Team:Imperial College London/Lab Diaries/Vectors team
From 2010.igem.org
| Line 21: | Line 21: | ||
====Week 6==== | ====Week 6==== | ||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| + | <td style="background-color:#e7e7e7;height:100px;width:150px;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;height:100px;width:150px;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;height:100px;width:150px;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;height:100px;width:150px;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;height:100px;width:150px;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="background-color:#FFCC66;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Afternoon</b> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | <li></li> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <ul> | ||
| + | <li></li> | ||
| + | <li></li> | ||
| + | </ul> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | </html> | ||
| - | <td style="background-color:# | + | <html> |
| + | <table width="850px" border="0"> | ||
| + | <tr> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| + | <b>Day</b> | ||
| + | </td> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> | ||
| + | </td> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> | ||
| + | </td> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> | ||
| + | </td> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> | ||
| + | </td> | ||
| + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> | ||
| + | </td> | ||
| + | |||
| + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Restriction digest of 5' dif XP with XbaI and PstI </li> | <li>Restriction digest of 5' dif XP with XbaI and PstI </li> | ||
| Line 60: | Line 141: | ||
</tr> | </tr> | ||
| - | + | <td style="background-color:#FFCC66;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Afternoon</b> | |
| - | + | ||
| - | <td style="background-color:# | + | |
</td> | </td> | ||
| + | <td style="background-color:#e7e7e7;text-align:top;font-family: helvetica, arial, sans-serif;color:#555555;font-family: helvetica, arial, sans-serif;color:#555555;"> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<b>Start assembly of PyrD vector</b> | <b>Start assembly of PyrD vector</b> | ||
| Line 81: | Line 161: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of resultant products from 5' dif XP digest</li> | <li>Gel analysis of resultant products from 5' dif XP digest</li> | ||
| Line 109: | Line 189: | ||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Restriction digestion of 5' ins [K143008] (using Eco and Spe)</li> | <li>Restriction digestion of 5' ins [K143008] (using Eco and Spe)</li> | ||
| Line 136: | Line 216: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis and extraction of 5' ins</li> | <li>Gel analysis and extraction of 5' ins</li> | ||
| Line 149: | Line 229: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Replica plating and colony PCR of transformed colonies (containing 5' dif in pSB1C3)</li> | <li>Replica plating and colony PCR of transformed colonies (containing 5' dif in pSB1C3)</li> | ||
| Line 157: | Line 237: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformation of overnight ligations: 5' ins, dif with pSB1C3 and pveg, SpecR-T and pSB1C3 </li> | <li>Transformation of overnight ligations: 5' ins, dif with pSB1C3 and pveg, SpecR-T and pSB1C3 </li> | ||
| Line 164: | Line 244: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Replica plating and colony PCR of: 5' ins, dif with pSB1C3 and pveg, SpecR-T and pSB1C3 </li> | <li>Replica plating and colony PCR of: 5' ins, dif with pSB1C3 and pveg, SpecR-T and pSB1C3 </li> | ||
| Line 173: | Line 253: | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>PCR purification of PSB1C3 vector </li> | <li>PCR purification of PSB1C3 vector </li> | ||
| Line 182: | Line 262: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Ligation of 5' ins and dif with pSB1C3 - a bench ligation (1 hour) and an overnight ligation were set up </li> | <li>Ligation of 5' ins and dif with pSB1C3 - a bench ligation (1 hour) and an overnight ligation were set up </li> | ||
| Line 191: | Line 271: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Overnight ligation of pveg and SpecR-T with pSB1C3 </li> | <li>Overnight ligation of pveg and SpecR-T with pSB1C3 </li> | ||
| Line 197: | Line 277: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of colony PCR products from the transformations</li> | <li>Gel analysis of colony PCR products from the transformations</li> | ||
| Line 266: | Line 346: | ||
====Week 8==== | ====Week 8==== | ||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
| - | + | ||
| - | + | ||
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
| - | + | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up overnight ligations for standard assembly (BBA) and 3A cloning (3A) of dif P</li> | <li>Set up overnight ligations for standard assembly (BBA) and 3A cloning (3A) of dif P</li> | ||
| Line 298: | Line 375: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Restriction digestion of 3' ins in the AK3 vector ( Using Xba and Pst) for 3A | <li>Restriction digestion of 3' ins in the AK3 vector ( Using Xba and Pst) for 3A | ||
| Line 305: | Line 382: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<ul> | <ul> | ||
| Line 313: | Line 390: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Replica plating of transformed colonies having dif P with 3' ins in AK3 -45 sigle colonies were plated</li> | <li>Replica plating of transformed colonies having dif P with 3' ins in AK3 -45 sigle colonies were plated</li> | ||
| Line 320: | Line 397: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of colony PCR from yesterday (first 15 replica plated colonies</li> | <li>Gel analysis of colony PCR from yesterday (first 15 replica plated colonies</li> | ||
| Line 327: | Line 404: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Miniprep of 4 overnight cultures - diff P with 3' ins in AK3; colonies 4,5,7 & 9</li> | <li>Miniprep of 4 overnight cultures - diff P with 3' ins in AK3; colonies 4,5,7 & 9</li> | ||
| Line 335: | Line 412: | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of PCR purified 3' ins in AK3 and gel purified 3' diff P to work out ratios for the liagation </li> | <li>Gel analysis of PCR purified 3' ins in AK3 and gel purified 3' diff P to work out ratios for the liagation </li> | ||
| Line 348: | Line 425: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformation of E.Coli with ligates from yesterday in AmpR;dif P with 3' ins in AK3 (insert and vector) and AK3 containing 3' ins vector only</li> | <li>Transformation of E.Coli with ligates from yesterday in AmpR;dif P with 3' ins in AK3 (insert and vector) and AK3 containing 3' ins vector only</li> | ||
| Line 358: | Line 435: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of gel extracted dif P and 3' ins (inserts) and pSB1C3 (vector) to work out ratios for the ligation </li> | <li>Gel analysis of gel extracted dif P and 3' ins (inserts) and pSB1C3 (vector) to work out ratios for the ligation </li> | ||
| Line 368: | Line 445: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up overnight 5 ml cultures containing dif P with 3' ins in AK3 for miniprep tomorrow - 4 cultures were set up by looking at the gel this morning; 2 positive looking (4 & 7), 1 negative (5) and one containing nothing (9</li> | <li>Set up overnight 5 ml cultures containing dif P with 3' ins in AK3 for miniprep tomorrow - 4 cultures were set up by looking at the gel this morning; 2 positive looking (4 & 7), 1 negative (5) and one containing nothing (9</li> | ||
| Line 374: | Line 451: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Diagnostic digests of minipreps containing dif P with 3' ins in AK3 - Two digests : One with Spe & Pst and other with Xba & Spe </li> | <li>Diagnostic digests of minipreps containing dif P with 3' ins in AK3 - Two digests : One with Spe & Pst and other with Xba & Spe </li> | ||
| Line 385: | Line 462: | ||
====Week 9==== | ====Week 9==== | ||
| - | |||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| Line 442: | Line 518: | ||
</tr> | </tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up 200 ml overnight culture of colony 4 (containing Dif P and K09) for midiprep tomorrow </li> | <li>Set up 200 ml overnight culture of colony 4 (containing Dif P and K09) for midiprep tomorrow </li> | ||
| Line 451: | Line 527: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Digests set up for Vector (SpecR) with Spe & Pst and Insert (dif P & K09)with Xba & Ps</li> | <li>Digests set up for Vector (SpecR) with Spe & Pst and Insert (dif P & K09)with Xba & Ps</li> | ||
| Line 460: | Line 536: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Dephosphorylation of vector </li> | <li>Dephosphorylation of vector </li> | ||
| Line 467: | Line 543: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Both ligations (Insert & Vector and Vector only) were plated in CmR and incubated overnight </li> | <li>Both ligations (Insert & Vector and Vector only) were plated in CmR and incubated overnight </li> | ||
| Line 473: | Line 549: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of colony PCR products </li> | <li>Gel analysis of colony PCR products </li> | ||
| Line 485: | Line 561: | ||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Colony PCR of 10 transformed C-Spec colonies</li> | <li>Colony PCR of 10 transformed C-Spec colonies</li> | ||
| Line 512: | Line 588: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Miniprep of C-Spec colonies 2, 5 & 9 in SpecR and CmR</li> | <li>Miniprep of C-Spec colonies 2, 5 & 9 in SpecR and CmR</li> | ||
| Line 520: | Line 596: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<ul> | <ul> | ||
| Line 529: | Line 605: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Start midiprep of C-Spec from colony 2 (isopropanol added elute was refrigerated for 4 hrs)</li> | <li>Start midiprep of C-Spec from colony 2 (isopropanol added elute was refrigerated for 4 hrs)</li> | ||
| Line 536: | Line 612: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Dilution of N-Spec and C-Spec (4x)</li> | <li>Dilution of N-Spec and C-Spec (4x)</li> | ||
| Line 548: | Line 624: | ||
</tr> | </tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis of colony PCR</li> | <li>Gel analysis of colony PCR</li> | ||
| Line 559: | Line 635: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Start midiprep of N-Spec (isopropanol added elute refrigerated overnight)</li> | <li>Start midiprep of N-Spec (isopropanol added elute refrigerated overnight)</li> | ||
| Line 567: | Line 643: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Continue midiprep of N-Spec (by ethanol preciptation)</li> | <li>Continue midiprep of N-Spec (by ethanol preciptation)</li> | ||
| Line 574: | Line 650: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Continue midiprep of C-Spec (by ethanol precipitation)</li> | <li>Continue midiprep of C-Spec (by ethanol precipitation)</li> | ||
| Line 583: | Line 659: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Single digest of diluted C-Spec (Eco only)</li> | <li>Single digest of diluted C-Spec (Eco only)</li> | ||
| Line 597: | Line 673: | ||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up digests of N-Spec and C-Spec (Eco and KpnI)- (CD 1)</li> | <li>Set up digests of N-Spec and C-Spec (Eco and KpnI)- (CD 1)</li> | ||
| Line 624: | Line 700: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel purification of N and C Specs from CD 2</li> | <li>Gel purification of N and C Specs from CD 2</li> | ||
| Line 631: | Line 707: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<ul> | <ul> | ||
| Line 639: | Line 715: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Backbone PCR of pSB1C3 using Barns buffer</li> | <li>Backbone PCR of pSB1C3 using Barns buffer</li> | ||
| Line 646: | Line 722: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel purification of pSB1C3</li> | <li>Gel purification of pSB1C3</li> | ||
| Line 655: | Line 731: | ||
</tr> | </tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up digests of N-Spec and C-Spec again, however, with a higher dilution of N-Spec - (CD 2)</li> | <li>Set up digests of N-Spec and C-Spec again, however, with a higher dilution of N-Spec - (CD 2)</li> | ||
| Line 666: | Line 742: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Dephosphorylate vector (C-Spec) from CD 2</li> | <li>Dephosphorylate vector (C-Spec) from CD 2</li> | ||
| Line 674: | Line 750: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformation of E.Coli with the two Spec final ligates - C-Spec (vector only) and N and C Specs (vector and insert)</li> | <li>Transformation of E.Coli with the two Spec final ligates - C-Spec (vector only) and N and C Specs (vector and insert)</li> | ||
| Line 681: | Line 757: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Gel analysis and extraction of pSB1C3</li> | <li>Gel analysis and extraction of pSB1C3</li> | ||
| Line 687: | Line 763: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Test digest of final spec minipreps with Eco and Kpn1 and Eco and Spe</li> | <li>Test digest of final spec minipreps with Eco and Kpn1 and Eco and Spe</li> | ||
| Line 699: | Line 775: | ||
====Week 12==== | ====Week 12==== | ||
| - | |||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
| - | |||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| Line 729: | Line 803: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Start midiprep of C-Spec</li> | <li>Start midiprep of C-Spec</li> | ||
| Line 736: | Line 810: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<li>Start midiprep of C-Spec colonies again</li> | <li>Start midiprep of C-Spec colonies again</li> | ||
<ul> | <ul> | ||
| Line 743: | Line 817: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Cloning digests of C-Spec with Kpn1 and Pst and N-Spec with Eco and Kpn1</li> | <li>Cloning digests of C-Spec with Kpn1 and Pst and N-Spec with Eco and Kpn1</li> | ||
| Line 750: | Line 824: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
</ul> | </ul> | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
</ul> | </ul> | ||
| Line 762: | Line 836: | ||
</tr> | </tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Miniprep cultures of C-Spec have not grown well enough therefore they will be left overnight</li> | <li>Miniprep cultures of C-Spec have not grown well enough therefore they will be left overnight</li> | ||
| Line 772: | Line 846: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Test digest C-Spec mini with ECo and KpnI</li> | <li>Test digest C-Spec mini with ECo and KpnI</li> | ||
| Line 782: | Line 856: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Continue Midiprep of C-Spec, pellets obtained</li> | <li>Continue Midiprep of C-Spec, pellets obtained</li> | ||
| Line 789: | Line 863: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Ligation gel with inserts; N-Spec (E & Kpn1) and C-Spec (Kpn1 & P) and Vector; pSB1C3 (E & P)</li> | <li>Ligation gel with inserts; N-Spec (E & Kpn1) and C-Spec (Kpn1 & P) and Vector; pSB1C3 (E & P)</li> | ||
| Line 796: | Line 870: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformation of E.Coli with the two Spec final ligates - C3 (vector only) and N and C Specs in C3 (vector and insert)</li> | <li>Transformation of E.Coli with the two Spec final ligates - C3 (vector only) and N and C Specs in C3 (vector and insert)</li> | ||
| Line 802: | Line 876: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformations failed, re-try next week!</li> | <li>Transformations failed, re-try next week!</li> | ||
| Line 812: | Line 886: | ||
====Week 13==== | ====Week 13==== | ||
| - | |||
<html> | <html> | ||
| - | <table width=" | + | <table width="850px" border="0"> |
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"> |
| - | <b> | + | <b>Day</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Monday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Tuesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Wednesday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Thursday</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFFF99;text-align:center; font-family: helvetica, arial, sans-serif;color:#555555;"><b>Friday</b> |
</td> | </td> | ||
| - | |||
</tr> | </tr> | ||
<tr> | <tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:center;font-family: helvetica, arial, sans-serif;color:#555555;"><b>Morning</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Set up 5 ml culture for miniprep of C-Spec of colnies 1 and 2 from synthesis in AmpR</li> | <li>Set up 5 ml culture for miniprep of C-Spec of colnies 1 and 2 from synthesis in AmpR</li> | ||
| Line 842: | Line 914: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Start midiprep of C-Spec</li> | <li>Start midiprep of C-Spec</li> | ||
| Line 849: | Line 921: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<li>Start midiprep of C-Spec colonies again</li> | <li>Start midiprep of C-Spec colonies again</li> | ||
<ul> | <ul> | ||
| Line 856: | Line 928: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Cloning digests of C-Spec with Kpn1 and Pst and N-Spec with Eco and Kpn1</li> | <li>Cloning digests of C-Spec with Kpn1 and Pst and N-Spec with Eco and Kpn1</li> | ||
| Line 863: | Line 935: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
</ul> | </ul> | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
</ul> | </ul> | ||
| Line 875: | Line 947: | ||
</tr> | </tr> | ||
| - | <td style="background-color:# | + | <td style="background-color:#FFCC66;width:100px;text-align:top;"><b>Afternoon</b> |
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Miniprep cultures of C-Spec have not grown well enough therefore they will be left overnight</li> | <li>Miniprep cultures of C-Spec have not grown well enough therefore they will be left overnight</li> | ||
| Line 885: | Line 957: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Test digest C-Spec mini with ECo and KpnI</li> | <li>Test digest C-Spec mini with ECo and KpnI</li> | ||
| Line 895: | Line 967: | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Continue Midiprep of C-Spec, pellets obtained</li> | <li>Continue Midiprep of C-Spec, pellets obtained</li> | ||
| Line 902: | Line 974: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Ligation gel with inserts; N-Spec (E & Kpn1) and C-Spec (Kpn1 & P) and Vector; pSB1C3 (E & P)</li> | <li>Ligation gel with inserts; N-Spec (E & Kpn1) and C-Spec (Kpn1 & P) and Vector; pSB1C3 (E & P)</li> | ||
| Line 909: | Line 981: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformation of E.Coli with the two Spec final ligates - C3 (vector only) and N and C Specs in C3 (vector and insert)</li> | <li>Transformation of E.Coli with the two Spec final ligates - C3 (vector only) and N and C Specs in C3 (vector and insert)</li> | ||
| Line 915: | Line 987: | ||
</td> | </td> | ||
| - | <td style="background-color:# | + | <td style="background-color:#e7e7e7;height:100px;width:200px;text-align:top;"> |
<ul> | <ul> | ||
<li>Transformations failed, re-try next week!</li> | <li>Transformations failed, re-try next week!</li> | ||
Revision as of 02:03, 25 October 2010
| Lab Diaries | Overview | Surface Protein Team | XylE Team | Vectors Team | Modelling Team |
| Here are the technical diaries for our project. We've split them up into three lab teams and the modelling team. We think it's really important that absolutely anyone can find out what we've been doing. For a really detailed look at what we did, and when, you've come to the right place! | |
| Objectives |
|
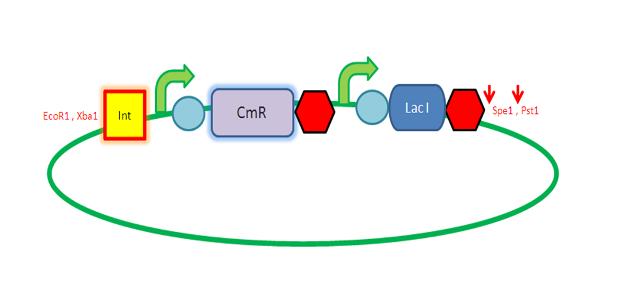
We are assembling the AmyE and PyrD vectors in order to transform B. Subtilis with our parts. Once completed, these vectors will be reusable and can then be used to introduce any relevant piece of DNA directly into B.subtilis genome. The vectors will be used both for the final assembly and for testing constructs. |
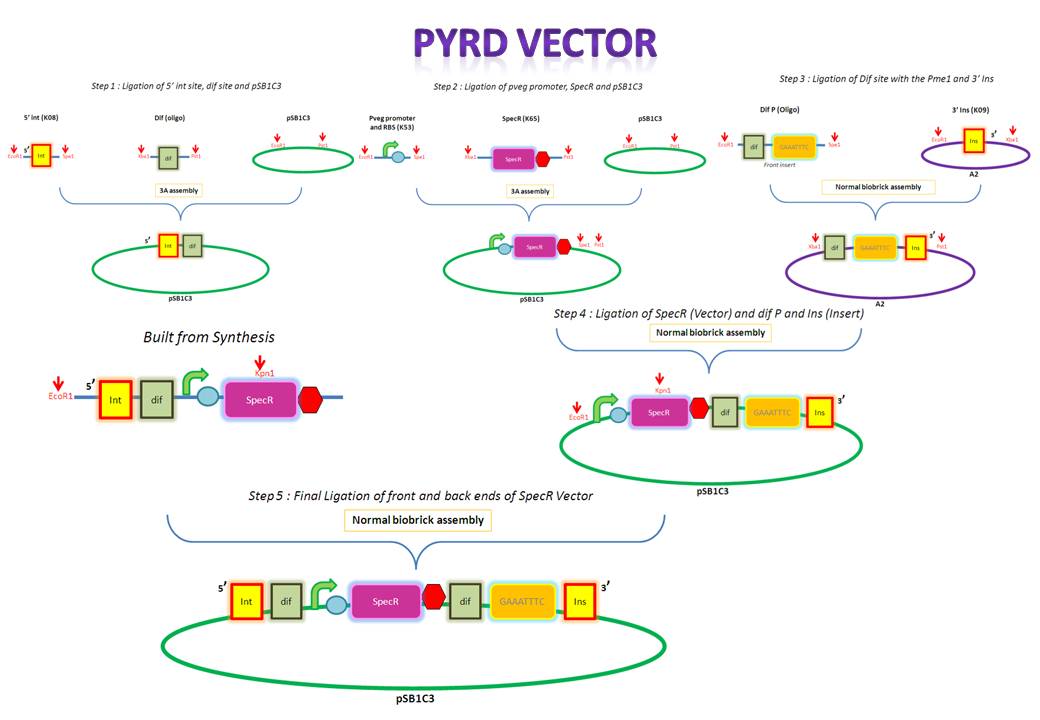
| PyrD Vector |
Week 6
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
|
|
|
|
| Afternoon |
|
|
|
|
|
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
Afternoon |
|
|
Thursday, August 12
- Annealing the forward and reverse strands of the dif XP oligo
The forward and reverse strands of the 5' dif site with XbaI and PstI restriction sites on either side have been synthesized separately. The synthesized fragments arrive in solid powder form. These were immediately diluted in ddH2O to obtain a stock concentration of 1 ng/ul. They were then allowed to anneal together by first heating them to 95 degs for denaturation and allowing them to cool down and anneal overnight.
Friday, August 13
- Restriction digest of dif XP
After annealing the two strands, the oligo was cut with XbaI and PstI to obtain overhangs that would later ligate with the compatible overhangs of a cut vector.
- PCR purification of the cut dif XP
The cut oligo was PCR purified in order to get rid of any contaminants. PCR purification gets rid of short pieces of DNA which are less than about 40 base pairs.
- Gel Analysis of dif XP
Week 7
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
|
|
|
|
| Afternoon |
|
|
|
|
Monday, August 16
- Restriction digest of 5' ins [K143008]
K143008 is the 5' integration site for the PyrD vector. This will be used as a front insert together with the dif XP for the pSB1C3 vector.
- PCR amplification of pSB1C3
The pSB1C3 vector backbone from the registry was amplified with the use of SB3 and SB2a primers. Submission of parts to the registry requires them to be in a pSB1C3 vector therefore any parts to be submitted will be inserted into this vector.
- PCR purification of pSB1C3
The PCR amplified vector was purified in order to get rid of any contaminants. For example, short pieces of DNA like the primers.
Tuesday, August 17
- Gel extraction and purification of 5' ins ES
The digested 5' ins was first analyzed on the gel to verify it's size and then extracted for purification. The 5' ins was gel purified in order to extract only the relevant piece of DNA.
- Restriction digest of pSB1C3
The pSB1C3 vector was digested so that it would contain compatible overhangs for ligation with inserts.
- PCR purification of pSB1C3 EP
The digested pSB1C3 was re-purified in order to get rid of any contaminant DNA that arose during the digestion.
- Restriction digests of pveg and Spec-T
pveg (promoter and RBS) and Spec-T (Spectinomycin with a terminator) were digested in preparation for 3A assembly.
- Ligation of 5' ins (ES) and dif (XP) with pSB1C3 (EP)
The digested 5' ins and dif (the front inserts) were ligated overnight with pSB1C3 (the vector). A bench ligation and an overnight ligation were set up.
- Transformation of E.Coli with bench ligate
E.Coli was transformed via the chemical method using the bench ligate.
- Gel extraction and purification of pveg and Spec-T
Since these are both inserts they were gel extracted and purified. PCR purification is not carried out for inserts since they are small and would therefore be lost during the process.
Wednesday, August 18
- Replica plating and colony PCR of 5' ins and dif in pSB1C3 (bench ligation)
- Ligation of pveg (ES) and SpecR-T (XP) with pSB1C3 (EP)
pveg and SpecR-T (the front inserts) were ligated overnight with pSB1C3 (the vector).
Thursday, August 19
- Transformation of E.Coli with the overnights ligates
- 5' ins and dif in pSB1C3
- pveg and SpecR-T in pSB1C3
Friday, August 20
- Replica plating and colony PCR of both transformations from yesterday.
- Annealing the forward and reverse strands of the dif P ES oligo
The forward and reverse strands of the 3' dif Pme1 sites with XbaI and PstI restriction sites on either side have been synthesized separately. The synthesized fragments arrive in solid powder form. These were immediately diluted in ddH2O to obtain a stock concentration of 1 ng/ul. They were then allowed to anneal together by heating them to 95 degs for denaturation and allowing them to cool down and anneal overnight.
Week 8
| Day | Monday | Tuesday | Wednesday | Thursday | Friday | |
| Morning |
|
|
|
|
|
|
| Afternoon |
|
|
|
|
|
|
Week 9
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
|
|
|
|
Afternoon |
|
|
|
|
|
Week 10
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
|
|
|
|
Afternoon |
|
|
|
|
|
Week 11
| Day | Monday | Tuesday | Wednesday | Thursday | Friday |
| Morning |
|
|
|
|
|
Afternoon |
|
|
|
|
|
Week 12
| Day | Monday | Tuesday | Wednesday | Thursday | Friday | |
| Morning |
|
|
|
|
|
|
Afternoon |
|
|
|
|
|
|
Week 13
| Day | Monday | Tuesday | Wednesday | Thursday | Friday | |
| Morning |
|
|
|
|
|
|
Afternoon |
|
|
|
|
|
|
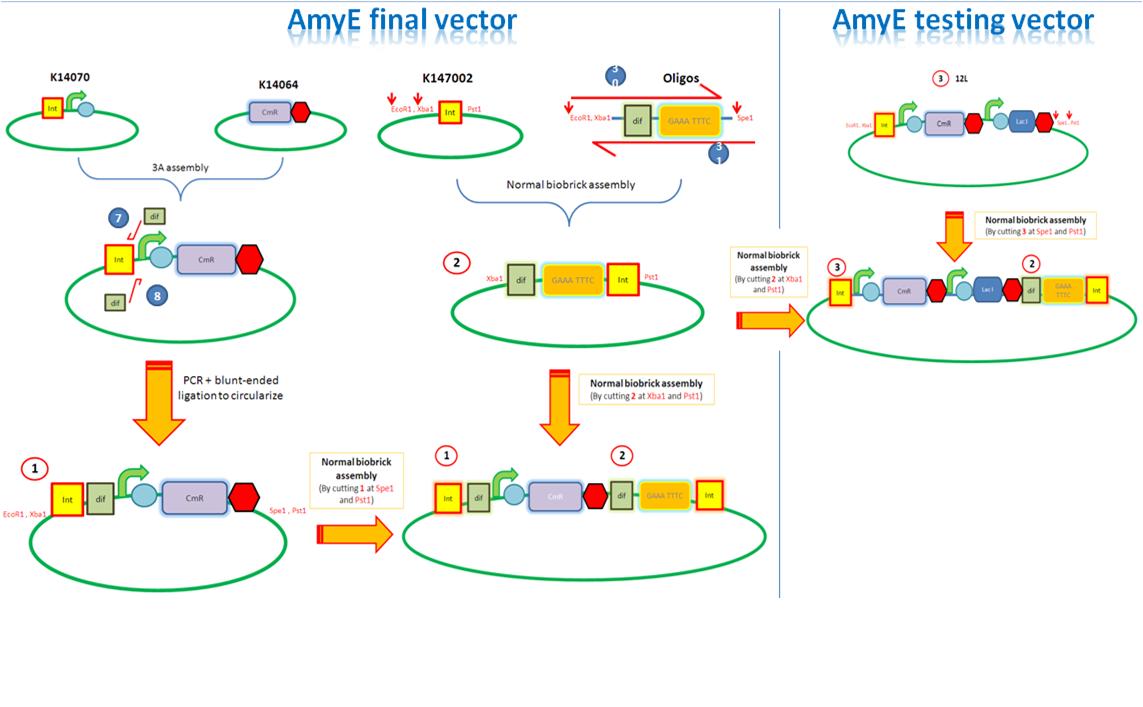
| AmyE Vector |
|
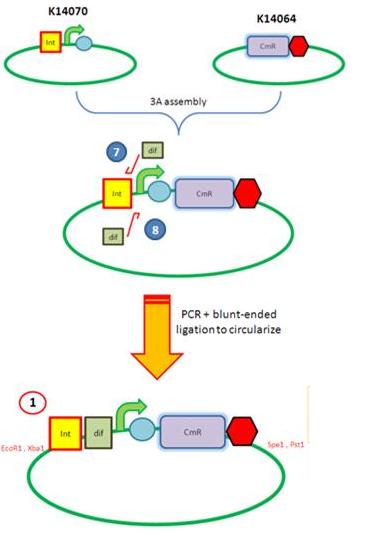
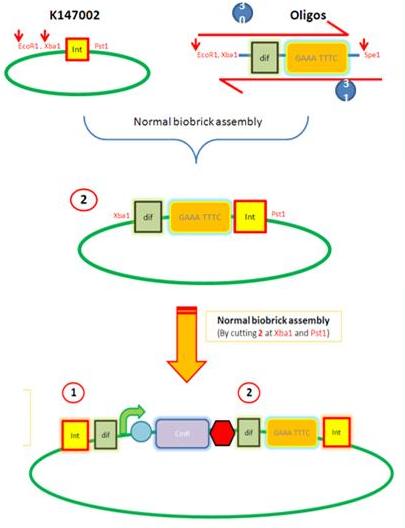
Starting from the top, we are assembling the first two fragments (K14070 and K14064) and K147002 with our oligos to add in a Dif site. Two dif sites on either side of resistance cassettes can be used to later excise antibiotic resistance from our final constructs.
Next Steps:
K14002 and oligos
Next Steps:
|
| Schedule & Lab Notes |
| Week 7 | ||||||||||||||||||
|
|
| Week 8 | |||||||||||||||||||||
|
|
| Week 9 | ||||||||||||||||||
|
|
| Week 10 |
|
|
| Week 11 |
|
|
| Week 12 |
|
|
| Week 13 | ||||||||||||||||||
|
|
 "
"