Team:Calgary/Parts/Characterization
From 2010.igem.org
| Line 81: | Line 81: | ||
| - | |||
</html> | </html> | ||
Revision as of 03:24, 27 October 2010

Characterization
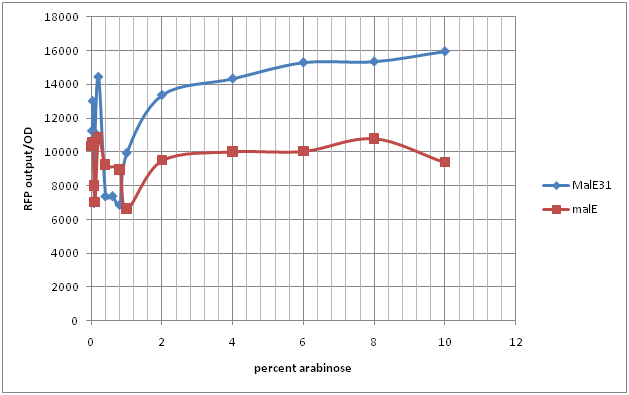
Experiment 1: Measuring RFP output by co-transformation of MalE and MalE31 coupled to arabinose promoter in the CpxR-RFP competent cells
Protocol:
Arabinose inducible promoter (I0500) coupled with standard ribosome binding site (B0034) and the respective maltose binding protein were transformed into competent cells containing pCpxR coupled with RFP generator (I13507). These cells were plated and incubated overnight. Colonies from each of the plates were selected and overnight cultures were prepared at 37 C. These 5 ml overnight cultures were then sub-cultured in 20 ml broth. These were shaken for 6-8 hours and aliquoted into 5 ml cultures and induced with varying levels of arabinose(percent). This was incubated in the shaker for 12-14 hours and RFP output was measured using 555 excitation and 632 nm emission frequency.
Discussion of Results and Conclusion
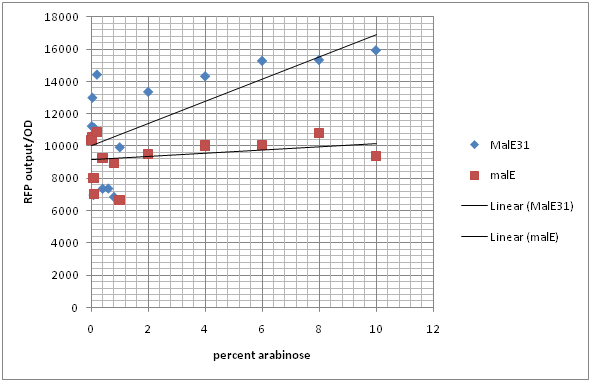
Figure 1 and 2 indicate the RFP output normalized with growth ratio (OD) at different levels of arabinose. Figure 1 shows that CpxR-I13507 is activated at the highest level when MalE31, the periplasmic misfolder, is expressed. This occurs around 0.2% arabinose concentration. Similar trends are observed in the case of MalE which is a periplasmic folder. MalE and MalE31 activate the system at different levels. MalE31 has similar trends to MalE but has a higher level of RFP expression. These results prove that MalE and MalE31 can both activate the CpxR system however, MalE31, which misfolds, activates it more rapidly and at a lower level of arabinose concentration compared to MalE. If the line of best fit is studied, it is seen that MalE has very minimal level of Cpx activation. Whereas, malE31 has a linear regression which flattens out as the system reaches its upper threshold of detection. Biologically, this could mean that the MalE31 is activated at levels that saturate the cellular chaperones and cause the system to reach its threshold level of proteolytic and chaperone activities. Another interesting pattern observed is the fact that when MalE is constructed with CpxR-I13507 on the same plasmid (Green), the cell RFP output is much lower compared to cells co-transfected with CpxR-I13507 and I0500-B0034 –MalE. This indicates that insertion of high copy plasmid also induces stress in the periplasmic region of the cell consequently inducing the activation of CpxR system.
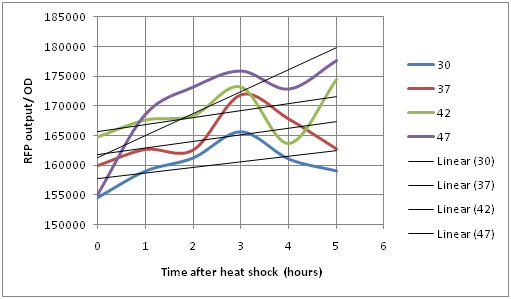
Experiment 2: Measuring RFP output of the CpxR-I13507 cells after exposure to different temperature for different time periods
Protocol:
Top 10 competent cells were transformed with CpxR-I13507 and plated. 5 ml overnight cultures were made from 5 different colonies using LB broth with appropriate antibiotics. Each of these cultures were aliquoted into six different tubes containing 600 µL of culture. These tubes were then placed in hot water baths at 30 C, 37C, 42C, 47C. Measurements were taken every hour for 5 hours after placing the tubes in different temperatures at 555 nm excitation and 632 nm emission.
Results
Discussion of Results and Conclusion
This graph shows that the CpxR system does respond to temperature activated stress. When the system is placed at 42 C the RFP output is much higher at t=0 compared to the system placed at 37 C or 30 C. This indicates that the system does get activated due to heat shock which matches the literature parameters. At 47 C, the system gets activated faster because the linear regression has a steeper slope. This indicates that the system is being stressed and it produces its downstream product which is RFP in this case and DegP and other chaperones in the genomic DNA much faster in order to cope with periplasmic protein denaturation. Also, it seems that the system gets activated dramatically after 3 hours regardless of the temperature, this could indicate that the system peaks after 3 hours and the genomic CpxR produces enough downstream chaperones and proteases in order for the system to be able to cope with stress which allows the RFP reading to decrease at 4 hours time because the cell reaches homeostasis. This allows the cell to get rid of misfolded protein and other factors that might be contributing to stressing it out and causing the Cpx regulon to be activated. The cell then shows a rapid rise again because it is still under heat shock stress. But, if the cell was placed at 37 degrees, the cell would show a flatline pattern rather than an oscillating pattern.
Experiment 4: Measuring the GFP output through insertion of the mutant malE and malE31 with the transport signal sequence deleted into competent cells containing a fusion promoter (ibpAB-fsxA) coupled to a GFP reporter.
Purpose
The purpose of this assay is to test the output that the cytoplasmic acting fusion promoter (ibpAB-fsxA) will produce with proteins that are known to fold correctly (malEΔSS) and with proteins that are known to misfold (malE31ΔSS) in the cytoplasm. The plasmids containing malEΔSS and malE31ΔSS are coupled to an IPTG inducible promoter and were received from the lab of Jean-Michel Betton.
 "
"