Team:Newcastle/16 July 2010
From 2010.igem.org
(→Results) |
PhilipHall (Talk | contribs) (→LacI BioBrick Construction) |
||
| Line 6: | Line 6: | ||
*To use PCR to extract ''lacI'' (promoter, ribosome-binding site (RBS) & coding sequence (CDS)) from plasmid pMutin4 and ligate into vector pSB1AT3 in front of red fluorescent protein (RFP). | *To use PCR to extract ''lacI'' (promoter, ribosome-binding site (RBS) & coding sequence (CDS)) from plasmid pMutin4 and ligate into vector pSB1AT3 in front of red fluorescent protein (RFP). | ||
| - | * | + | ==Materials== |
| + | *[[Team:Newcastle/15_July_2010|Overnight culture]]of transformants]] | ||
| + | *Restriction enzymes ''Eco''R1 and ''Pst''1 | ||
| - | == Protocol == | + | ==Protocol== |
| - | + | *Plasmid extraction of transformants | |
| - | + | *Single (''Eco''R1) and double (''Eco''R1 and ''Pst''1) restriction digest of plasmid extracts | |
| - | + | *Run digests on a gel | |
| - | + | ||
| - | + | ||
| - | + | ||
==Results== | ==Results== | ||
| - | |||
| - | |||
| - | |||
[[Image:Newcastle LacI BioBrick Construction Gel.png|700px|centre]] | [[Image:Newcastle LacI BioBrick Construction Gel.png|700px|centre]] | ||
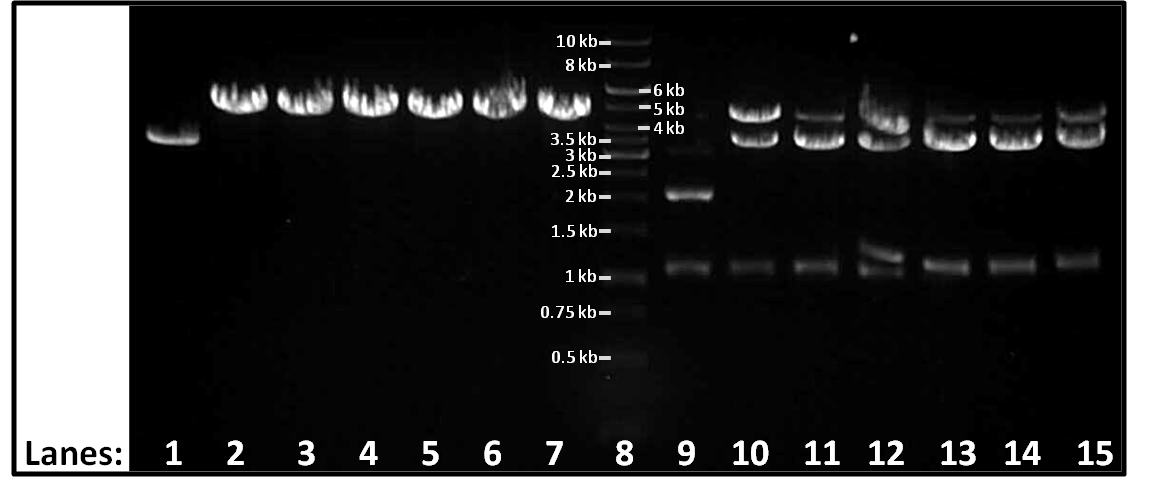
| + | ''Figure 1.'' Gel electrophoresis of the single and double digests using ''Eco''R1 and ''Pst''1. | ||
| - | + | ''Lane 1-7:'' Single digests of colonies 2-8 in order | |
| + | ''Lane 8:'' 1 Kbp Molecular weight marker | ||
| + | ''Lane 9-15:'' Double digests of colonies 2-8 in order | ||
| + | This gel shows that colony 2 contains only pSB1AT3 (3 Kbp) and that the rest contain pSB1AT3 (3 Kbp) with an RFP (1.5 Kbp)insert (total 4.5 Kbp). This is as was shown in the other tests that were performed this week. | ||
| - | + | ==Conclusion== | |
| + | We can conclude from these results that our ligation attempt has failed and that in order to carry on the construction we would need to re-perform the ligation using more of the PCR product. However this will not be done for the moment for these reasons: Firstly we believe that we can use another BioBrick already in the registry that will suit our needs, this is only made possible by the looming prospect of using Gibson cloning method to construct our Bricks; Secondly there are other Bricks that our more important for our project that we need to concentrate on. | ||
| - | + | {Team:Newcastle/footer}} | |
Revision as of 15:20, 10 August 2010

| |||||||||||||
| |||||||||||||
Contents |
LacI BioBrick Construction
Aims
- To use PCR to extract lacI (promoter, ribosome-binding site (RBS) & coding sequence (CDS)) from plasmid pMutin4 and ligate into vector pSB1AT3 in front of red fluorescent protein (RFP).
Materials
- Overnight cultureof transformants]]
- Restriction enzymes EcoR1 and Pst1
Protocol
- Plasmid extraction of transformants
- Single (EcoR1) and double (EcoR1 and Pst1) restriction digest of plasmid extracts
- Run digests on a gel
Results
Figure 1. Gel electrophoresis of the single and double digests using EcoR1 and Pst1.
Lane 1-7: Single digests of colonies 2-8 in order Lane 8: 1 Kbp Molecular weight marker Lane 9-15: Double digests of colonies 2-8 in order
This gel shows that colony 2 contains only pSB1AT3 (3 Kbp) and that the rest contain pSB1AT3 (3 Kbp) with an RFP (1.5 Kbp)insert (total 4.5 Kbp). This is as was shown in the other tests that were performed this week.
Conclusion
We can conclude from these results that our ligation attempt has failed and that in order to carry on the construction we would need to re-perform the ligation using more of the PCR product. However this will not be done for the moment for these reasons: Firstly we believe that we can use another BioBrick already in the registry that will suit our needs, this is only made possible by the looming prospect of using Gibson cloning method to construct our Bricks; Secondly there are other Bricks that our more important for our project that we need to concentrate on.
{Team:Newcastle/footer}} "
"