Team:Calgary/Project/IbpAB
From 2010.igem.org

IbpAB Circuit
Protein misfolding can occur as a result of several factors. It can be due to overproduction of the protein in the cell, in which case the cell lacks resources such as chaperones to fold the protein fast enough. This can cause the proteins to misfold and form aggregate bodies. Proteins can also misfold due to mutations that occur in the coding region of the protein that can alter the amino acid sequence and thereby interrupting the native structure of the protein, causing it to misfold and be dysfunctional. Proteins can also misfold due to cellular stress such as a change in pH, temperature and change in media. Due to lack of optimal folding conditions proteins can form aggregate bodies and cause activation of heat shock systems, chaperone systems and proteolytic pathways which are involved in either refolding the proteins into their native form or degrading the aggregate bodies. Proteins can also misfold due to lack of localization. For example: if a periplasmic protein lacked a signal sequence it will misfold in the cytoplasm because the conditions are different in the two cellular compartments.
There are several heat shock pathways in E. coli which are actively transcribed in response to cellular stress. This class of proteins are called small heat shock proteins (sHsps). sHsps consist of proteins such as ibpA, ibpB, DnaK, DnaJ, GroEL and GroES. Amongst these, IbpA and ibpB are two different proteins that are activated as a result of cytoplasmic stress response. IbpA and ibpB proteins are chaperones that are responsible for refolding aggregated bodies and inclusion bodies into their native conformation.
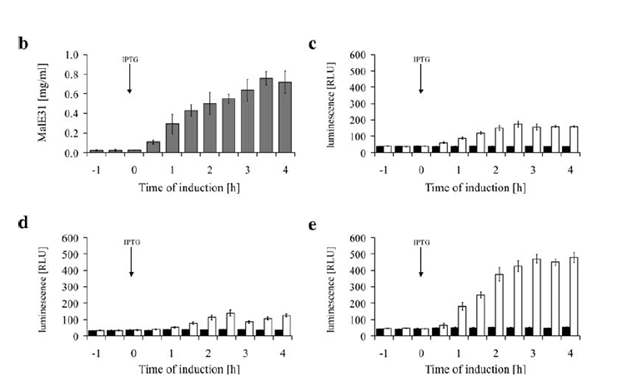
In our cytoplasmic stress detector circuit, we decided to fuse two different promoter regions from two heat shock proteins, which are ibpAB and fxsA. In a study done by Kraft et al, they demonstrate that a fusion of IbpAB/fxsA promoters combined along with T7 DNA has a significantly higher output as a result of heat shock compared to the promoters individually.
This fusion promoter will be connected to the registry part I13504 which is RBS-GFP-B0015. The ibpAB/fxsA circuit will be activated in the presence of aggregation in the cell. We will be using MalE31 with a signal sequence deletion (MalE31∆SS) which was designed by Betton et al. The native E. coli protein MalE generally exported into the periplasmic space but this mutated protein does not get exported to the periplasmic space due to the signal sequence deletion. Also Betton et al designed MalE31such that there are two amino acid changes in the protein and it misfolds. The MalE31∆SS protein coding region will be used in order to induce cytoplasmic protein stress in E. coli. Ideally, this misfolded MalE31∆SS should activate the plasmid system containing ibpAB/fxsA-I13504 which will produce GFP alerting the researcher that their protein is not being expressed in the cell because it is misfolding and as a result getting degraded. Our circuit should also be activated much faster than the native stress system because the ibpAB/fxsA promoter is much more sensitive to the presence of aggregate bodies in the cell. The promoter also gives a much higher output compared to the promoters individually, which is the case in the E. coli genome which should allow us to detect the fluorescence level much faster.

 "
"