Team:Lethbridge/Notebook/Lab Work/June
From 2010.igem.org
Adam.smith4 (Talk | contribs) (→June 2/2010) |
Adam.smith4 (Talk | contribs) (→June 2010) |
||
| Line 129: | Line 129: | ||
Incubated reactions overnight at room temperature (total of 19.5 hours)<br> | Incubated reactions overnight at room temperature (total of 19.5 hours)<br> | ||
Killed enzymes by incubating reactions for 10 minutes at 80<sup>o</sup>C</ul> | Killed enzymes by incubating reactions for 10 minutes at 80<sup>o</sup>C</ul> | ||
| + | |||
| + | ===June 2/2010 - Evening=== | ||
| + | <b>Objective:</b> Set up new ligations of pLacI and sRBS-Lum-dT according to Tom Knight's protocol. Previous ligation had very little DNA.<br> | ||
| + | <b>Relevant Information:</b><br> | ||
| + | <ol> | ||
| + | <li>Want a final mass of 25ng of each pDNA in the ligation mix. | ||
| + | <li>Final concentration of pDNA in restriction digest should be 25-50ng/µL. | ||
| + | <li>Tom Knight's restriction reaction is 50µL, therefore there should be 1000ng pDNA in each restriction digest. | ||
| + | <li>Identified the following plasmids in our [[Team:Lethbridge/Notebook/Working_Plasmids|working plasmids box]]: | ||
| + | <table><table border ="3"> | ||
| + | <tr><td><b>Common Name</b></td><td><b>Location</b></td><td><b>Concentration (ng/µL)</b></td><td><b>Volume/rxn (µL)</b></td></tr> | ||
| + | <tr><td>pLacI Maxiprep</td><td>A9</td><td>990</td><td>~1</td></tr> | ||
| + | <tr><td>pLacI (B1)</td><td>A6</td><td>440</td><td>~2</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (2)</td><td>A1</td><td>965</td><td>~1</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (1)</td><td>A2</td><td>1145</td><td>~1</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT Maxiprep</td><td>B8</td><td>4780</td><td>~.2</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT</td><td>B7</td><td>4375</td><td>~.25</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (1)</td><td>G2</td><td>335</td><td>~3</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (2)</td><td>G3</td><td>965</td><td>~2</td></tr></table> | ||
| + | *Make a 1:10 dilution of sRBS-Lum-dt maxiprep (D8) and sRBS-Lum-dT (B7). 0.5µL pDNA in 4.5µL water. | ||
| + | <li>Cut pLacI with EcoRI and SpeI | ||
| + | <li>Cut sRBS-Lum-dT with XbaI and PstI | ||
| + | <li>Cut pSB1T3 with EcoRI and PstI | ||
| + | <li>Will have total of 12 ligation reactions, want 12x2µL of pSB1T3 to add to each, therefore want 25µL of pSB1T3.</ol> | ||
| + | <b>Method:</b><br> | ||
| + | Experimental Setup<br> | ||
| + | <table><table border ="3"> | ||
| + | <tr><td><b>Name</b></td><td><b>[pDNA] (ng/µL)</b></td><td><b>Volume<br>pDNA (µL)</b></td><td><b>Volume<br>Water (µL)</b></td><td><b>Volume<br>Buffer (µL)</b></td><td><b>Enzymes</b></td><td><b>Total Volume</b></td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr> | ||
| + | <tr><td>sRBS-Lum-dT (A1)</td><td>965</td><td>1</td><td>43.5</td><td>5</td><td>0.25µL XbaI<br>0.25µL PstI</td><td>50</td></tr></table> | ||
| + | |||
===June 3/2010=== | ===June 3/2010=== | ||
<b>Objective:</b> Ligate pLacI to sRBS-Lum-dT using the three antibiotic assembly method (according to Tom Knight's protocol). Previous ligation had miniscule quantities of DNA in ligation, not surprising it didn't work.<br> | <b>Objective:</b> Ligate pLacI to sRBS-Lum-dT using the three antibiotic assembly method (according to Tom Knight's protocol). Previous ligation had miniscule quantities of DNA in ligation, not surprising it didn't work.<br> | ||
Revision as of 01:18, 11 June 2010
Contents |
June 2010
June 1/2010
JV quantified the amount of DNA in gels run to date using ImageJ software. Results to be posted in working plasmids box.
Objective: Transform plasmids into DH5α
Method: Follow competent cell transformation protocol to transform the following:
From our ligations:
- pLacI-sRBS-Lumazine-dT
- pLacI-sRBS-Lumazine-dT
- mms6 (A6)
- mms6 (B6)
- xylE (C4)
- xylE (B4)
From the 2010 Parts Distribution:
- ECFP (Bba_E0020)
- EYFP (Bba_E0030)
- BglII Endonuclease (Bba_K112106)
June 2/2010
(In Lab: JV)
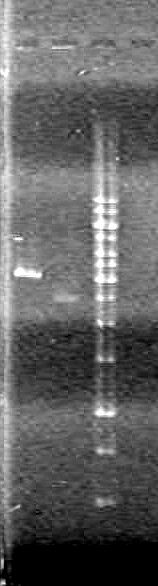
Objective: Isolate plasmid DNA of RBS-xylE (BBa_J33204) from DH5α cells and confirm results.
Method: "Mini-prep" the plasmid DNA using boiling lysis miniprep. Then restrict the DNA once and run on a 1% agarose gel (TAE).
Restriction Reaction
| Ingredient | Volume(µL) |
| MilliQ H20 Water | 15.75 |
| Orange Buffer (10x) | 2 |
| pDNA (rbs-xylE) | 2 |
| EcoRI | 0.25 |
Unrestricted Control
| Ingredient | Volume(µL) |
| MilliQ H20 Water | 16 |
| Orange Buffer (10x) | 2 |
| pDNA (rbs-xylE) | 2 |
DNA was restricted for 80 minutes at 37oC.
Analyzed results on a 1% agarose gel. Load order as follows:
| Lane | Sample | Volume Sample (µL) | Volume Loading Dye (µL) |
| 1 | Restricted RBS-xylE | 10 | 2 |
| 1 | Unestricted RBS-xylE† | 1 | 2 |
| 1 | 1kb Ladder†† | 2 | 2 |
† Added 9µL MilliQ H2O
†† Added 8µL MilliQ H2O
Ran gel at 100V from 2 hours.
Results:
Conclusions: Plasmid DNA prep and restriction was successful.
Objective: Ligate rbs-xylE (Bba_J33204) to our double terminator, and insert it into the pSB1T3 plasmid backbone.
Method:
- Restrictions
- Restrict rbs-xylE wit EcoRI and SpeI (Red Buffer)
- Restrict the double terminator with XbaI and PstI (Tango Buffer)
- Restrict pSB1T3 with EcoRI and PstI (Red Buffer)
Component Volume (µL) MilliQ H2O 15.5 Buffer 2 pDNA 2 Enzyme 0.25 + 0.25 Set up control reaction as follows:
- MilliQ H2O - 16µL
- Buffer - 2µL
- pDNA - 2µL
Incubated reactions for 65 minutes at 37oC
Killed enzymes by incubating reactions for 10 minutes at 65oC
- Ligation
Reaction set up as follows:- T4 DNA ligase - 0.25µL
- rbs-xylE - 5µL
- dT - 3µL
- pSB1T3 - 8µL
- 10x Ligation Buffer - 2µL
- MilliQ H2O - 1.75µL
Killed enzymes by incubating reactions for 10 minutes at 80oC</ul>June 2/2010 - Evening
Objective: Set up new ligations of pLacI and sRBS-Lum-dT according to Tom Knight's protocol. Previous ligation had very little DNA.
Relevant Information:
- Want a final mass of 25ng of each pDNA in the ligation mix.
- Final concentration of pDNA in restriction digest should be 25-50ng/µL.
- Tom Knight's restriction reaction is 50µL, therefore there should be 1000ng pDNA in each restriction digest.
- Identified the following plasmids in our working plasmids box:
Common Name Location Concentration (ng/µL) Volume/rxn (µL) pLacI Maxiprep A9 990 ~1 pLacI (B1) A6 440 ~2 sRBS-Lum-dT (2) A1 965 ~1 sRBS-Lum-dT (1) A2 1145 ~1 sRBS-Lum-dT Maxiprep B8 4780 ~.2 sRBS-Lum-dT B7 4375 ~.25 sRBS-Lum-dT (1) G2 335 ~3 sRBS-Lum-dT (2) G3 965 ~2 - Make a 1:10 dilution of sRBS-Lum-dt maxiprep (D8) and sRBS-Lum-dT (B7). 0.5µL pDNA in 4.5µL water.
- Cut pLacI with EcoRI and SpeI
- Cut sRBS-Lum-dT with XbaI and PstI
- Cut pSB1T3 with EcoRI and PstI
- Will have total of 12 ligation reactions, want 12x2µL of pSB1T3 to add to each, therefore want 25µL of pSB1T3.</ol> Method:
Experimental Setup
Name [pDNA] (ng/µL) Volume
pDNA (µL)Volume
Water (µL)Volume
Buffer (µL)Enzymes Total Volume sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50 sRBS-Lum-dT (A1) 965 1 43.5 5 0.25µL XbaI
0.25µL PstI50
June 3/2010
Objective: Ligate pLacI to sRBS-Lum-dT using the three antibiotic assembly method (according to Tom Knight's protocol). Previous ligation had miniscule quantities of DNA in ligation, not surprising it didn't work.
- Restrict rbs-xylE wit EcoRI and SpeI (Red Buffer)
 "
"


 ]
]
 ]
]
 ]
]
 ]
]
 ]
]
 ]
]