Team:TU Munich/Parts
From 2010.igem.org
(→Malachitegreen-Binding Aptamer - BBa_K494000) |
(→References) |
||
| Line 69: | Line 69: | ||
<html><a name="ref2"></a></html>[2] Babendure, J.R., S.R. Adams, and R.Y. Tsien, Aptamers switch on fluorescence of triphenylmethane dyes. J. Am. Chem. Soc, 2003. 125(48): p. 14716-14717. | <html><a name="ref2"></a></html>[2] Babendure, J.R., S.R. Adams, and R.Y. Tsien, Aptamers switch on fluorescence of triphenylmethane dyes. J. Am. Chem. Soc, 2003. 125(48): p. 14716-14717. | ||
| - | <html><a name="ref3"></a></html>[3] | + | <html><a name="ref3"></a></html>[3] Weigand, J.E. and B. Suess, Aptamers and riboswitches: perspectives in biotechnology. Applied microbiology and biotechnology, 2009. 85(2): p. 229-236. |
<html><a name="ref4"></a></html>[4] Stojanovic, M.N. and D.M. Kolpashchikov, Modular aptameric sensors. J. Am. Chem. Soc, 2004. 126(30): p. 9266-9270. | <html><a name="ref4"></a></html>[4] Stojanovic, M.N. and D.M. Kolpashchikov, Modular aptameric sensors. J. Am. Chem. Soc, 2004. 126(30): p. 9266-9270. | ||
Revision as of 22:17, 25 October 2010
|
||||||||
|
|
Submitted PartsSingle PartsMalachite Green Binding Aptamer - BBa_K494000
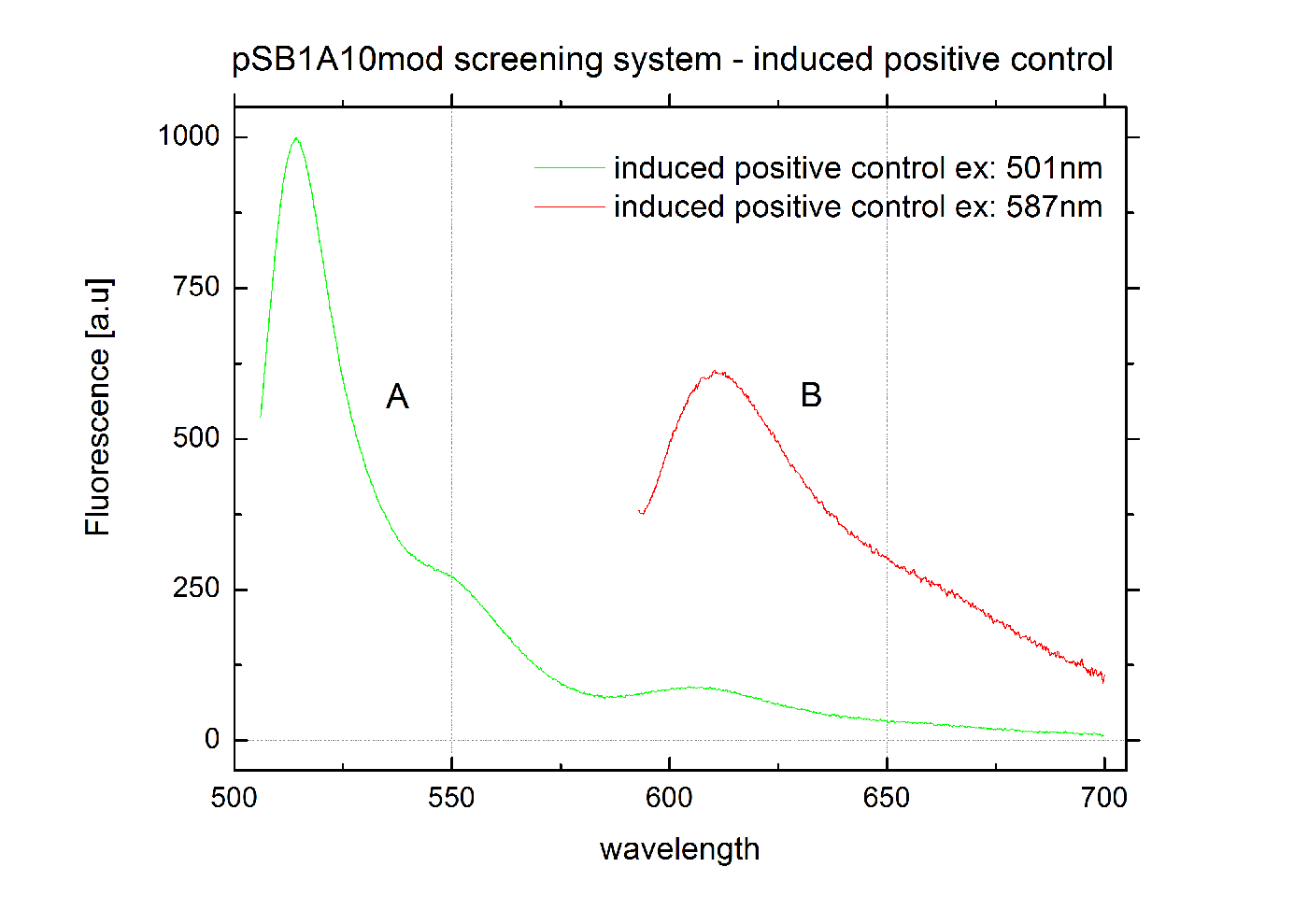
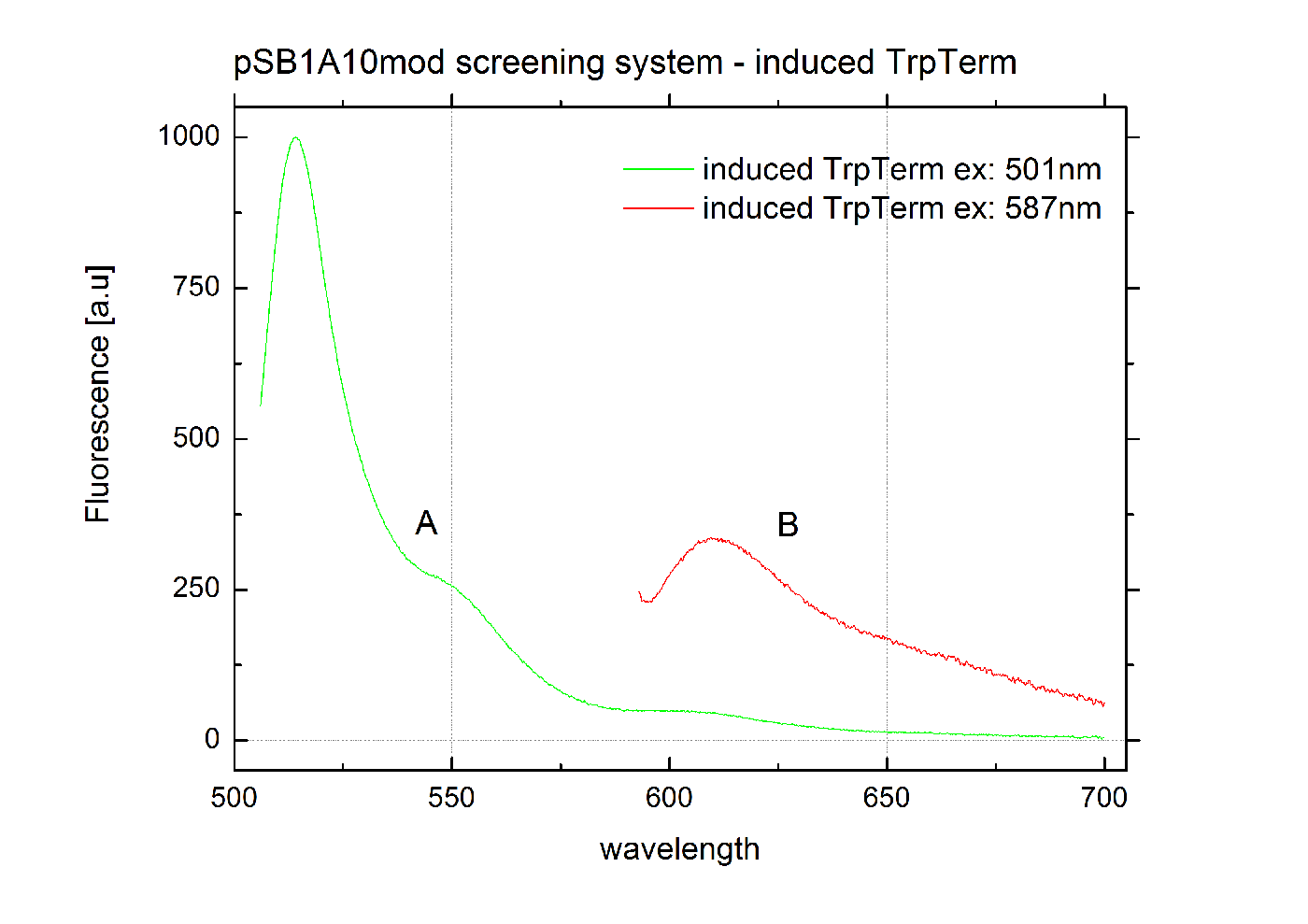
The malachite green binding aptamer has been successfully used in screening systems being both robust and easy to produce. Aptamers provide specifities in the range of antibodies and can be evolved to target small molecules and proteins.[3] PlasmidsIn general we want to provide a new principle of gene regulation which can be further developed, tested and optimizted by everybody. Therefore we focus on providing the parts needed for verification and testing of new individual switches. We provide a plasmid which can be used for further cloning, a positive control to test the general functionality and the constructs we characterized for comparison. Screening system: Backbone BBa_K494001We designed a new screening systems based on the non-functional pSB1A10 plasmid. We improved its features for ‘’in vivo’’ characterization of PoPS-based devices using fluorescent reporters . The plasmid still contains the Pbad arabinose-inducible induction system as a tunable input and eGFP as an internal standard for induction. However, we altered the reporter protein to mCherry. Furthermore we adjusted the BioBrick cloning site to allow cloning of additional parts independent from the Input/Output measurement. This screening plasmid is designed to be used with a second Arabinose inducible promoter <partinfo>I13453</partinfo> which is not included in this part. The improved screening system is optimized for the evaluation of PoPS-based devices in fluorescence measurements. RFP which was known to contain an RNase restriction site was replaced by mCherry which combines good expression yield, short maturation times and an acceptable and well-characterized quantum yield. A unique challenge for the characterization of our switches is the expression of a corresponding signal independent of the input/output measurement. Thus, we moved the BioBrick cloning site resulting in the fluorescent reporter being inside the cloning site and giving the possibility to clone independent parts behind the reporter protein. To fully function our screening plasmid need the arabinose inducible promoter BBa_I13453 in front of the PoPS-based device to screen. Using a second arabinose inducible promoter, we were able to keep eGFP as an internal standard for the tunable input via the Pbad arabinose-inducible induction system. The two identical promoters ensure the same rate of induction for eGFP and the tested PoPS-based device. Thus, obtaining comparable screening results is easy. Unfortunately, this design implicates a minor disadvantage. Two cloning steps are needed to gain an functional construct for testing any PoPS-based device. CloseScreening System: Positive Control BBa_K494002Here we present a ready to use composite part consisting of our screening system pSB1A10mod <partinfo>BBa_K494001</partinfo>, the PBad Promotor <partinfo>BBa_I13453</partinfo> and the reporter protein mCherry <partinfo>BBa_J06702</partinfo> including RBS and double Terminator <partinfo>BBa_B0015</partinfo>. This part belongs to the screening system and is intended as positive control. Screening System: Negative Control BBa_K494003Here we present a ready to use composite part consisting of our screening system pSB1A10mod BBa_K494001, the PBad Promotor BBa_I13453, the His Terminator and the reporter protein mCherry BBa_J06702 including RBS and double Terminator BBa_B0015. This part belongs to the screening system and is intended as negative control. Screening System: Control BBa_K494004Here we present a ready to use composite part consisting of our screening system pSB1A10mod BBa_K494001, the PBad Promotor BBa_I13453 and the reporter protein mCherry BBa_J06702 including RBS and double Terminator BBa_B0015. This part belongs to the screening system and is intended as control for light termination. Screening System: BBa_K494005Screening System: BBa_K494006Verified PartsFalsified PartspSB1A10The Screening Plasmid pSB1A10 is intended for the characterization of an Input/Output for a PoPS based device. However, our experiments designed to test our switching devices revealed the part to be non-functional. Although induction with arabinose worked fine indicated by GFP fluorescence, we could not detect any RFP output. Even by cloning a nonsense sequence into the BioBrick cloning site, we were unable to measure any RFP fluorescence [Data not shown]. We first assumed degradation of RFP-mRNA which is known to have an RNase site might be responsible. But comparison with our new screening system reveals a transcriptional problem to be most likely as replacing RFP did not improve the construct. Though, introducing a second arabinose promoter finally resulted in the desired output signal. In order to create a functional screening device for measurements we suggest cloning an additional PBad promoter BBa_I13453 in front of the part to be tested. Thus, eGFP is still able to function as an internal standard for expression rate via the Pbad arabinose-inducible induction. Therefore comparable results for PoPS-based devices are achievable. CloseReferences[1] Grate, D. and C. Wilson, Laser-mediated, site-specific inactivation of RNA transcripts. Proceedings of the National Academy of Sciences of the United States of America, 1999. 96(11): p. 6131. [2] Babendure, J.R., S.R. Adams, and R.Y. Tsien, Aptamers switch on fluorescence of triphenylmethane dyes. J. Am. Chem. Soc, 2003. 125(48): p. 14716-14717. [3] Weigand, J.E. and B. Suess, Aptamers and riboswitches: perspectives in biotechnology. Applied microbiology and biotechnology, 2009. 85(2): p. 229-236. [4] Stojanovic, M.N. and D.M. Kolpashchikov, Modular aptameric sensors. J. Am. Chem. Soc, 2004. 126(30): p. 9266-9270. [5] Smolke and so on.... [6] http://en.wikipedia.org/wiki/Logic_gate#Symbols |
|||||||
 "
"