Team:SDU-Denmark/labnotes2
From 2010.igem.org
(→Group: Flagella) |
(→Group: Flagella) |
||
| Line 477: | Line 477: | ||
--[[User:Tipi|Tipi]] 18:15, 19 July 2010 (UTC)<br><br> | --[[User:Tipi|Tipi]] 18:15, 19 July 2010 (UTC)<br><br> | ||
| - | === Coloni PCR of flhD/C in pSB1C3 === | + | === Coloni PCR of flhD/C (native coding sequence) in pSB1C3 === |
Start date: 21/07 End date: 21/07<br> | Start date: 21/07 End date: 21/07<br> | ||
''Methods:'' Coloni PCR and gel electrophoresis <br><br> | ''Methods:'' Coloni PCR and gel electrophoresis <br><br> | ||
| Line 611: | Line 611: | ||
--[[User:Tipi|Tipi]] 11:46, 24 July 2010 (UTC) <br><br> | --[[User:Tipi|Tipi]] 11:46, 24 July 2010 (UTC) <br><br> | ||
| - | === Coloni PCR of flhD/C in pSB1C3 === | + | === Coloni PCR of flhD/C (native coding sequence) in pSB1C3 === |
Start date: 22/07 End date: 22/07<br> | Start date: 22/07 End date: 22/07<br> | ||
''Methods:'' Coloni PCR and gel electrophoresis <br><br> | ''Methods:'' Coloni PCR and gel electrophoresis <br><br> | ||
| Line 795: | Line 795: | ||
The 2 bands at app. 100bp and 200bp respectively indicates that the E and S restiction sites have been preserved, suggesting that a ligation has occured.<br> | The 2 bands at app. 100bp and 200bp respectively indicates that the E and S restiction sites have been preserved, suggesting that a ligation has occured.<br> | ||
An ON culture of coloni 4 and 9 is made to use for mini-prep.<br> | An ON culture of coloni 4 and 9 is made to use for mini-prep.<br> | ||
| - | --[[User:Tipi|Tipi]] 14:37, 24 July 2010 (UTC) | + | --[[User:Tipi|Tipi]] 14:37, 24 July 2010 (UTC)<br><br> |
| + | |||
| + | === Digestion of flhD/C mut with PstI === | ||
| + | Start date: 21/07 End date: 21/07<br> | ||
| + | ''Methods:'' Restriction digest and gel electrophoresis <br><br> | ||
| + | ''Protocol:''[https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1] <br><br> | ||
| + | ''Experiment done by:'' Maria<br><br> | ||
| + | ''Notes:''<br> | ||
| + | To verify if the silent mutation has been made PCR product of flhD/C mut (see [[https://2010.igem.org/Team:SDU-Denmark/labnotes2#Amplification_of_FlhDCmut flhD/C mut PCR]] was digested with restiction enzyme PstI.<br> | ||
| + | If the mutation has been made the flhD/C fragment should not be digested with PstI.<br> | ||
| + | PCR product of flhD/C (native coding sequence) was used as controle.<br> | ||
| + | Undigested PCR product of flhD/C (native coding sequence) was loaded onto the gel as well.<br>Samples were loaded onto a 2% agarose gel. Hyper Ladder II was used as marker.<br> | ||
| + | Loading scheme:<br> | ||
| + | <table style="text-align: left; width: 100%;" border="1" cellpadding="2" | ||
| + | cellspacing="2"> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Lane<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">1<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">2<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">3<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;"><br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">Digested flhD/C (native coding sequence) <br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">Digested flhD/C mut<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">Undigested flhD/C (native coding sequence)<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table><br><br> | ||
| + | ''Results:''<br> | ||
| + | Gel electrophoresis:<br> | ||
| + | [[Image:Team-SDU-Denmark-Digestion test flhD,C.jpg|600px]]<br><br> | ||
| + | ''Analysis:''<br> | ||
| + | The intensity of the bands are too low to conclude anything. The experiment was remade using a gel with smaller wells.<br> | ||
| + | --[[User:Tipi|Tipi]] 14:37, 24 July 2010 (UTC)<br><br> | ||
===Purification of pSB1A2 containing J13002=== | ===Purification of pSB1A2 containing J13002=== | ||
<br> | <br> | ||
Revision as of 15:37, 24 July 2010
Lab notes (7/19 - 7/25)
Group: Flagella
Annealing of the two mutated strands of FlhDC (FlhDCmut)
Experiment done by: Sheila
Date: July 19th
Protocol: CP1.1
Method: PCR of the two mutated strands of the FlhDC operon
Notes: To samples were run at two different temperatures: 56,1˚C and 64,5˚C respectively.
Polymerase used: Pfu
Primers used: None, as the two strands are supposed to anneal to each other
| Pfu buffer |
5,0uL |
| dNTP |
1,5uL |
| FlhDC mutated long
strand |
1,5uL |
| FlhDC mutated
short strand |
1,5uL |
| H2O |
41,0uL |
| Pfu |
1,0uL |
The samples were run in the following PCR program:
| Program |
1st sample |
2nd sample |
Time |
| 1: Start |
94˚C |
94˚C |
3min |
| 2: Denaturing |
94˚C |
94˚C |
2min |
| 3: Annealing |
56,1˚C |
64,5˚C |
30sec |
| 4: Elongation |
72˚C |
72˚C |
2min |
| 5: |
GOTO |
Repeat |
X29 |
| 6: End |
72˚C |
72˚C |
2min |
| 7: Hold |
4˚C |
4˚C |
Results: A gel, containing the PCR product was run, using [http://www.fermentas.de/product_info.php?info=p1110 GeneRuler(TM) DNA Ladder Mix, ready-to-use (#SM0333)] loaded in lane one (from the left:
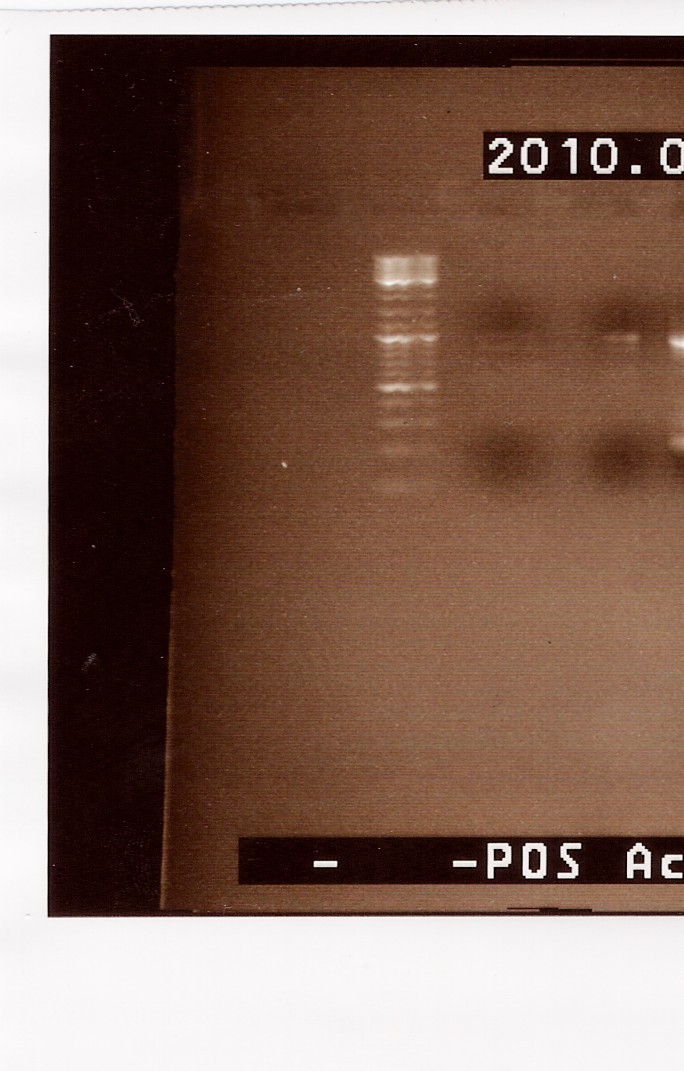
We can se very faint bands in lane 2 and 3, at around 1000bp. The band in lane 3 is a little more visible than lane 2. Because we haven't used primers ind this PCR, it is possible that the two mutated strands has a lower possibility of annealing to each other. It s however possible to amplify the few complete FlhDCmut strands in a new PCR, which is what we will do next.
--Sheila 09:10, 22 July 2010 (UTC)
Amplification of FlhDCmut
Experiment done by: Sheila
Date: July 19th
Protocol: CP1.1
Method: The experiment was done in two rounds. First, the annealing product, made by Pernille on july 19th was amplified. The we amplified Pernilles amplification simultaneously as we amplified Sheilas previous annealing.
Primers used: FlhDC fw and FlhDC rev
Polymerase used: Pfu
Notes:
Results:
Gel extraction of FlhDCmut
Experiment done by: Sheila
Date: July 20th
Protocol:
Method:
Notes:
Results:
Restriction digest of FlhDCmut and pSB1C3
Experiment done by: Sheila
Date: July 20th
Protocol:
Method:
Notes:
Results:
Gel extraction of FlhDCmut and pSB1C3
Experiment done by: Sheila
Date: July 20th
Protocol:
Method:
Notes:
Results:
Amplification of FlhDCmut
Experiment done by: Sheila
Date: July 20th-21st
Protocol: CP1.1
Method:
Notes:
Results:
Gel extraction of FlhDCmut
Experiment done by: Sheila
Date: July 21st
Protocol:
Method:
Notes:
Results:
Gel extraction of flhD/C (native coding sequence)
Start date: 19/07 End date: 19/07
Methods: Gel extraction, nanodrop
Protocol:DE1.1
Experiment done by:Maria
Notes:
70uL of flhD/C PCR product from Amplification of flhD/C was loaded onto a 1.5 agarose gel and extracted according to protocol.
DNA was eluted in 20uL elution buffer.
Results:
Nanodrop:
| Sample ID |
ng/uL |
260/280 |
260/230 |
| flhDC 1 |
19.82 |
2.43 |
0.08 |
| flhDC 2 |
25.24 |
2.22 |
0.12 |
Analysis:
Nanodrop measurements indicated a possible contamination. However the DNA was pooled and used for Digestion
--Tipi 17:22, 19 July 2010 (UTC)
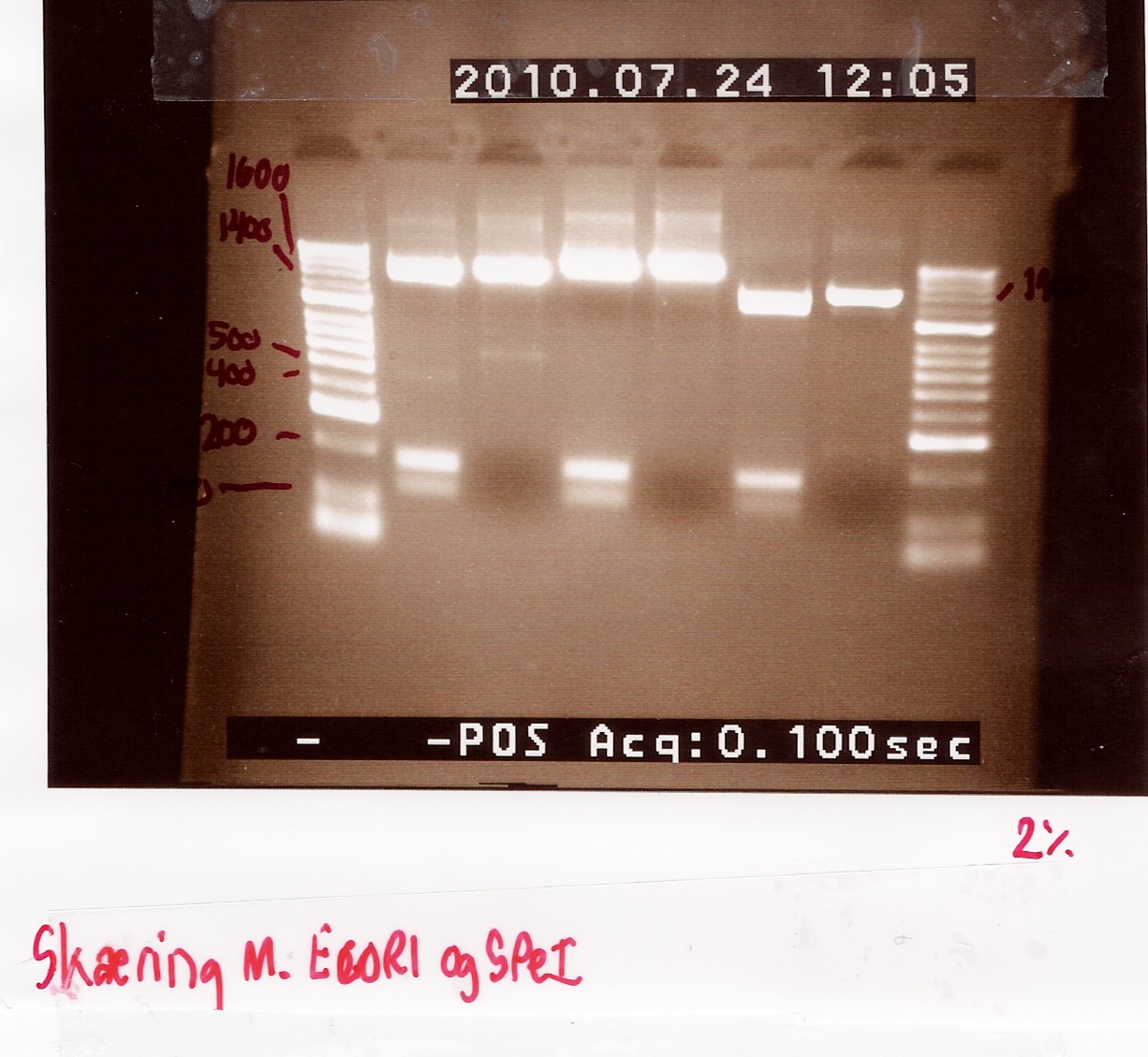
Digestion of flhD/C (native coding sequence) and pSB1A2 with EcoRI SpeI
Start date: 19/07 End date: 19/07
Methods: Digestion, Gel electrophoresis
Protocol:RD1.1
Experiment done by:Maria
Notes:
purified pSB1C3 (tube 18 blue) and flhD/C from Gel extraction was digested with EcoRI and SpeI.
Restriction mixture:
| flhD/C |
pSB1C3 |
|
| H2O |
30uL |
24uL |
| EcoRI |
4uL |
2uL |
| SpeI |
4uL |
2uL |
| FD green buffer |
8uL |
4uL |
| DNA |
38uL |
10uL |
| total vol. |
84 |
42 |
samples were loaded onto a 1.5 agarose gel. Gene ruler red were used as marker.
Results:
Gel electrophoresis:
Analysis:
In lane 2 containing pSB1C3 2 bands are detected indicating a succesful digestion of the plasmid (the band at 1000 bp corresponds to J04450). A succesful digestion of the flhD/C cannot be concluded from the gel. However both bands was excised and extracted from gel (Gel extraction)
--Tipi 17:42, 19 July 2010 (UTC)
Gel extraction of digested flhD/C (native coding sequence) and pSB1C3
Start date: 19/07 End date: 19/07
Methods: Gel extraction, nanodrop
Protocol:DE1.2
Experiment done by:Maria
Notes:
Digested flhD/C and pSB1C3 from Digestion were extracted from gel according to protocol.
DNA was eluted in 20uL H20.
Results:
Nanodrop:
| Sample ID |
ng/uL |
260/280 |
260/230 |
| flhD/C |
18.18 |
4.59 |
0.18 |
| pSB1C3 |
14.30 |
1.89 |
0.04 |
Analysis:
nanodrop measurements indicated contamination. However both samples were used for Ligation.
--Tipi 17:57, 19 July 2010 (UTC)
Ligation of flhD/C (native coding sequence) and pSB1C3
Start date: 19/07 End date: 20/07
Methods: Ligation
Protocol:LG1.2
Experiment done by:Maria
Notes:
3 ligation reactions was prepared.
| LG1 |
LG2 |
LG3 |
|
| 10x T4 ligase buffer |
2uL |
2uL |
2uL |
| flhD/C |
5uL |
5uL |
5uL |
| pSB1C3 |
1uL |
2.8uL |
5uL |
| H20 |
11uL |
9.2uL |
7uL |
| T4 ligase |
1uL |
1uL |
1uL |
Ligation mixtures were not run on gel but were directly used for transformation
surplus ligation mixture was stored at -20 degrees as L1, L2 and L3.
--Tipi 18:15, 19 July 2010 (UTC)
Coloni PCR of flhD/C (native coding sequence) in pSB1C3
Start date: 21/07 End date: 21/07
Methods: Coloni PCR and gel electrophoresis
Protocol:CP1.2
Experiment done by: Maria and LC
Notes:
Coloni PCR was made on flhD/C in pSB1C3 from Transformation.
10 colonies from plates plated with cells transformed with L2 and L3 ligation mixtures (see Ligation).
Coloni 1-5 is taken from L2 plates
coloni 6-10 is taken from L3 plates
PCR was made with only half the amount of premix.
Premix for 12 PCR reactions:
| 10xtaq buffer |
30uL |
| MgCl2 |
12uL |
| VF2 |
12uL |
| VR |
12uL |
| dNTP |
6uL |
| H2O |
42uL |
| Total vol. |
114uL |
0.5uL Taq polymerase from ampliqon was used.
Taq PCR program:
| 1:Start |
94C |
2 min |
| 2: Denaturing |
94C |
1 min |
| 3: Annealing |
55C |
1 min |
| 4: Elongation |
72C |
1 min 30 s |
| 5: |
GO TO |
rep. 29x |
| 6: End |
72C |
3 min |
| 7: Hold |
4C |
PCR product was loaded onto a 2% agarose gel. Gene ruler DNA ladder mix (red) was used as marker.
VF2 - coding sequence (in pSB1C3) : 140bp
Coding sequence - VR (in pSB1C3) : 176bp
flhD/C : 932bp
VF2 - VR for pSB1C3 w. flhD/C :1248bp
VF2 - VR for pSB1C3 without insertion : 316bp
Results:
Gel electrophoresis:
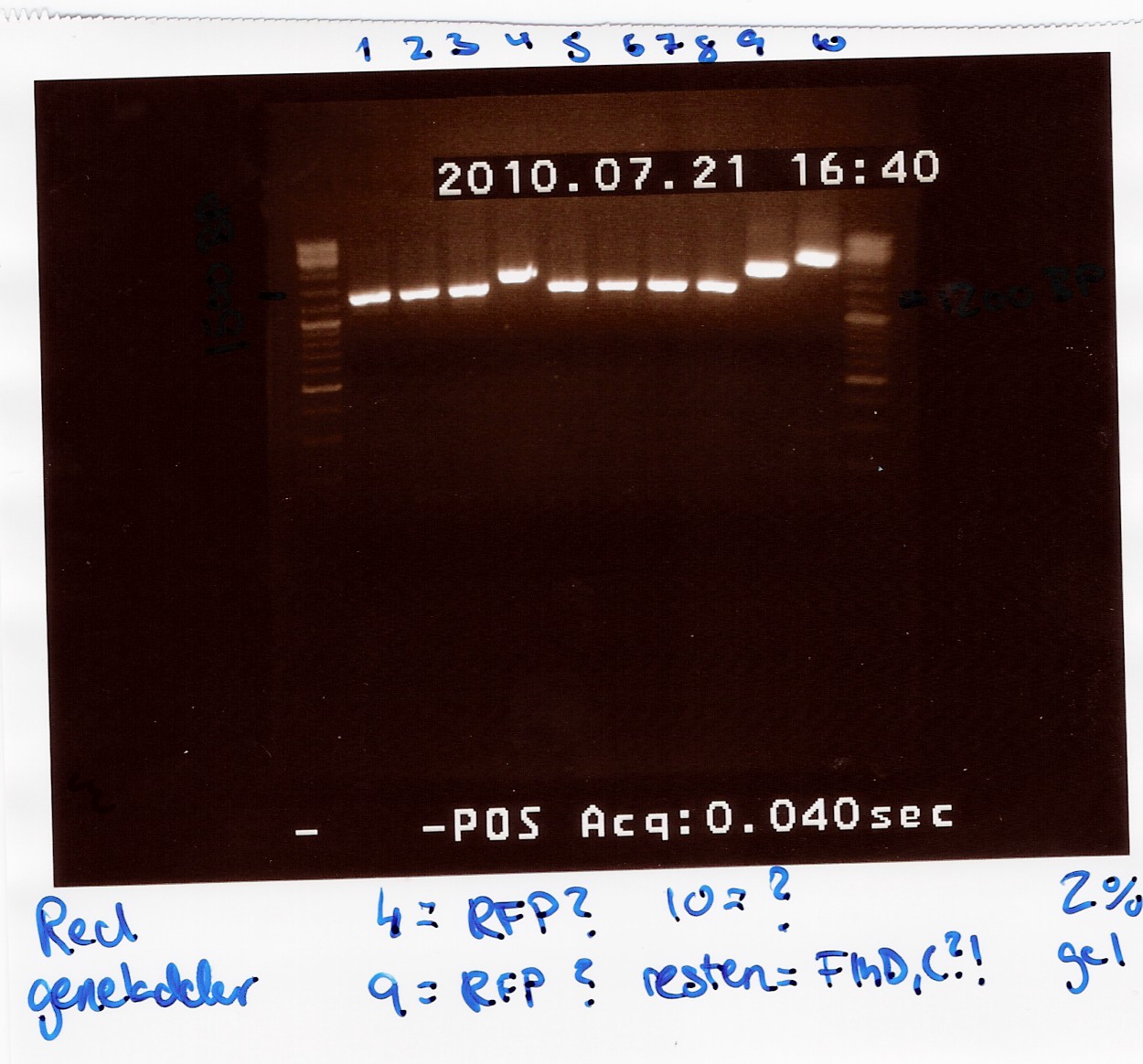
Analysis:
All lanes except 4, 9 and 10 showed a band at app. 1200bp. which might indicate plasmids where flhD/C has been inserted.
Lane 4 and 9 has a band at app. 1500bp which could indicate undigested pSB1C3 (VF2 - VR for pSB1C3 w. J04450 is 1385bp).
Streak plates of coloni 1, 2, 3, 5, 6, 7, 8 and 10 showed red colonies, indicating that these contained undigested pSB1C3.
Streak plates of coloni 4 and 9 had only white colonies, suggesting an insertion has occured.
To verify if flhD/C has been inserted coloni PCR using VF2 and flhD/C rev. as primers was carried out for colonies from streak plates of coloni 4 and 9 and of all white colonies from the transformation plates. (see coloni PCR)
--Tipi 11:46, 24 July 2010 (UTC)
Coloni PCR of flhD/C (native coding sequence) in pSB1C3
Start date: 22/07 End date: 22/07
Methods: Coloni PCR and gel electrophoresis
Protocol:CP1.3
Experiment done by: Maria
Notes:
Coloni PCR was made on flhD/C in pSB1C3 from Transformation.
All remaining white colonies from plates plated with cells transformed with L2 and L3 ligation mixtures (see Ligation) was selected.
coloni 1-6 is taken from L2.3 plates
coloni 7-15 is taken from L3.2 plates
coloni 16-27 is taken from L3.3 plates
coloni 28 is taken from streak plate of PCR no. 4
coloni 29 is taken from streak plate of PCR no. 9 (see Coloni PCR)
Premix for 31 PCR reactions:
| 10xtaq buffer |
77.5uL |
| MgCl2 |
31uL |
| VF2 |
31uL |
| flhD/C rev. |
31uL |
| dNTP |
15.5uL |
| H2O |
108.5uL |
| Total vol. |
294.5uL |
0.5uL Taq polymerase from fermentas was used.
Taq PCR program:
| 1:Start |
94C |
2 min |
| 2: Denaturing |
94C |
1 min |
| 3: Annealing |
55C |
1 min |
| 4: Elongation |
72C |
1 min 30 s |
| 5: |
GO TO |
rep. 29x |
| 6: End |
72C |
3 min |
| 7: Hold |
4C |
PCR product was loaded onto a 2% agarose gel. Gene ruler DNA ladder mix (red) was used as marker.
Analysis:
None of the samples contained amplified DNA.
This indicates either that the flhD/C gene has not been inserted to the pSB1C3 backbone or an insertion has been made but the flhD/C primer has not been able to anneal to the template.
To verify that a ligation has occurred, PCR product 4 and 9 from Coloni PCR was digested with EcoRI and SpeI.
--Tipi 13:53, 24 July 2010 (UTC)
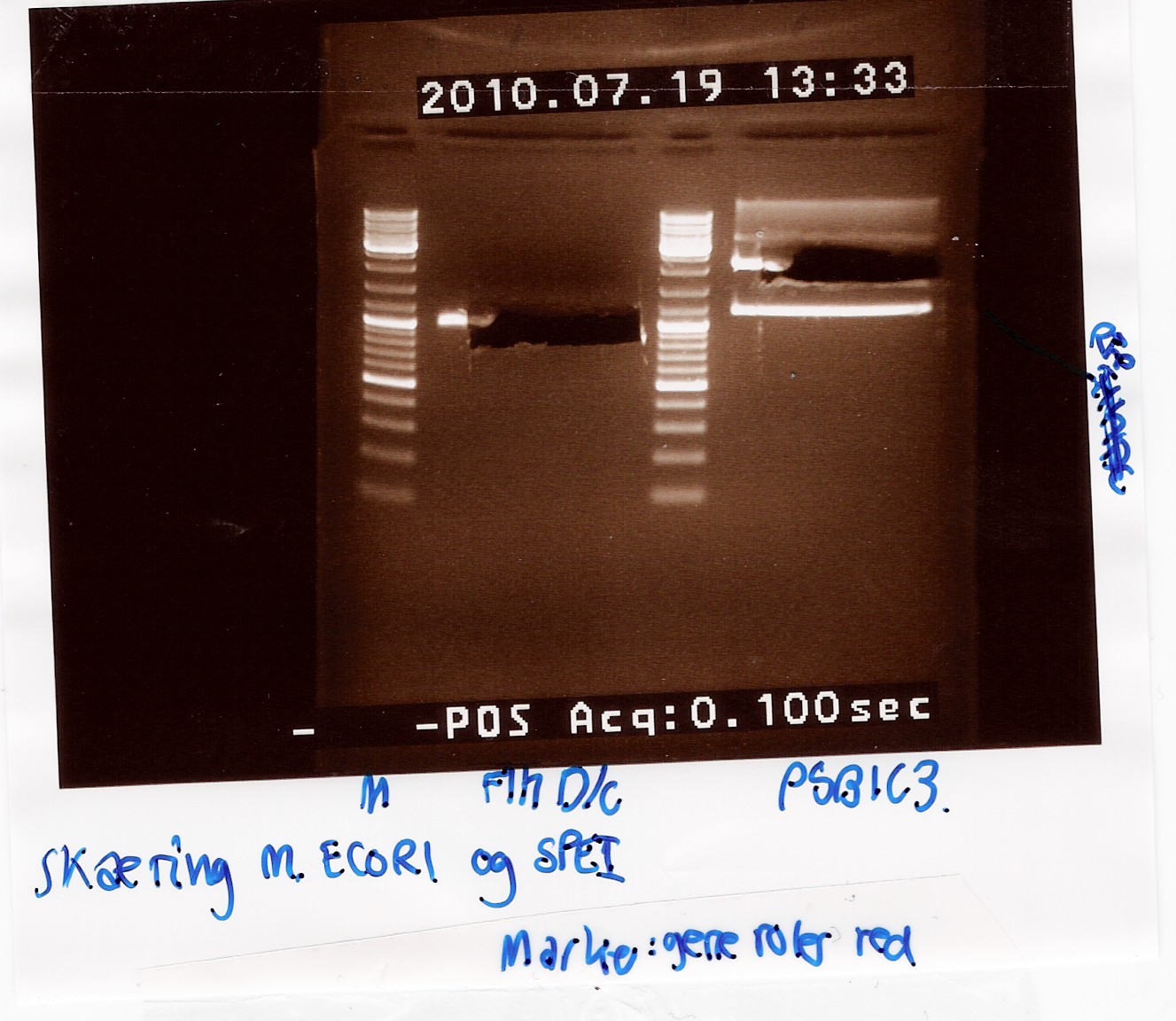
Digestion of flhD/C PCR product with EcoRI and SpeI
Start date: 24/07 End date: 24/07
Methods: Restriction digest and gel electrophoresis
Protocol:RD1.1
Experiment done by: Maria
Notes:
To verify if a ligation has occurred Taq PCR product of coloni 4 and 9 (see Coloni PCR) was digested with the same restiction enzymes used for the digestion of flhD/C DNA and pSB1C3 prior to ligation.
If an ligation has occurred, then the restriction sites of EcoRI and SpeI should be intact, and two fragments of 122bp and 170bp should be observed on the gel, indicating VF2-E site and S site-VR respectively.
RFP (PCR poduct of coloni no.8) was used as controle.
2.5uL of undigested PCR product of coloni 4, 8 and 9 were loaded onto the gel as well.
Samples were loaded onto a 2% agarose gel. Hyper Ladder II was used as marker.
Loading scheme:
| Lane |
1 |
2 |
3 |
4 |
5 |
6 |
| digested no. 4 |
undigested no. 4 |
digested no. 9 |
undigested no. 9 |
digested no. 8 |
undigested no. 8 |
Results:
Gel electrophoresis:
Analysis:
The 2 bands at app. 100bp and 200bp respectively indicates that the E and S restiction sites have been preserved, suggesting that a ligation has occured.
An ON culture of coloni 4 and 9 is made to use for mini-prep.
--Tipi 14:37, 24 July 2010 (UTC)
Digestion of flhD/C mut with PstI
Start date: 21/07 End date: 21/07
Methods: Restriction digest and gel electrophoresis
Protocol:RD1.1
Experiment done by: Maria
Notes:
To verify if the silent mutation has been made PCR product of flhD/C mut (see [flhD/C mut PCR] was digested with restiction enzyme PstI.
If the mutation has been made the flhD/C fragment should not be digested with PstI.
PCR product of flhD/C (native coding sequence) was used as controle.
Undigested PCR product of flhD/C (native coding sequence) was loaded onto the gel as well.
Samples were loaded onto a 2% agarose gel. Hyper Ladder II was used as marker.
Loading scheme:
| Lane |
1 |
2 |
3 |
| Digested flhD/C (native coding sequence) |
Digested flhD/C mut |
Undigested flhD/C (native coding sequence) |
Results:
Gel electrophoresis:
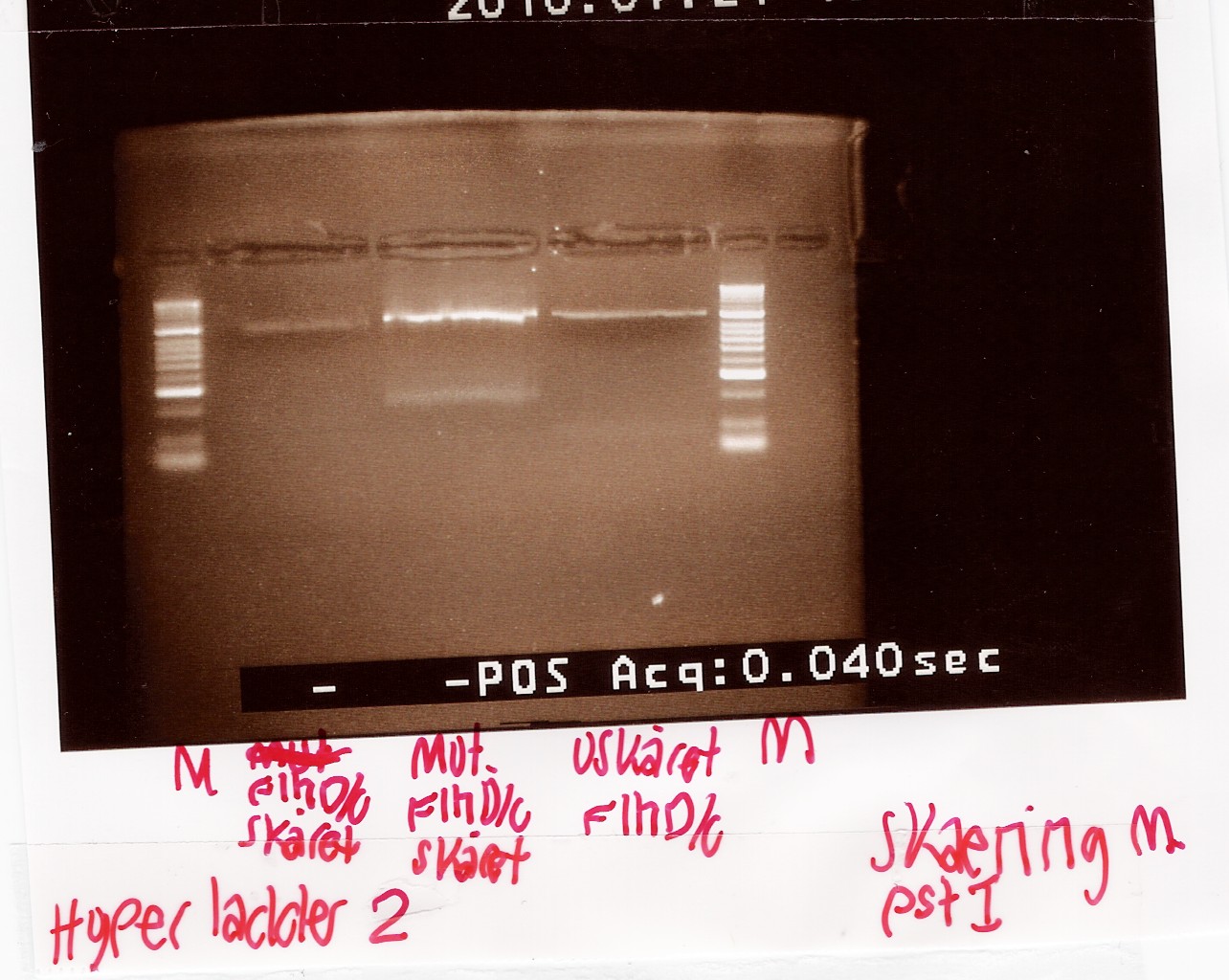
Analysis:
The intensity of the bands are too low to conclude anything. The experiment was remade using a gel with smaller wells.
--Tipi 14:37, 24 July 2010 (UTC)
Purification of pSB1A2 containing J13002
Experiment done by: Pernille
Date: the 19 of July
Protocol: MP1.1 and CP1.1
Method: Miniprep, Nano drop, gel electrophoresis and PCR
Notes: The pSB1A2 plasmid are a high-copy-plasmid and therefore 5mL ON culture are used for purification of the plasmid. for the PCR the following PRC program are used:
1: 94°C 3min
2: 94°C 2min
3: 55°C 30 sec
4: 72°C 1min
5: x 29 rep.
6: 72°C 5min
7: hold 4°C
Results:After the purification the concentration of nucleic acid concentration was measured on the Nano Drop. There was made four purification tubes. Only tube 1 and 3 was used for further experiments because the other contained a very low concentration of nucleic acid. The concentration in tube 1 and 3 was 25,72ng/uL and 25,76ng/uL.
The purified DNA was loaded in a 1,5% agarose gel together with a 10.000bp long ladder (red).

PCR was run on the DNA using the VR and VF2 primers wich ensures the only the brick part are amplificated. The pfu polymerase was used but unfortunately i did not get any results since i did not add any MgSO4 to the premix.
--Pernm07 08:48, 22 July 2010 (UTC)
Amplification of pSB1A2 w. J13002, pSB1A2 w. B0015 and pSB1A2 w. B0034
Experiment done by: Pernille
Date: The 20th of July
Protocol: CP1.1and DNA extraction
Method: PCR and DNA extraction
Notes: The purified DNA was amiplificated with PRC using the following program:
1: 94°C 3min
2: 94°C 2min
3: 55°C 30 sec
4: 72°C 1min
5: x 29 rep.
6: 72°C 5min
7: hold 4°C
the experiment for each biobrick was do in duplicats.
Results:After amplification the nucleic acid concentration was measured on the Nano Drop. The concentrations was measued to:
tube 1: pSB1A2 w. J13002 was 825.2ng/uL tube 2: pSB1A2 w. B0015 was 547.76ng/mL tube 3: pSB1A2 w. B0034 was 777.84ng/uL
The PCR product was loaded on a 2% agarose gel together with a 1000 bp long marker. xxxxxxxxxx
During the DNA extraction 50uL DNA anf 7uL loading buffer was loaded in a lane. The samples was loaded from left to right in continuous numbers.
xxxxxxx
The concentration of the extracted DNA from tube 1,2 and was found to 13.60ng/uL, 16,58ng/uL and 12,6313.60ng/uL respectively. while we have to use the dobbelt terminator and the constitutive active promotor in a lot of experiment the next step is to do another round of PRC and DNA extraction on thise biobricks.
--Pernm07 10:06, 22 July 2010 (UTC)
Group: Photosensor
Miniprep of pSB3T5 w. J04450 and pSB3C5 w. J04450 from transformation 20/7
Start date: 22/07 End date: 22/07
Methods: Miniprep, gel electrophoresis and nano drop
Protocol:MP1.2
Experiment done by: LC
Notes: /
Results:
Gel electrophoresis:
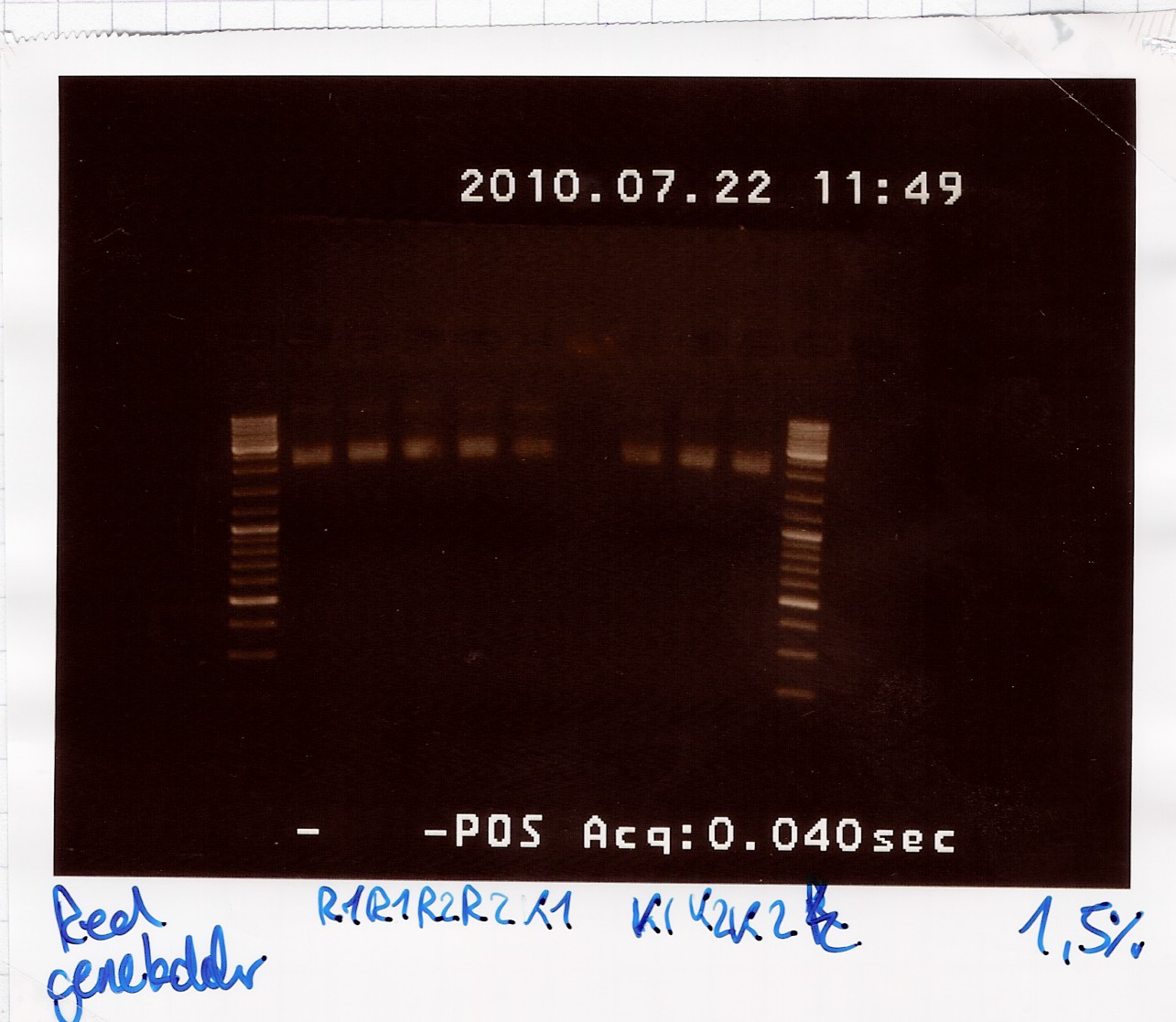
Nanodrop:
The concentration was around 23 ng/ul for all samples. Not overwhelming, but usable.
Analysis:
Sample 1-4 of BBa_K081005 were pooled and frozen and sample 1-4 of BBa_R0011 were too.
Group: Retinal
Transformation of K081005 in pSB1A2 (constitutive promoter and RBS combined),R0011 in pSB1A2, pSB3C5 w. J04450 and pSB3T5 w. J04450 in Top 10 E.Coli
Start date: 19/07 End date: 20/07
Methods: ON culture, making competent cells, transformation
Protocol:CC1.1 TR1.1
Experiment done by: Maria
Notes:ON colony was made of 110 ml lb medium inoculated with a top10 coloni.
| Time |
Optical density |
| 8:12 |
2.9 |
| 8:17 |
0.02 |
| 9:17 |
0.035 |
| 10:17 |
0.204 |
| 10:40 |
0.380 |
| 10:50 |
0.49 |
pSB1A2 w. R0011 and pSB1A2 w. K081005 was plated with 150uL on plates containing LA, LA + Amp, LA + Tetracycline, LA + Chloramphenicol and LA + Kanamycine.Upconcentration of these samples was not made. pSB3T5 w. J04450 and pSB3C5 w. J04450 was plated according to protocol
Results:
Analysis:
Both pSB3T5 and pSB3C5 was succesfully transformed, and ON cultures with appropiate antibiotics were made for mini-prep.
For pSB1A2 w. R0011 only 6 colonies was observed on the LA+amp plate.
For pSB1A2 w. K081005 only 4 colonies was observed on the LA+amp plate.
All 10 colonies were used for Coloni PCR.
--Tipi 16:33, 19 July 2010 (UTC)
Transformation of flhD/C in pSB1C3 and test plasmid in Top 10 E.Coli
Start date: 20/07 End date: 19/07
Methods: ON culture, making competent cells, transformation
Protocol:CC1.1 TR1.1
Experiment done by: Maria
Notes:
Ligated flhD/C from Ligation and test plasmid from Whatman was transformed.
ON colony was made of 25 ml lb medium inoculated with a top10 coloni.
| Time |
Optical density |
| 8:17 |
3.5 |
| 8:20 |
0.02 |
| 9:15 |
0.035 |
| 10:30 |
0.222 |
| 10:52 |
0.350 |
| 11:05 |
0.52 |
Compotent cells were washed using 10mL 50mM CaCl2.
3 parallel transformations were carried out for L1, L2 and L3 respectively (see Ligation). L1.1, L1.2, L2.1, L2.2 and L3.1 were transformed using compotent cells from 19/7 (Compotent cells).
Prior to transformation test plasmid was washed with 10xTE pH 8.0.
Results:
Analysis:
Test plasmid was succelsfully transformed and we can carry on transforming the plasmid containing the NinaB gene.
Colonies was seen on plates transformed with L2 and L3. 10 colonies were selected and used for coloni PCR.
--Tipi 08:17, 21 July 2010 (UTC)
Coloni PCR of R0011 in pSB1A2 and K081005 in pSB1A2
Start date: 20/07 End date: 20/07
Methods: Coloni PCR and gel electrophoresis
Protocol:CP1.2
Experiment done by: Maria and LC
Notes:
6 colonies transformed with pSB1A2 w. R0011 and 4 colonies transformed with pSB1A2 w. K081005 (see Transformation) was used for coloni PCR.
Colonies with K081005 was denoted A1, A2 A3 and A4 respectively.
Colonies with R0011 was denotes B1, B2, B3, B4, B5 and B6 respectively
PCR was made with only half the amount of premix.
Premix for 12 PCR reactions:
| 10xtaq buffer |
30uL |
| MgCl2 |
12uL |
| VF2 |
12uL |
| VR |
12uL |
| dNTP |
6uL |
| H2O |
42uL |
| Total vol. |
114uL |
0.5uL Taq polymerase from ampliqon was used.
Taq PCR program:
| 1:Start |
94C |
2 min |
| 2: Denaturing |
94C |
1 min |
| 3: Annealing |
55C |
1 min |
| 4: Elongation |
72C |
30 s |
| 5: |
GO TO |
rep. 29x |
| 6: End |
72C |
3 min |
| 7: Hold |
4C |
PCR product was loaded onto a 2% agarose gel. Gene ruler 100bp DNA ladder was used as marker.
Results:
Gel electrophoresis:
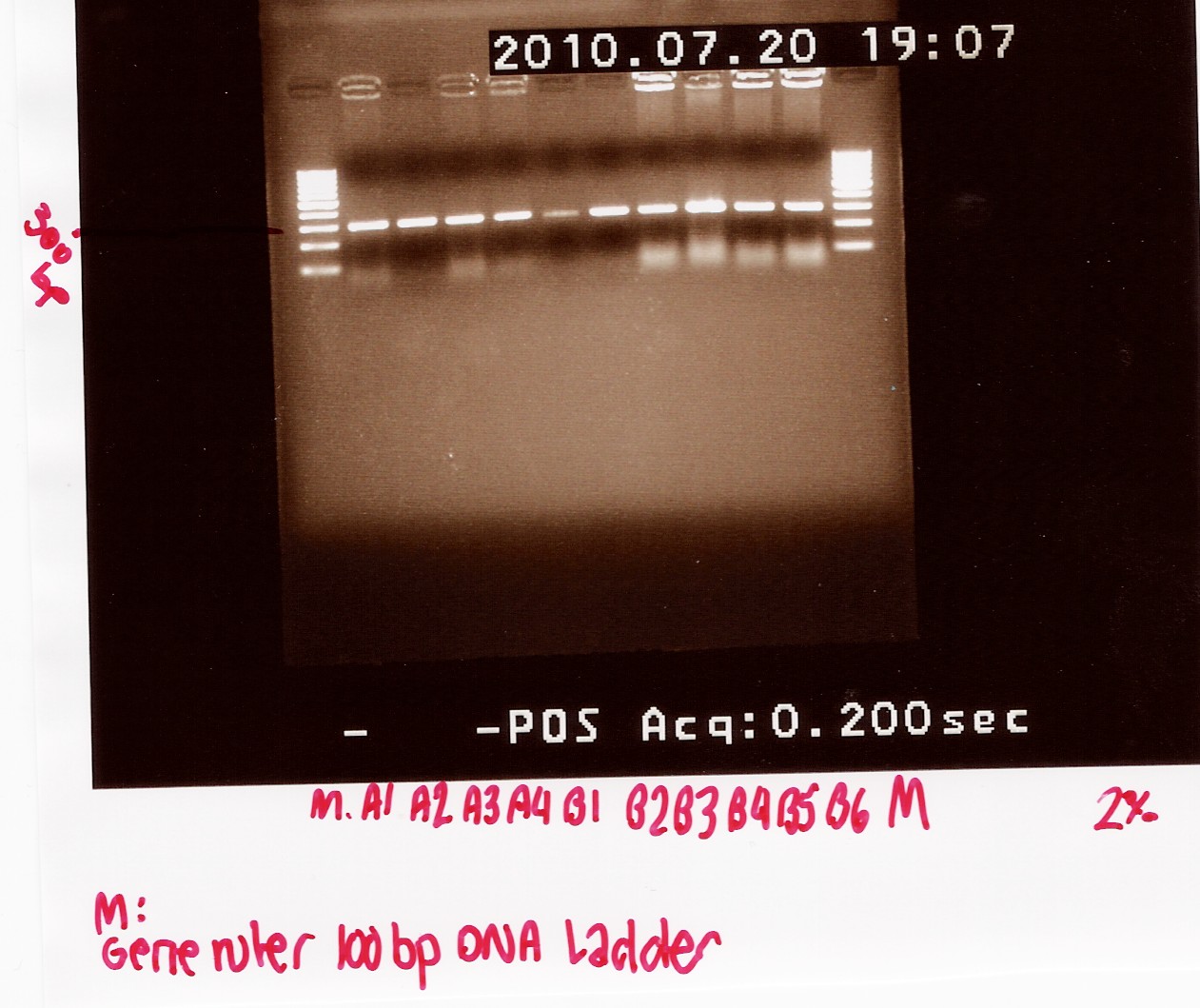
Analysis:
A band at app. 300bp was observed in all lanes, indicating that all 10 colonies contained the correct plasmids.
ON cultures were made for mini-prep.
--Tipi 06:35, 21 July 2010 (UTC)
Miniprep of pSB3T5 w. J04450 and pSB3C5 w. J04450 from transformation 20/7
Start date: 21/07 End date: 21/07
Methods: Miniprep, gel electrophoresis and nano drop
Protocol:MP1.2
Experiment done by: Maria and LC
Notes:
For pSB3T5 no.3 one half of the material was lost during the neutralization step.
pSB3T5 no.1 was eluted in 200uL elution buffer.
Samples were loaded onto a 1.5 agarose gel. Generuler DNA ladder-mix (red) was used as marker
Results:
Gel electrophoresis:
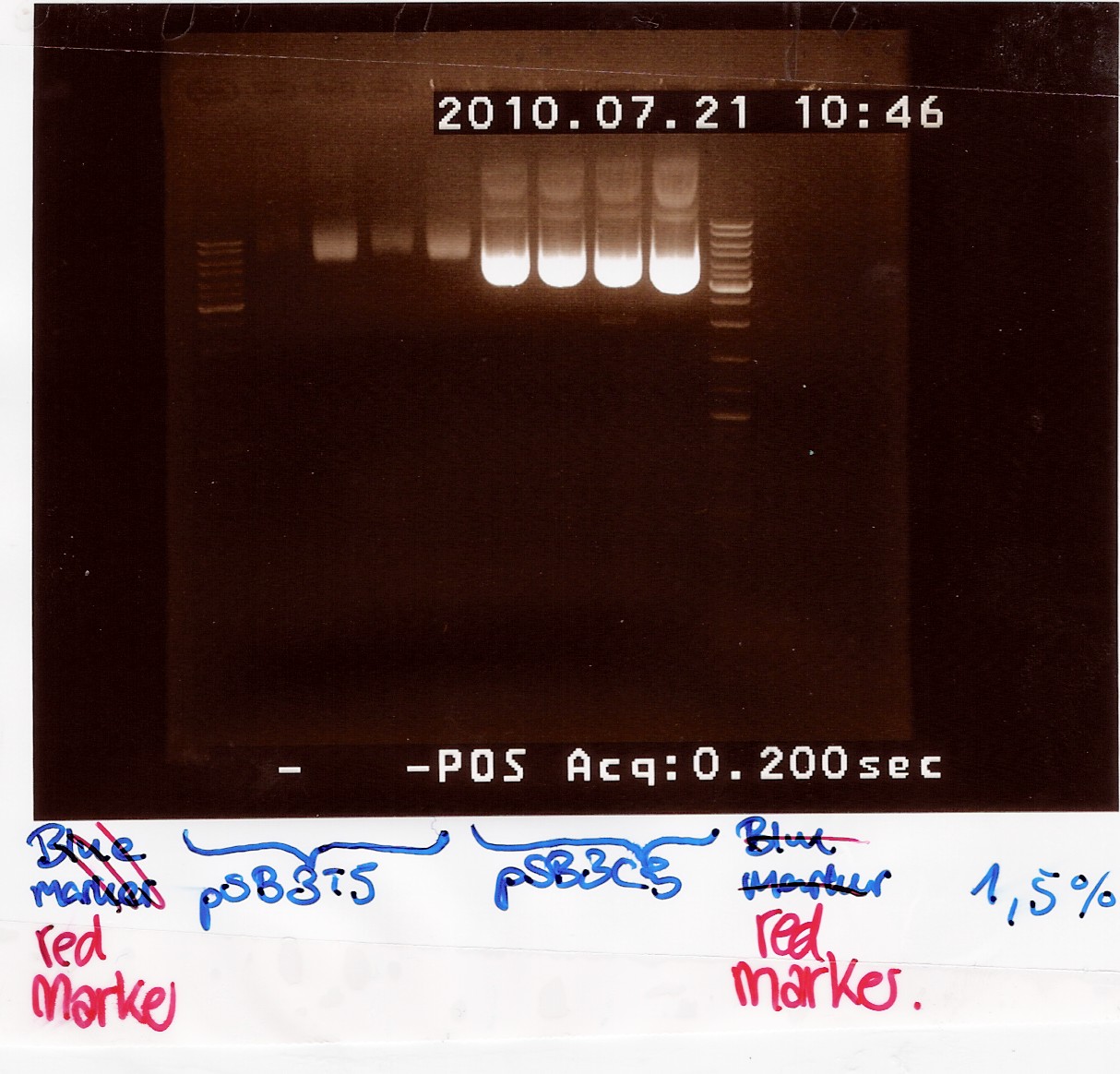
Nanodrop:
| Sample ID |
ng/uL |
260/280 |
260/230 |
| pSB3C5 1 |
208.2 |
1.93 |
2.31 |
| pSB3C5 2 |
225.09 |
1.93 |
2.3 |
| pSB3C5 3 |
215.2 |
1.93 |
2.3 |
| pSB3C5 4 |
260.68 |
1.93 |
2.33 |
| pSB3T5 1 |
14.16 |
2.06 |
2.0 |
| pSB3T5 2 |
52.50 |
1.96 |
2.15 |
| pSB3T5 3 |
26.45 |
1.99 |
2.3 |
| pSB3T5 4 |
53.43 |
2.0 |
2.19 |
Analysis:
Both pSB3T5 and pSB3C5 was succesfully purified.
pSB3C5 1-4 was pooled and stored as no.28 (white) at -20 degrees.
pSB3T5 1+3 was pooled and stored as no.30 (white) at -20 degrees.
pSB3T5 2+4 was pooled and stored as no.29 (white) at -20 degrees.
Nanodrop of pooled samples:
| Sample ID |
ng/uL |
260/280 |
260/230 |
| pSB3C5 1-4 |
256.71 |
1.92 |
2.3 |
| pSB3T5 1+3 |
54.01 |
1.95 |
2.19 |
| pSB3T5 2+4 |
16.0 |
1.88 |
2.11 |
--Tipi 09:41, 21 July 2010 (UTC)
Transformation of TOP 10 e. coli with ninaB gene in pOT2:chlor vector
Date: 21/7
Done by: Christian
Transformation of cells with gene in plasmid
Date: 21/7
Methods: Transformation
Protocols: TR1.1 and paperblotter protocol from plasmid cDNA package.
Notes: Filterpaper was washed with 50ul TE buffer for 2 seconds. Paper punch was then transfered to a sterile eppendorff tube, and transformation protocol TR1.1 was followed from here. The usual positive control was used.
Plates were dried for ca. 12 minutes, showed no sign of cracking.
The gene is inserted into a pOT2 plasmid. A plasmid map can be found [http://www.fruitfly.org/about/methods/pOT2vector.html here]:
Two of the chlor plates were damaged (one very slightly) and one ampicilin plate was torn up, again. (good thing two were dried). Plates were incubated ON at 37°.
Coloni PCR of flhD/C in pSB1C3
Start date: 21/07 End date: 21/07
Methods: Coloni PCR and gel electrophoresis
Protocol:CP1.2
Experiment done by: Maria and LC
Notes:
Coloni PCR was made on flhD/C in pSB1C3 from Transformation.
10 colonies from plates plated with cells transformed with L2 and L3 ligation mixtures (see Ligation).
Coloni 1-5 is taken from L2 plates
coloni 6-10 is taken from L3 plates
PCR was made with only half the amount of premix.
Premix for 12 PCR reactions:
| 10xtaq buffer |
30uL |
| MgCl2 |
12uL |
| VF2 |
12uL |
| VR |
12uL |
| dNTP |
6uL |
| H2O |
42uL |
| Total vol. |
114uL |
0.5uL Taq polymerase from ampliqon was used.
Taq PCR program:
| 1:Start |
94C |
2 min |
| 2: Denaturing |
94C |
1 min |
| 3: Annealing |
55C |
1 min |
| 4: Elongation |
72C |
1 min 30 s |
| 5: |
GO TO |
rep. 29x |
| 6: End |
72C |
3 min |
| 7: Hold |
4C |
PCR product was loaded onto a 2% agarose gel. Gene ruler DNA ladder mix (red) was used as marker.
VF2 - coding sequence (in pSB1C3) : 140bp
Coding sequence - VR (in pSB1C3) : 176bp
flhD/C : 932bp
VF2 - VR for pSB1C3 w. flhD/C :1248bp
VF2 - VR for pSB1C3 without insertion : 316bp
Results:
Gel electrophoresis:

Analysis:
All lanes except 4, 9 and 10 showed a band at app. 1200bp. which might indicate plasmids where flhD/C has been inserted.
Lane 4 and 9 has a band at app. 1500bp which could indicate undigested pSB1C3 (VF2 - VR for pSB1C3 w. J04450 is 1385bp).
Include column content here.
 "
"
