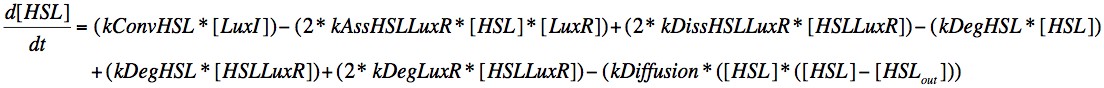Team:St Andrews/project/modelling/model 2
Model 2: Single Cell With Loop
Theory & assumptions
Our second model introduced the concept of an external environment into which the HSL could diffuse into. This was assumed to be of infinite volume and thus the concentration of HSL in this region would always be zero, regardless of the quantity of HSL leaving the cell. In order to implement this modification we produced a new differential to desrcibe the change in HSL.
Figure 1: Differential equation describing HSL change
The creation of HSL is dictated by kConv, which is associated with the ability of LuxI to catalyse the conversion of resources into HSL. The concentration is reduced by the association with LuxR and by natural degredation. It is also affected by the degredation of molecules in the HSL-LuxR complex in the same way that LuxR is. We also introduce a diffusion term which accounts for HSL diffusing out of the cell due to a concentration gradient between the cell and the external environment.
The code on which this model is based can be found here.
Results
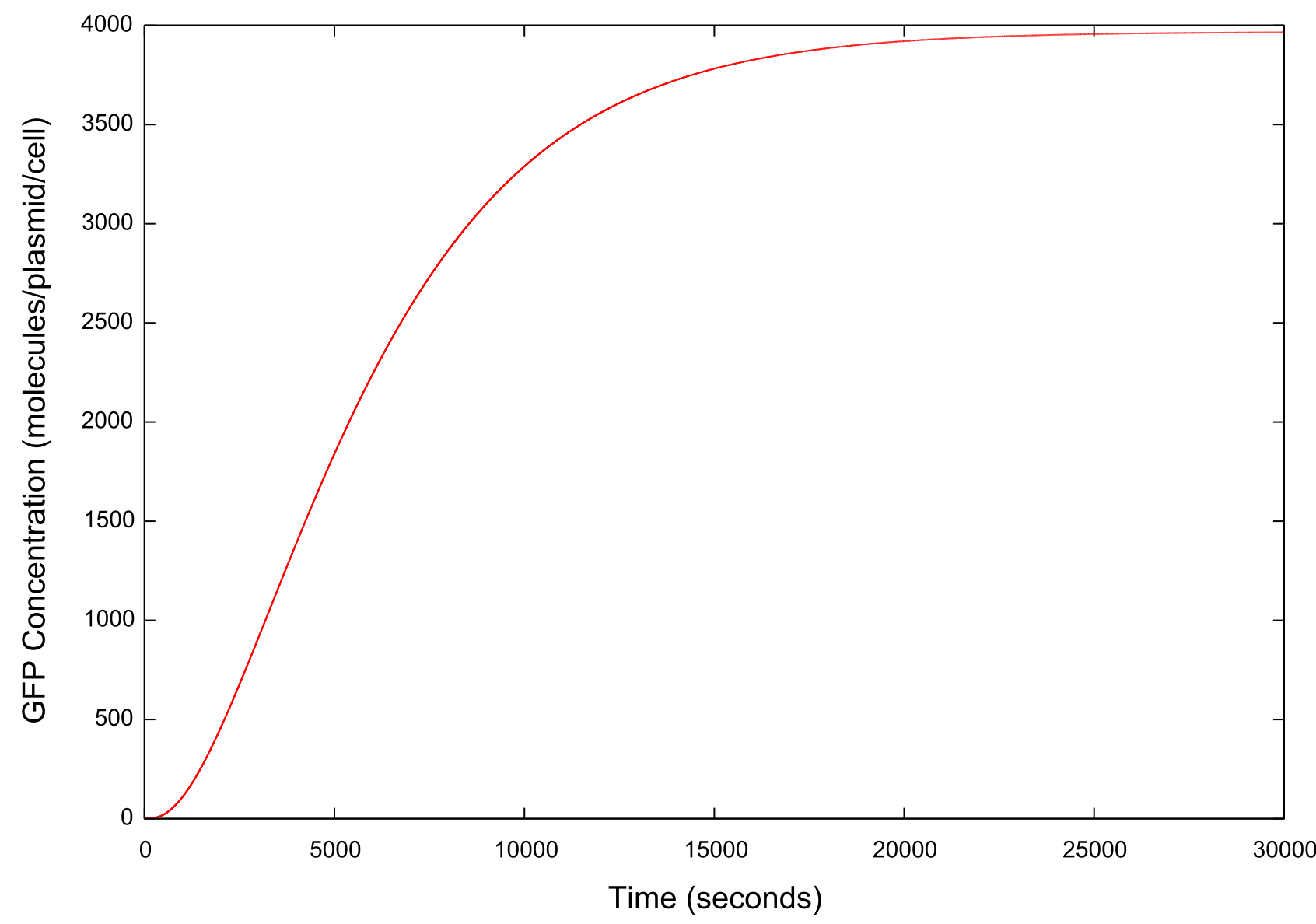
The switching on behaviour produced by this model is shown in figure . This switching on can be interpreted as the cell turning itself on due to the HSL it produces not diffusing out of the cell at a high enough rate. Therefore there is a considerable concentration of HSL remaining in the cell to maintain the feedback loop and produce more HSL. Thus regardless of the initial concentrations our cell always switches on. This does not match with the true biological scenario at all. Quorum sensing is designed such that cells will only become up-regulated when present at a certain critical cell density. Therefore the suggestion made by our results that a single cell can self-activate undermines this principle entirely.
Figure 2: Graph of GFP concentration against time
There is an apparent switching on of the system, but this is in fact not what we would expect seeing as our model is for a single cell in a surrounding environment of infinite volume. Therefore any HSL produced by the cell should become diluted to the point where the concentration externally can be said to be negligible.
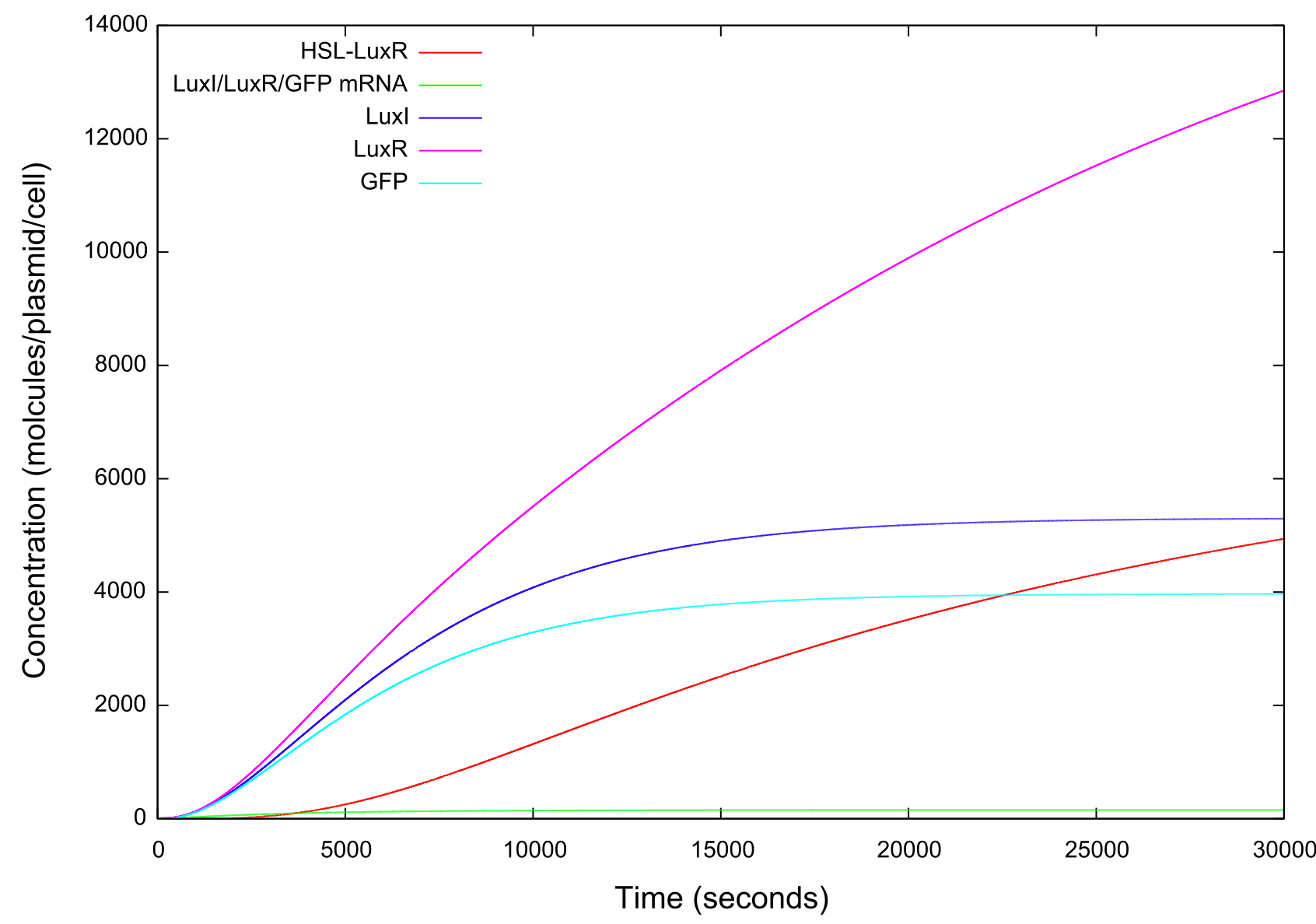
Figure 3: Graph of GFP, LuxI, LuxR, HSL-LuxR and mRNA concentrations against time
We can see from figure 3 that the model predicts huge concentrations of the HSL-LuxR complex to be present in the cell, which strongly contradicts previous work. The complex is very unstable and should therefore exist in only small concentrations within the cell. This gives us some idea as to the fallibility of this model. It is also of note that in general the order of magnitude if the concentrations output from this model are a few below that from model 1.
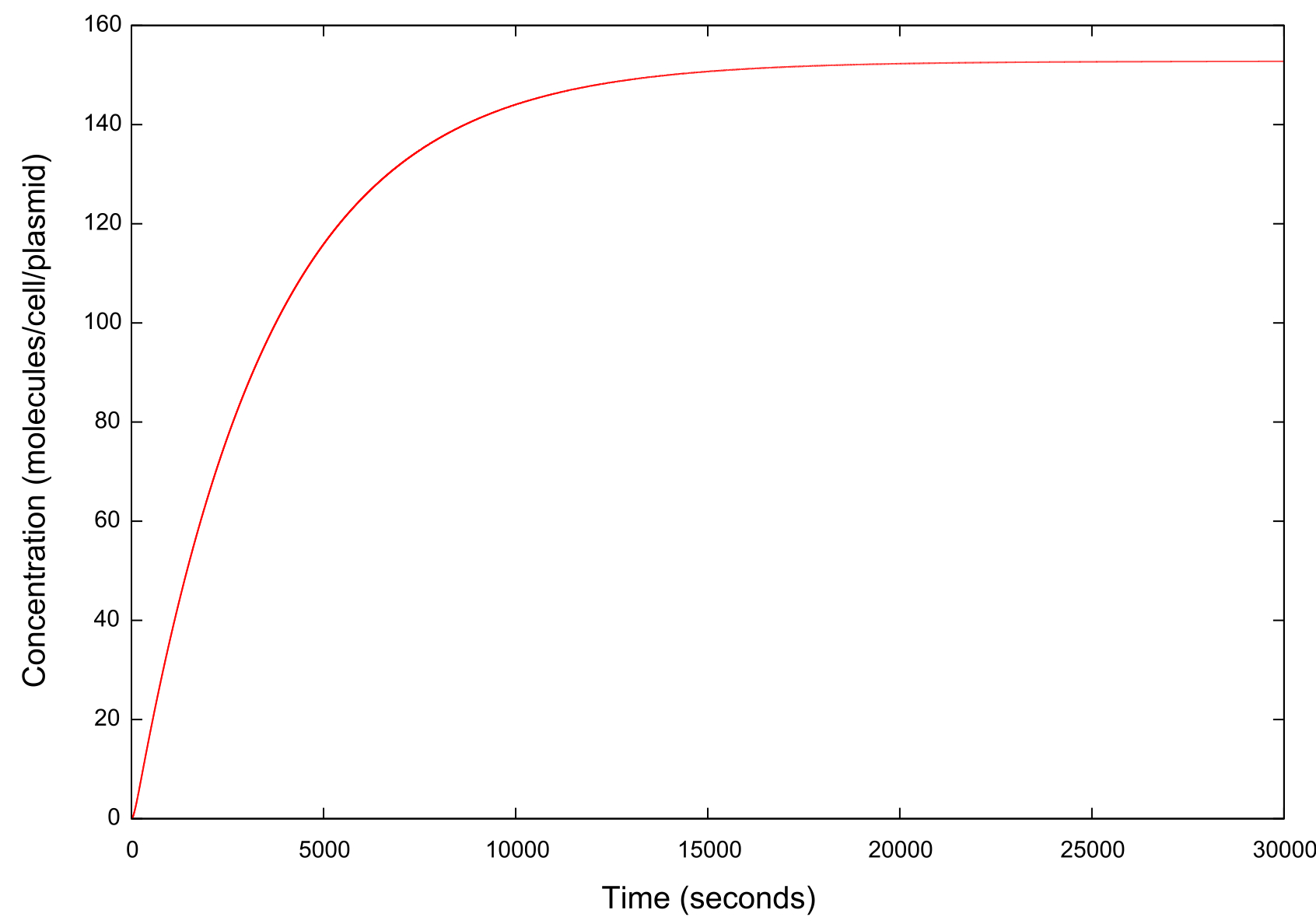
Figure 4: Graph of mRNA concentration against time
The concentrations of mRNA are equal for GFP, LuxR and LuxI, and whatsmore are comparable to those seen in our previous model.
Conclusions
Our model demonstrated switching behaviour and also contained some concept of diffusion. However, it also came with its fair share of problems. Among these was the fact that a single cell seems to switch itself on regardless of the initial conditions or of the rate constants. In addition there was no concept of other cells. As such we felt this particular model was unsuitable for use and began work on an improved version.
 "
"