Team:Imperial College London/Lab Diaries/Vectors team
From 2010.igem.org
| Line 5: | Line 5: | ||
__NOTOC__ | __NOTOC__ | ||
| - | == | + | {| style="width:900px;background:#f5f5f5;text-align:justify;font-family: helvetica, arial, sans-serif;color:#555555;margin-top:5px;" cellspacing="20" |
| - | + | |style="font-family: helvetica, arial, sans-serif;font-size:2em;color:#ea8828;"|Objectives | |
| + | |- | ||
| + | | | ||
We are assembling the AmyE and PyrD vectors in order to transform B. Subtilis with our parts. Once completed, these vectors will be reusable and can then be used to introduce any relevant piece of DNA directly into B.subtilis genome. The vectors will be used both for the final assembly and for testing constructs. | We are assembling the AmyE and PyrD vectors in order to transform B. Subtilis with our parts. Once completed, these vectors will be reusable and can then be used to introduce any relevant piece of DNA directly into B.subtilis genome. The vectors will be used both for the final assembly and for testing constructs. | ||
| + | |} | ||
| + | {| style="width:900px;background:#f5f5f5;text-align:justify;font-family: helvetica, arial, sans-serif;color:#555555;margin-top:5px;" cellspacing="20" | ||
| + | |style="font-family: helvetica, arial, sans-serif;font-size:2em;color:#ea8828;"|PyrD Vector | ||
| + | |- | ||
| + | | | ||
| - | |||
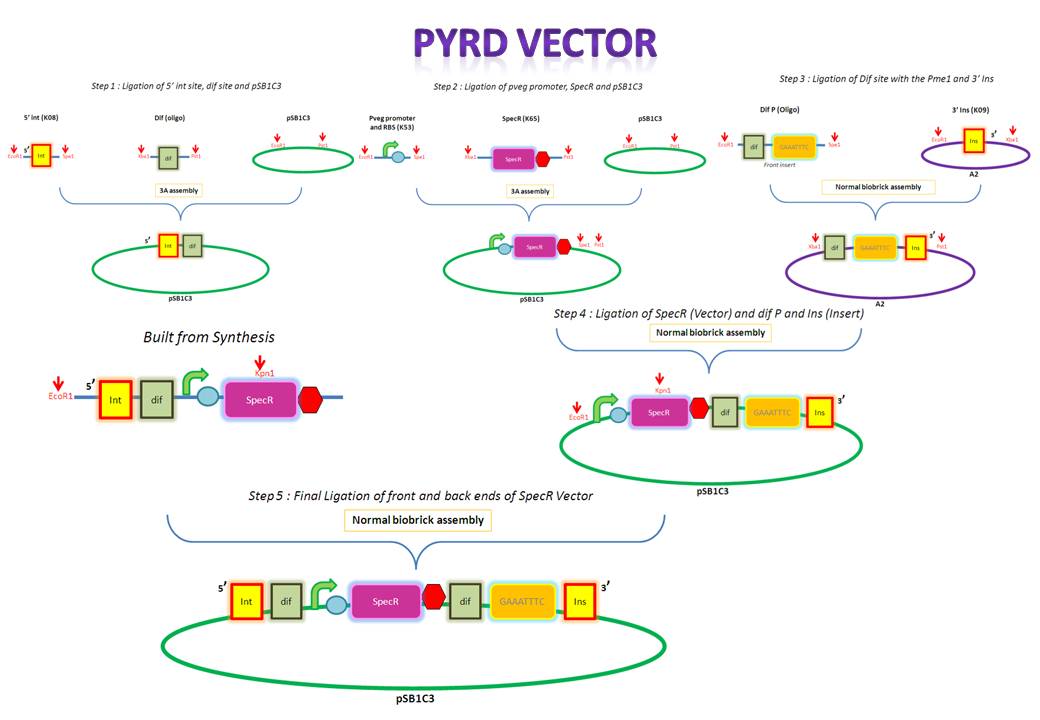
[[Image:IC_PyrD.JPG |750px|thumb|center|alt=A|Overveiw of PyrD assembly]] | [[Image:IC_PyrD.JPG |750px|thumb|center|alt=A|Overveiw of PyrD assembly]] | ||
| Line 916: | Line 922: | ||
</table> | </table> | ||
</html> | </html> | ||
| + | |||
| + | |} | ||
| + | |||
==AmyE Vector== | ==AmyE Vector== | ||
| Line 932: | Line 941: | ||
# Run on a gel to confirm correct cutting and estimate relative ratios of DNA for ligation | # Run on a gel to confirm correct cutting and estimate relative ratios of DNA for ligation | ||
# Ligated overnight | # Ligated overnight | ||
| - | # Transformed into E.Coli to Amplify the DNA | + | # Transformed into ''E.Coli'' to Amplify the DNA |
# Colony PCR has been used to confirm the correct insert size. | # Colony PCR has been used to confirm the correct insert size. | ||
Revision as of 19:22, 23 October 2010
| Lab Diaries | Overview | Surface Protein Team | XylE Team | Vectors Team | Modelling Team |
| Here are the technical diaries for our project. We've split them up into three lab teams and the modelling team. We think it's really important that absolutely anyone can find out what we've been doing. For a really detailed look at what we did, and when, you've come to the right place! | |
| Objectives |
|
We are assembling the AmyE and PyrD vectors in order to transform B. Subtilis with our parts. Once completed, these vectors will be reusable and can then be used to introduce any relevant piece of DNA directly into B.subtilis genome. The vectors will be used both for the final assembly and for testing constructs. |
| PyrD Vector | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Week 6
Thursday, August 12
The forward and reverse strands of the 5' dif site with XbaI and PstI restriction sites on either side have been synthesized separately. The synthesized fragments arrive in solid powder form. These were immediately diluted in ddH2O to obtain a stock concentration of 1 ng/ul. They were then allowed to anneal together by first heating them to 95 degs for denaturation and allowing them to cool down and anneal overnight. Friday, August 13
After annealing the two strands, the oligo was cut with XbaI and PstI to obtain overhangs that would later ligate with the compatible overhangs of a cut vector.
The cut oligo was PCR purified in order to get rid of any contaminants. PCR purification gets rid of short pieces of DNA which are less than about 40 base pairs.
Week 7
Monday, August 16
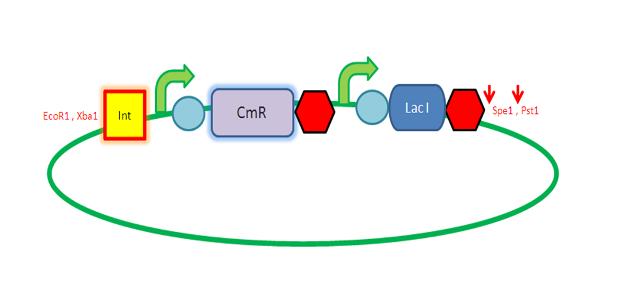
K143008 is the 5' integration site for the PyrD vector. This will be used as a front insert together with the dif XP for the pSB1C3 vector.
The pSB1C3 vector backbone from the registry was amplified with the use of SB3 and SB2a primers. Submission of parts to the registry requires them to be in a pSB1C3 vector therefore any parts to be submitted will be inserted into this vector.
The PCR amplified vector was purified in order to get rid of any contaminants. For example, short pieces of DNA like the primers. Tuesday, August 17
The digested 5' ins was first analyzed on the gel to verify it's size and then extracted for purification. The 5' ins was gel purified in order to extract only the relevant piece of DNA.
The pSB1C3 vector was digested so that it would contain compatible overhangs for ligation with inserts.
The digested pSB1C3 was re-purified in order to get rid of any contaminant DNA that arose during the digestion.
pveg (promoter and RBS) and Spec-T (Spectinomycin with a terminator) were digested in preparation for 3A assembly.
The digested 5' ins and dif (the front inserts) were ligated overnight with pSB1C3 (the vector). A bench ligation and an overnight ligation were set up.
E.Coli was transformed via the chemical method using the bench ligate.
Since these are both inserts they were gel extracted and purified. PCR purification is not carried out for inserts since they are small and would therefore be lost during the process. Wednesday, August 18
pveg and SpecR-T (the front inserts) were ligated overnight with pSB1C3 (the vector). Thursday, August 19
Friday, August 20
The forward and reverse strands of the 3' dif Pme1 sites with XbaI and PstI restriction sites on either side have been synthesized separately. The synthesized fragments arrive in solid powder form. These were immediately diluted in ddH2O to obtain a stock concentration of 1 ng/ul. They were then allowed to anneal together by heating them to 95 degs for denaturation and allowing them to cool down and anneal overnight. Week 8
Week 9
Week 10
Week 11
Week 12
Week 13
|
AmyE Vector
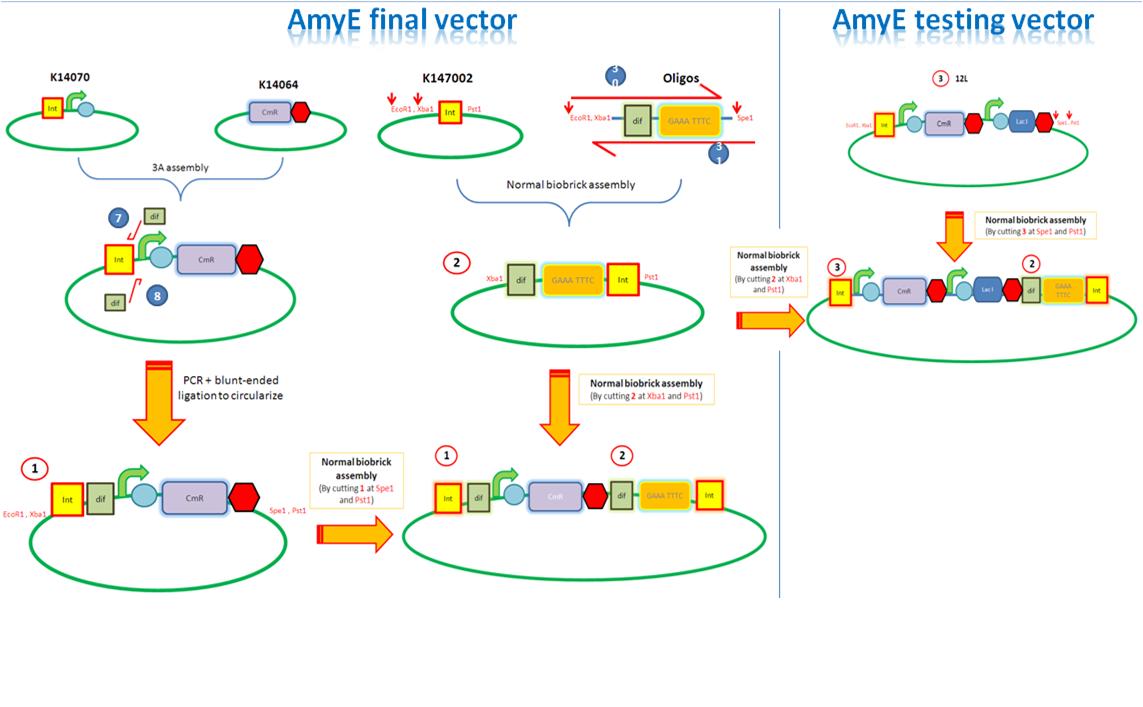
Assembly of the AmyE vector Assembly
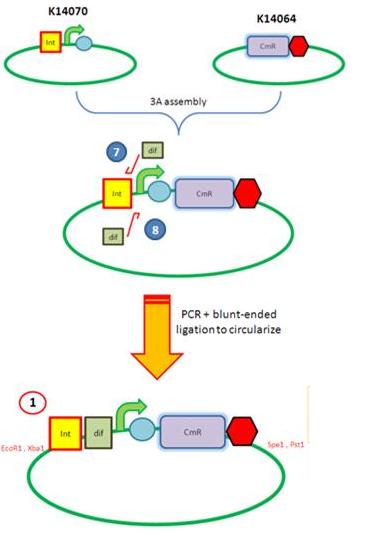
Starting from the top, we are assembling the first two fragments (K14070 and K14064) and K147002 with our oligos to add in a Dif site. Two dif sites on either side of resistance cassettes can be used to later excise antibiotic resistance from our final constructs.
K14070 and K14064 fragments
- DNA was taken out of the reigstry
- Cut with restriction enzymes
- Run on a gel to confirm correct cutting and estimate relative ratios of DNA for ligation
- Ligated overnight
- Transformed into E.Coli to Amplify the DNA
- Colony PCR has been used to confirm the correct insert size.
Next Steps:
- Extract the DNA with a miniprep
- Proceed to the next step - Reverse PCR
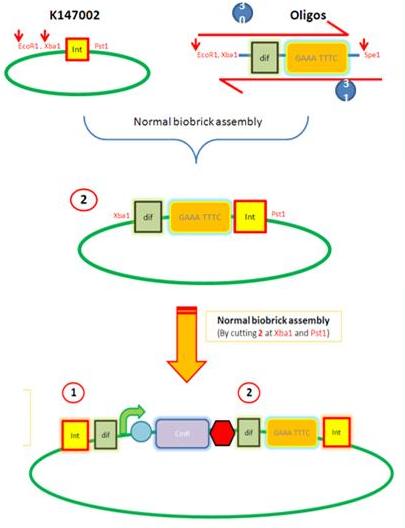
K14002 and oligos
- Ligated two single stranded oligos together to produce Dif and insertion site
- Standard biobrick assembly of oligos to K14002
- Ligation and transformation into E.coli competent cells (strain)
- Screen plated colonies for correct insertion
Next Steps:
- Purify the correct insert out of E.coli
- Next step assembly - LacI test vector and Final assembly vector
LacI Testing vector
Currently waiting for Part B step 2 Midi-prep results to start this step.
Schedule & Lab notes
Week 7
| Week 6 | Monday | Tuesday | Wednesday | Thursday | Friday |
|---|---|---|---|---|---|
| MORNING | Starting assembly of AmyE vector
|
|
|
k64 and k70, k70 only, Spec and 5' PyrD diff |
Spec and 5' PyrD diff
k64 and k70. k70 only was discarded since this was purely for a baground check |
| AFTERNOON |
|
|
|
|
Week 8
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | Saturday |
|---|---|---|---|---|---|---|
| MORNING |
Set up overnight ligations for standard assembly (BBA) and 3A cloning (3A) of dif P
|
|
|
|
|
|
| AFTERNOON |
|
|
|
|
Backbone PCR of pSB1C3 using PFU (3rd attempt)
|
|
Week 9
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | |
|---|---|---|---|---|---|---|
| MORNING |
|
|
|
|
| |
| AFTERNOON |
|
|
|
|
|
Week 10
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | |
|---|---|---|---|---|---|---|
| MORNING |
|
|
|
|
| |
| AFTERNOON |
|
|
|
|
|
Week 11
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | |
|---|---|---|---|---|---|---|
| MORNING |
|
|
|
|
| |
| AFTERNOON |
|
|
|
|
|
Week 12
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | |
|---|---|---|---|---|---|---|
| MORNING |
|
|
|
|
| |
| AFTERNOON |
|
|
|
|
|
Week 13
| Week 8 | Monday | Tuesday | Wednesday | Thursday | Friday | |
|---|---|---|---|---|---|---|
| MORNING |
|
|
|
|
| |
| AFTERNOON |
|
|
|
|
|
 "
"