Team:TU Munich/Lab
From 2010.igem.org
|
||||||||
|
|
Experiment DesignIn vivo Measurements
Our initial idea to prove our concept of antitermination was to use flourescent proteins as reporters. This approach gives the opportunity to measure the termination and antitermination efficiency of our designed BioBricks in vivo as well as in vitro, the latter using a translation kit based on E. coli lysate.
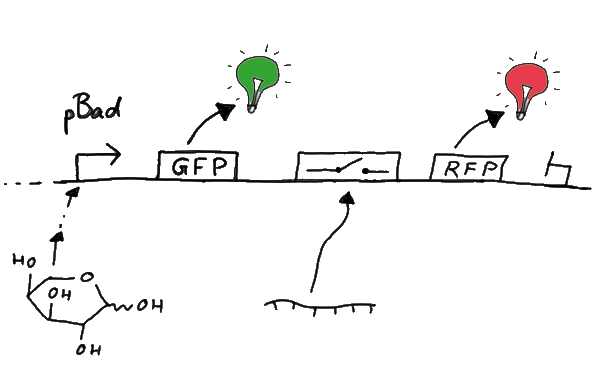
Upon binding of the signal the stem loop of the switch would resolve leading to red fluorescent additional to the green one of GFP. The internal control carries the advantage that errors in the measurement set can be detected easily. Lack of arabinose or promotor insensitivity can be recognized as well as problems with the fluorescence measurement itself. Plus, we have a way to normalize our measurements and compare different preparations in relation to each other.
Our measuring plasmid is based on the BioBrick pSB1A10, A1, distribution 2010. Unfortunately after two months of cloning we had to recognize that the plasmid in use did not work (see also Biobrick validation--> link). So after the first unsuccessful attempts we had to reclone the system, substituing RFP to mCherry, a dsRED derivative with a spectra in the far red, and adding arabinose inducible promoters in front of both fluorescent proteins.
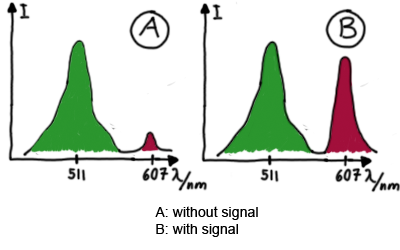
For switch evaluation, IPTG was added to the cells after about two hours after arabinose induction (baseline). ??? Stimmt das?? A rise of RFP/mCherry emission should be visible in case of a working switch.
When measuring the termination of our BioBricks and the antitermination by their corresponding signal-RNA, we should be able to observe an increasing RFP emission compared to the GFP emission upon induced signal-RNA production in the cells/in the kit:
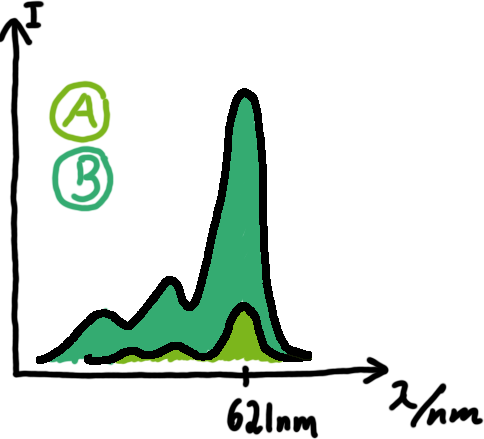
With these measurements, it should also be possible to observe differences in efficiency of termination as well as antitermination between our designed switches. In vitro Translation
So haben wir in vivo translation gemessen CloseIn vitro Measurements
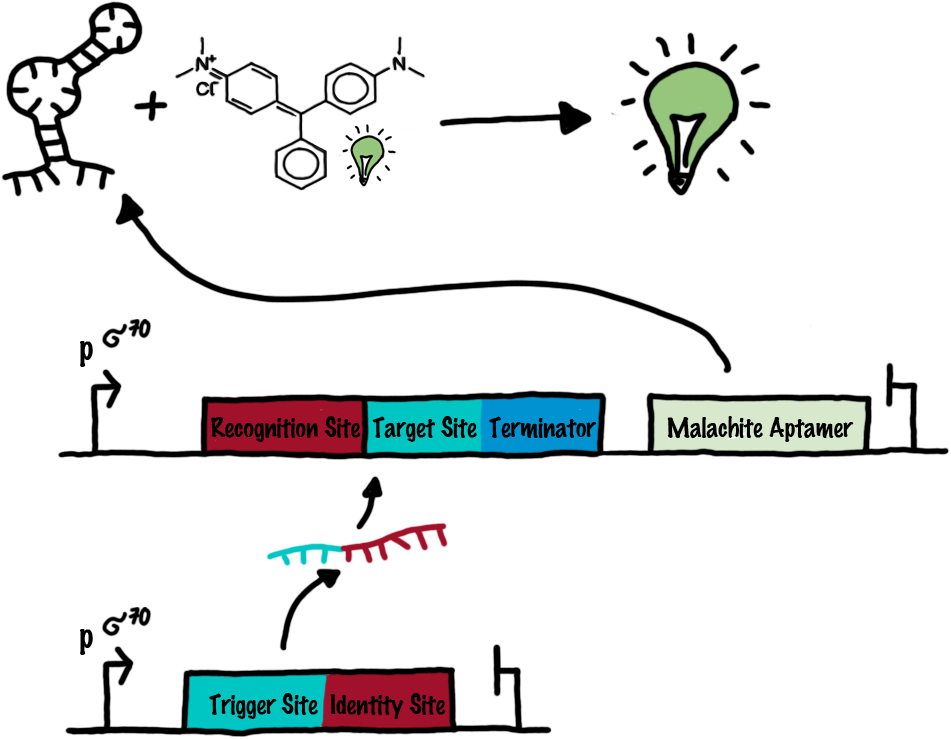
A second possibility to measure parameters of our switches we came up with, was the idea to investigate our system on the transcriptional level only. Therefore, we decided to use malachite green as reporter. Malachite green in a fluorescent dye, whose emission increasing dramaticly (about 3000 times) upon binding of a specific RNA-aptamer.
---concept to be described, as well as literature---
<ref>refs</ref>
To study the switches on the transcriptional level gives the advantage, that we would have less interferences and possible artefacts. Also, we are not sure how cellular mechanisms like degradation of RNases or interacting factors as well as molecular crowding influence our systems. We made constructs comprising of a sigma(70)-binding promoter followed by a short nonsense sequence, the switches and the aptamer sequence. ProtocolsMolecular BiologyPCR
So geht ne PCR CloseDigestion
So geht ne PCR CloseLigation
In vivo measurement
So haben wir in vivo gemessen CloseIn vitro expression
So haben wir in vitro gemessen CloseIn vitro measurement
So haben wir in vivo gemessen CloseLab BookExplanation:
|
|||||||
 "
"