construct in psb3k3. The cicrularized plasmid is then transformed into electrocompetent E. coli Dh5a and the resulting colonies were screened for the insert of expected length via double restriction digest followed by agarose gel electrophoresis. The plasmids that showed inserts of expected length were sequenced, and it was determined that multiple plasmids had the correct F2620-Tse2/Tsi2 inserts.
Recombineering the type 6 secretion system
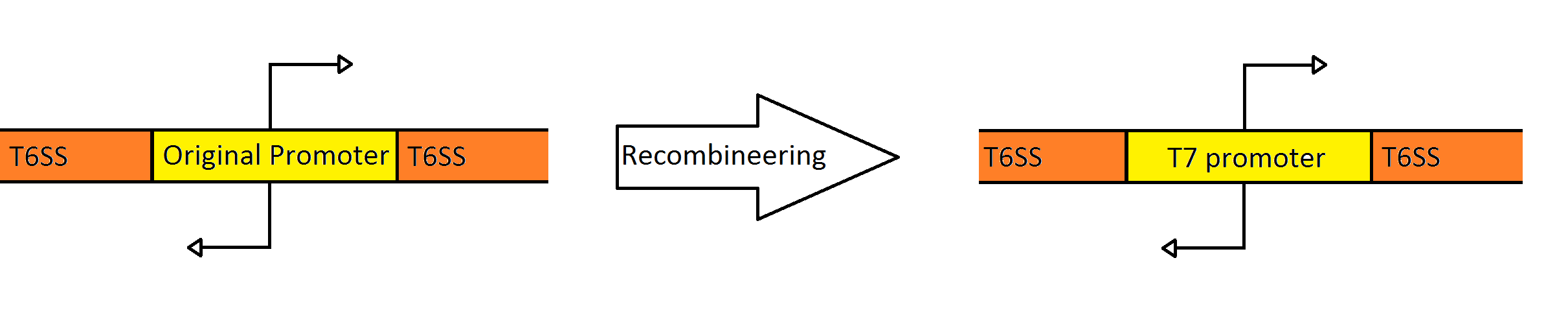 In order to replace the native promoters with more robust T7 promoters we used a technique called Recombineering to flip out the region containing both promoters. We designed primers to create a cassette using PCR, containing the galK galactose metabolism gene flanked by 50 base pairs of homology with the promoter region. The cassette was inserted, giving us a selection factor for our final insertion of our new promoters. A new promoter cassette was designed in total using oligos that encoded the two T7 promoters flanked by 50 base pairs of homology, and inserted using recombineering to create a robust Type VI Secretion System.
In order to replace the native promoters with more robust T7 promoters we used a technique called Recombineering to flip out the region containing both promoters. We designed primers to create a cassette using PCR, containing the galK galactose metabolism gene flanked by 50 base pairs of homology with the promoter region. The cassette was inserted, giving us a selection factor for our final insertion of our new promoters. A new promoter cassette was designed in total using oligos that encoded the two T7 promoters flanked by 50 base pairs of homology, and inserted using recombineering to create a robust Type VI Secretion System.
 "
"