Team:Minnesota/Project
From 2010.igem.org
| Line 40: | Line 40: | ||
To a synthetic biologist, an empty proteinacious compartment by itself is not useful without a way of targeting favorite proteins to the inside. It had been demonstrated that signal sequences at the N-terminus of BMC associated enzymes are capable of causing their compartmentalization inside shells (Cheng et al.). To be able to target a reporter to the inside of our recombinant Eut BMCs, our group used a predicted signal sequence from the enzyme EutC, which is known to be sequestered inside the Eut BMC. EutC encodes the small subunit of the ethanolamine ammonia lyase enzyme that is known to be targeted to the Eut microcompartment. By comparison to homologous enzymes, Cheng et al. suggested that a sequence of 19 amino acids at the N-terminus of EutC may be a likely candidate for serving this function. To verify this idea, a construct containing this signal sequence fused to GFP was developed and co-transformed in E. coli cells alongside the Eut SMNLK vector. These dual-transformed cells were characterized by fluorescence microscopy and the results are summarized below. | To a synthetic biologist, an empty proteinacious compartment by itself is not useful without a way of targeting favorite proteins to the inside. It had been demonstrated that signal sequences at the N-terminus of BMC associated enzymes are capable of causing their compartmentalization inside shells (Cheng et al.). To be able to target a reporter to the inside of our recombinant Eut BMCs, our group used a predicted signal sequence from the enzyme EutC, which is known to be sequestered inside the Eut BMC. EutC encodes the small subunit of the ethanolamine ammonia lyase enzyme that is known to be targeted to the Eut microcompartment. By comparison to homologous enzymes, Cheng et al. suggested that a sequence of 19 amino acids at the N-terminus of EutC may be a likely candidate for serving this function. To verify this idea, a construct containing this signal sequence fused to GFP was developed and co-transformed in E. coli cells alongside the Eut SMNLK vector. These dual-transformed cells were characterized by fluorescence microscopy and the results are summarized below. | ||
| - | [[Image:Flourescence images 1.jpg| | + | [[Image:Flourescence images 1.jpg|200px|center|frame]] |
| - | [[Image:Flourescence images 2.jpg| | + | [[Image:Flourescence images 2.jpg|200px|center|frame]] |
| - | [[Image:Flourescence images 3.jpg| | + | [[Image:Flourescence images 3.jpg|200px|center|frame]] |
Flourescence imaging shows that cells transformed solely with GFP or GFP with the fused signal sequence cause the entirety of the cell cytoplasm to fluoresce. The cells transformed with untagged-GFP and the microcompartment proteins also have distributed fluorescence. As predicted, E. coli transformed with tagged GFP and all of the microcompartment genes (EutSMNLK)display localization of the fluorescence to a discrete point within the cell. These experiments have shown that the S. enterica microcompartment proteins can be expressed in E. coli and that expression of these proteins can cause GFP with an attached signal sequence to be localized to a distinct point inside the cell. | Flourescence imaging shows that cells transformed solely with GFP or GFP with the fused signal sequence cause the entirety of the cell cytoplasm to fluoresce. The cells transformed with untagged-GFP and the microcompartment proteins also have distributed fluorescence. As predicted, E. coli transformed with tagged GFP and all of the microcompartment genes (EutSMNLK)display localization of the fluorescence to a discrete point within the cell. These experiments have shown that the S. enterica microcompartment proteins can be expressed in E. coli and that expression of these proteins can cause GFP with an attached signal sequence to be localized to a distinct point inside the cell. | ||
Revision as of 18:37, 25 October 2010
| Home | Team | Project | Protocols | Notebook | Judging Criteria | Safety |
|---|
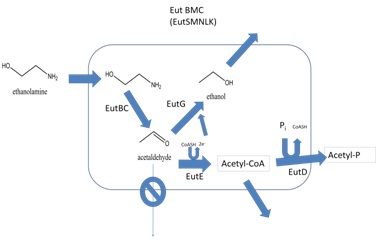
A bacterial microcompartment (BMC) is a polyhedral protein complex that encapsulates enzymes involved in specific metabolic pathways. BMCs were first observed by microscope in cyanobacteria the 1950’s. By the late 1990’s, it had become clear that these proteinaceous structures are produced by many types of bacteria, and serve various functions (yeates et al. 2008). There are multiple examples of BMC’s occurring in nature: the carboxysome found in cyanobacteria and chemoautotrophic bacteria, the propanediol utilization compartment found in enterobacteria, as well as the ethanolamine utilization (Eut) compartment used in this project, also found in enterobacteria. By sequestering enzymes and substrates together in an enclosed space within the cell, the overall reaction efficiency is greatly improved. In some species, BMCs localize toxic compounds with enzymes that convert them into benign compounds. In this way a bacterial cell can ensure that the problematic intermediate is not released into the cell cytoplasm. BMCs are ideal for iGEM because they could be harnessed as nanobioreactors for metabolic engineering. Engineering a strain to express a BMC shell is possible because the genes that encode the BMC associated proteins are frequently organized tightly together into an operon. For this reason, theses are fairly well characterized. Engineering a BMC to house an enzymatic pathway would increase the efficiency of the process, and would also quarantine any toxic intermediates (Fig 1).
Our goals were to develop E. coli that express a recombinant BMC, and develop a system for targeting proteins to be encapsulated within that compartment. We chose to use the Eut shell from S. enterica because this species has been shown to produce these structures and they have been well characterized (Penrod & Roth, 2006). The ethanolamine utilization operon from Salmonella enterica strain LT2 is composed of 17 genes (Fig 5). Of these, only five possess BMC associated domains and are believed to be components of the Eut microcompartment. In Salmonella enterica this organelle-like structure localizes the enzymes required for ethanolamine utilization and prevents acetaldehyde, a volatile intermediate, from diffusing into the cytoplasm.
The five proteins possessing BMC domains are the EutS, M, N, L, and K gene products. The first step in this project was to isolate these genes from genomic S. enterica DNA, specifically strain LT-2. Specific primers containing BglII and NotI sites were designed for the five genes, EutL and EutK were cloned as a single PCR fragment, as were EutM and EutN. EutS was cloned separately. Each PCR product was ligated into the biobrick-compatible pUCBB plasmid (Johnson et al., manuscript under preparation). The EutMN construct was digested with EcoRI and SpeI and gel purified to prepare an entry fragment. The EutLK construct was linearized with EcoRI and XbaI. A EutMNLK construct was obtained by ligating the EutMN fragment into the linearized pUCBB-EutLK plasmid. This EutMNLK plasmid was then linearized with EcoRI/ XbaI and a final ligation was performed with EcoRI/SpeI digested EutS as the entry fragment. For submission to the iGEM competition the full EutSMNLK fragment was ligated into the psb1C3 vector. All completed constructs were confirmed by DNA sequencing.
To demonstrate that these genes can be expressed in a new bacterial host, these plasmids were transformed into E. coli and analysis of the protein produced was performed by SDS-PAGE. The results as shown in fig(??#) indicate that E. coli are able to express the Eut Genes. The observed overexpressed-protein bands match the predicted sizes of the recombinant proteins. pUCBB-GFP total cell lysate was used as a negative control in this experiment. Gel pictures
To a synthetic biologist, an empty proteinacious compartment by itself is not useful without a way of targeting favorite proteins to the inside. It had been demonstrated that signal sequences at the N-terminus of BMC associated enzymes are capable of causing their compartmentalization inside shells (Cheng et al.). To be able to target a reporter to the inside of our recombinant Eut BMCs, our group used a predicted signal sequence from the enzyme EutC, which is known to be sequestered inside the Eut BMC. EutC encodes the small subunit of the ethanolamine ammonia lyase enzyme that is known to be targeted to the Eut microcompartment. By comparison to homologous enzymes, Cheng et al. suggested that a sequence of 19 amino acids at the N-terminus of EutC may be a likely candidate for serving this function. To verify this idea, a construct containing this signal sequence fused to GFP was developed and co-transformed in E. coli cells alongside the Eut SMNLK vector. These dual-transformed cells were characterized by fluorescence microscopy and the results are summarized below.
Flourescence imaging shows that cells transformed solely with GFP or GFP with the fused signal sequence cause the entirety of the cell cytoplasm to fluoresce. The cells transformed with untagged-GFP and the microcompartment proteins also have distributed fluorescence. As predicted, E. coli transformed with tagged GFP and all of the microcompartment genes (EutSMNLK)display localization of the fluorescence to a discrete point within the cell. These experiments have shown that the S. enterica microcompartment proteins can be expressed in E. coli and that expression of these proteins can cause GFP with an attached signal sequence to be localized to a distinct point inside the cell.
We wanted to determine which of Eut BMC genes is required for localization. Co-transformations of EutMN, EutLK, and EutMNLK with the tagged reporter do not demonstrate the localizing phenomena. On the other hand, co-transformation with EutS alone was sufficient for localization. This observation was surprising and indicates that EutS may be the specific protein that recognizes the EutC signal sequence and causing localization. This experiment has caused us to become interested in investigating whether EutS is able to form a proper BMC on its own. Future experiments will probe further into this possibility—expect to hear more about this in next year’s jamboree!
We are pleased to show that our fluorescent images yielded such definite results. For many of us this was our first exposure to cloning experiments. The work performed at the outset of this project went smoothly, but by the end of the summer we were facing issues and setbacks of all kinds. We are satisfied that our results have some apparent applications. An example application would be to tag enzymes with the EutC signal sequence and co-transform these with EutS. The enzymes would associate to a definite location in the cytoplasm and biocatalysis would be improved by bringing multiple steps of a pathway into proximity. We hope to see future iGEM teams develop imaginative new uses for our Biobrick submission.
To move forward with our project, our goal will be to demonstrate that these proteins are forming a structured microcompartment and not a cytoplasmic agglutination. Further imaging experiments will be needed. The next step that our group will take in exploring BMCs and their properties will make use of scanning and transmission electron microscopes to visualize whether they form the hypothesized icosahedral structure. Other future experiments will attempt to show that this system can localize enzymes to engineer pathways with improved efficiency as well as prevent the accumulation of toxic intermediate in the cytoplasm, allowing cells to perform reactions that would otherwise result in their death. Nanobioreactors!
References
Fan, C. G., S. Q. Cheng, et al. (2010). "Short N-terminal sequences package proteins into bacterial microcompartments." Proceedings of the National Academy of Sciences of the United States of America 107(16): 7509-7514.
Penrod, J. T. and J. R. Roth (2006). "Conserving a volatile metabolite: a role for carboxysome-like organelles in Salmonella enterica." Journal of Bacteriology 188(8): 2865-2874.
Yeates, T. O., C. A. Kerfeld, et al. (2008). "Protein-based organelles in bacteria: carboxysomes and related microcompartments." Nature Reviews Microbiology 6(9): 681-691.
 "
"