Objective
The main purpose of modeling was to characterize the topography of the kinetics behind foreign protein/product production in recombinant E.Coli biofilm with a diffusing inducer, and the effect on the various species involved by primarily the following factors:
- Time-derivative Spatial-Gradient Inducer Concentration
- Repressor Concentration
- Ribosome Binding Site (Rate of Transcription)
In terms of our iGEM project, this model was employed to explore the effect of IPTG concentration and diffusion, lacI concentration (determined by the combination part of constitutive promoter, ribosome binding site, lacI gene, double terminator, lac promoter/operon), and the ribosome binding site on the concentrations of all species involved - described in the following sections - and especially on Chitin Synthase and Chitin concentration and the corresponding rates.
Modeling
Overall Model
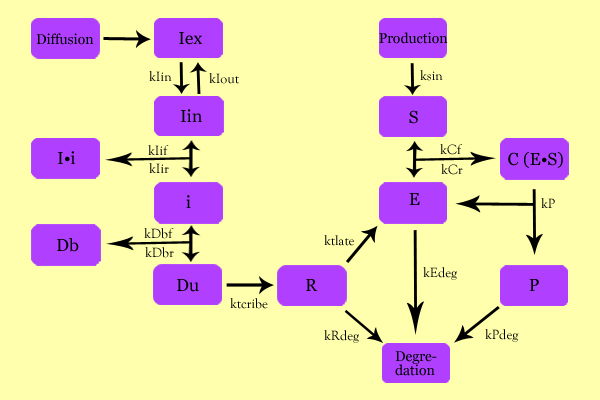
Using enzyme kinetics equations, we elected to mathematically simulate the following model:
Variables
- Iex: External Inducer, determined by diffusion through Fick's law (IPTG in our experiment)
- Iin: Internal Inducer (IPTG)
- Ii: Inducer bound to Repressor (IPTG bound to lacI)
- i: Repressor (lacI)
- Db: Repressor-bound DNA (lacI-bound DNA(CHS3) region in plasmid)
- Dunb: transcribe-able or Repressor-unbound DNA (lacI-unbound DNA(CHS3))
- Re: mRNA for Enzyme (CHS3 mRNA)
- E: Enzyme (CHS3)
- S: Substrate (N-Acetyl Glucosamine)
- C: Enzyme Substrate Complex (CHS3-(N-Acetyl-Glucosamine)-Chitin or (NAG)n Complex)
- P: Protein Product (Chitin or (NAG)n+1)
Equations
The differential of the variables were found as follows:
- dIin = kIin*Iex - kIex*Iin + kIir*Ii - kIif*Iin*i
- dIi = kIif*Iin*i - kIir*Ii
- di = kIir*Ii - kIif*Iin*i + kDbr*Db - kDbf*i*Dunb
- dDb = kDbf*i*Dunb - kDbr*Db
- dDunb = kDbr*Db - kDbf*i*Dunb
- dRe = ktscribe*Dunb - kRdeg*Re
- dE = ktslate*Re - kEdeg*E + kCr*CC + kP*CC - kCf*E*Si
- dSi = ksin*So - kCf*E*Si + kCr*CC
- dCC = kCf*E*Si - kCr*CC - kP*CC
- dP = kP*CC - kPdeg*P
IPTG Diffusion
First, Fick's Law of Diffusion was modeled through MATLAB. The diffusion constant used was 220um^2/s.[4]
IPTG was sprayed at the top of the colony, which then diffuses as according to Fick's law.
The spatially different local IPTG concentration will then differentially induce downstream processes.
This distinction was necessary in our project in order to establish a Chitin layer on the top of the biofilm.
Semi-Empirical Variable/Constant Determination
Status: Under Development
The initial plan was to use lacI-constitutive expression / lac-operon (CP-LacpI) part with Green Fluorescent Protein to acquire empirical data.
By testing various combinations of CP/LacpI, RBS, and IPTG concentrations with GFP, one could acquire fluorescence data over time using a plate reader, and use that data to fit the current model, where instead of producing Chitin Synthase, the protein product would be GFP, the concentration of which could be measured as fluorescence.
However, at the time of the wiki-freeze, data acquisition is incomplete.
Fitted Model
Matlab was used to generate a theoretical model where IPTG would diffuse down the biofilm as according to Fick's Law of Diffusion and initiate the process. The extracellular substrate concentration was assumed to be much greater than the uptake/use, and so would diffuse in at a constant rate.
This model was fitted with empirical data using cp-lacpi-gfp to estimate the rate constant, and therefore the effects of varying cp, lacpi, and rbs on enzyme and final product production.
References
1. A novel structured kinetic modeling approach for the analysis of plasmid instability in recombinant bacterial cultures
William E. Bentley, Dhinakar S. Kompala
Article first published online: 18 FEB 2004
DOI: 10.1002/bit.260330108
http://onlinelibrary.wiley.com/doi/10.1002/bit.260330108/pdf
2. Mathematical modeling of induced foreign protein production by recombinant bacteria
Jongdae Lee, W. Fred Ramirez
Article first published online: 19 FEB 2004
DOI: 10.1002/bit.260390608
http://onlinelibrary.wiley.com/doi/10.1002/bit.260390608/pdf
3. Pool Levels of UDP N-Acetylglucosamine and UDP NAcetylglucosamine-Enolpyruvate in Escherichia coli and Correlation with Peptidoglycan Synthesis
DOMINIQUE MENGIN-LECREULX, BERNARD FLOURET, AND JEAN VAN HEIJENOORT*
E.R. 245 du C.N.R.S., Institut de Biochimie, Universit' Paris-Sud, Orsay, 91405, France
Received 9 February 1983/Accepted 15 March 1983
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC217602/pdf/jbacter00247-0262.pdf
4. Diffusion in Biofilms
Philip S. Stewart
Center for Biofilm Engineering and Department of Chemical Engineering, Montana
State University–Bozeman, Bozeman, Montana, 59717-3980
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC148055/pdf/0965.pdf
5. Regulation of the Synthesis of the Lactose Repressor
PATRICIA L. EDELMANN' AND GORDON EDLIN
Department of Genetics, University of California, Davis, California 95616
Received for publication 21 March 1974
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC245824/pdf/jbacter00335-0105.pdf
MATLAB file provided upon request.
|  "
"