Team:TU Delft/26 July 2010 content
From 2010.igem.org
(New page: =Emulsification Assay= We have been looking for a robust protocol for the characterization of the emulsifiers for quite some time now. Somehow most literature refers to spectrophotometric ...) |
|||
| Line 4: | Line 4: | ||
The method is based on a simple article in Biochemical and Biophysical Methods: ''Spectrophotometric method for quantitative determination of nonionic, ionic and zwitterionic detergents'' by Rajakumari et al. 2006. | The method is based on a simple article in Biochemical and Biophysical Methods: ''Spectrophotometric method for quantitative determination of nonionic, ionic and zwitterionic detergents'' by Rajakumari et al. 2006. | ||
| - | [[Image:TU_Delft_Rajakumari.jpg|thumb|right|Calibration graph showing a linear increase in absorbance at 660 nm for increasing concentrations of SDS (● | + | [[Image:TU_Delft_Rajakumari.jpg|thumb|right|Calibration graph showing a linear increase in absorbance at 660 nm for increasing concentrations of SDS (●). Under the standard detergent assay conditions, various components like 200 mM NaCl (□), 2.0 mM CaCl2 (♦), 10% glycerol (■), 100 μg microsomal membranes (open diamond), 0.2 mM Triton X-100 (▲) and 2.5 mM CHAPS (○) were added and the turbidity was measured. Error bars represent the deviation from five independent experiments and each one performed in duplicate.]] |
The article shows the graph on the right, which we tried to reproduce. However, instead of triolein we used hexane. And the buffer used was 200 mM Tris-HCl, pH 8. | The article shows the graph on the right, which we tried to reproduce. However, instead of triolein we used hexane. And the buffer used was 200 mM Tris-HCl, pH 8. | ||
| + | |||
| + | {| | ||
| + | |'''#''' | ||
| + | |'''[SDS] (mM)''' | ||
| + | |'''Absorbance 660 nm (AU)''' | ||
| + | |- | ||
| + | |1 | ||
| + | |10 | ||
| + | |0.598 | ||
| + | |- | ||
| + | |2 | ||
| + | |8 | ||
| + | |0.425 | ||
| + | |- | ||
| + | |3 | ||
| + | |6 | ||
| + | |0.830 | ||
| + | |- | ||
| + | |4 | ||
| + | |4 | ||
| + | |0.432 | ||
| + | |- | ||
| + | |5 | ||
| + | |2 | ||
| + | |0.379 | ||
| + | |- | ||
| + | |6 | ||
| + | |0 | ||
| + | |0.001 | ||
| + | |} | ||
Revision as of 12:20, 27 July 2010
Emulsification Assay
We have been looking for a robust protocol for the characterization of the emulsifiers for quite some time now. Somehow most literature refers to spectrophotometric assays based on turbidity after mixing. This a somewhat arbitrary method, and so far we were unable to get reproducible results. Today we decided to explore this method once more, using a lot of different conditions.
The method is based on a simple article in Biochemical and Biophysical Methods: Spectrophotometric method for quantitative determination of nonionic, ionic and zwitterionic detergents by Rajakumari et al. 2006.
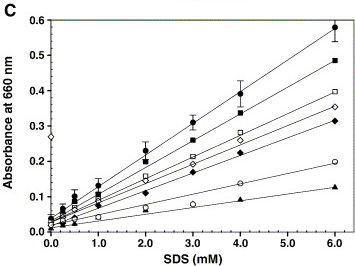
The article shows the graph on the right, which we tried to reproduce. However, instead of triolein we used hexane. And the buffer used was 200 mM Tris-HCl, pH 8.
| # | [SDS] (mM) | Absorbance 660 nm (AU) |
| 1 | 10 | 0.598 |
| 2 | 8 | 0.425 |
| 3 | 6 | 0.830 |
| 4 | 4 | 0.432 |
| 5 | 2 | 0.379 |
| 6 | 0 | 0.001 |
 "
"