Team:Calgary/20 July 2010
From 2010.igem.org
H.dastidar (Talk | contribs) |
|||
| (15 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{CalgaryNotebookTemplate| | {{CalgaryNotebookTemplate| | ||
| - | + | Tuesday July 20, 2010| | |
| - | + | ||
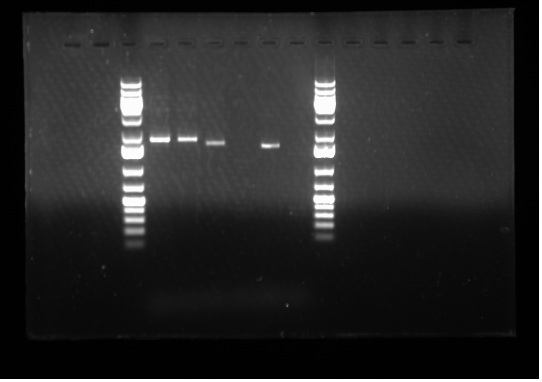
[[Image:07.20.2010. Raida PCR BBK primer.jpg|thumb|400px|Gel electrophoresis of Raida's PCR products: BBK primers that anneal before the multiple cloning sites. Lane 4 and 5 : Lux0047 A + B0015 psB1AK3 Lane 6: J23002 + Lux0047E, Lane7: R0040 + I3502, Lane 8: Lux0047E, Lane 9: Master Mix]] | [[Image:07.20.2010. Raida PCR BBK primer.jpg|thumb|400px|Gel electrophoresis of Raida's PCR products: BBK primers that anneal before the multiple cloning sites. Lane 4 and 5 : Lux0047 A + B0015 psB1AK3 Lane 6: J23002 + Lux0047E, Lane7: R0040 + I3502, Lane 8: Lux0047E, Lane 9: Master Mix]] | ||
| - | <u> Raida </u> | + | [[Image:07.20.2010 Chris CpxP Gel 2.jpg|thumb|400px|Agarose Gel Electrophoresis of Chris's CpxP colony PCR in plasmid form. The gel was allowed to run for one hour and it did not move from 25 minutes to 1 hour.]] |
| + | |||
| + | <u>Raida</u> | ||
Today I set up and ran two 1% gels: one for my PCR products from yesterday and the second one for Emily's PCR products- both of which were done to test whether our primers are working or not. Please see the gel to the side: | Today I set up and ran two 1% gels: one for my PCR products from yesterday and the second one for Emily's PCR products- both of which were done to test whether our primers are working or not. Please see the gel to the side: | ||
| - | As we can see in the gel electrophoresis image, the expected results has been obtained. For example, Lane 8 | + | As we can see in the gel electrophoresis image, the expected results has been obtained. For example, Lane 8 contains only the Lux0047E gene where as Lane 6 has J23002 + Lux0047E, hence the band in Lane 8 goes slightly further relative to Lane 6 because it is a shorter band. Lane 7 shows no band because the primers did not and was not supposed to anneal to the site because R0040 + I3502 was in a standard plasmid from this year and has a different sequence than all the 4 other plasmids that were used for this particular PCR. The primers are not complimentary to the sequence of the plasmids being used this year. So the fact that Lane 7 shows no band means there was no amplification- so the result is positive. Lanes 4 and 5 show exactly the same band because they are the same genes in the same plasmid backbone. Furthermore, Lane 9 shows no band because it is the Master Mix and our negative control. Therefore, it can be concluded that the '''primers are functional.''' |
| - | + | ||
| - | <u> Jeremy </u> | + | Apart from the Wet Lab, I've also helped Jeremey with the summary of our Lethbridge Workshop. |
| + | |||
| + | |||
| + | <u>Jeremy</u> | ||
Today I completed the ligation and transformation of the I0500+E0034 plasmid switch. I also ran the PCR products and the results came back negative. So I set up another PCR and Gel. | Today I completed the ligation and transformation of the I0500+E0034 plasmid switch. I also ran the PCR products and the results came back negative. So I set up another PCR and Gel. | ||
| - | }} | + | |
| + | <u>Patrick</u> | ||
| + | |||
| + | Today I helped plasmid prep the ''degP'' promoter and ''cpxR'' promoter on a GFP and RFP reporter device each. I also continued looking to literature regarding high and low-copy plasmids for the modelling project. | ||
| + | |||
| + | |||
| + | <u>Himika</u> | ||
| + | |||
| + | Today I plated the construction that I did last night. I also overnight cultured the construction from last night by inoculating from the cells to be plated. Tomorrow I will plasmid prep the cultures. I also looked into the literature regarding the plasmid which will be presented to Paul on Thursday. | ||
| + | |||
| + | |||
| + | <u>Chris</u> | ||
| + | |||
| + | Today, I did a colony PCR of the CpxP promoter that was transformed into both ampicillin-kanamycin and ampicillin-chloramphenicol plasmids. The results of the colony PCR were then run on a 1.2% gel and the bands were very irregular as shown on the right. The gel was run for approximately 1 hour and the bands did not move after 35 minutes. Another colony PCR was set up and let to run again overnight. As well, overnight culures were made of all 11 samples so they could be plasmid prepped appropriately if the Colony PCR showed up possible correct bands. As well, I helped Emily test the plate reader with several cultures that had arabinose inducible promoters induced overnight of GFP and RFP. | ||
| + | |||
| + | |||
| + | <u>Alex</u> | ||
| + | |||
| + | Today I did a plasmid prep of the overnight colonies of CpxR and DegP devices. I also made a gel and ran the colony PCR product. Three of the colonies appear to be the anticipated size, others appear to be failed constructions. | ||
| + | |||
| + | |||
| + | <u>Emily</u> | ||
| + | |||
| + | Today I retransformed the psB1AC3 and psB1AK3 vectors with the P1010 ccdb insert in them and I transformed them into d53.30 competant cells and plated on A,C and K plates. I also did a plasmid switch of B0034 from an Amp vector into AC and AK vectors. I antarctic phosphotased the vector and then ligated and transformed into TOP10 E coli cells. I then plated on A,C and K and left for overnight growth. I also spent a large portion of my day testing out the plate reader. I made serial dilutions of the ovenright cultures that I had made of a constitutive GFP circuit, a constitutive RFP circuit and B0015. I tested these all as well as a negative control of just LB broth and our 4 constructs of I0500-B0034-E0040 that have been induced with arabinose. Today I also filled more pipette tip boxes and autoclaved a bunch of necessary lab supplies like pipette tip boxes and 1.5 mL centrifuge tubes. | ||
| + | |||
| + | |||
| + | <u> Himika </u> | ||
| + | |||
| + | Today I did a colony PCR o}} | ||
Latest revision as of 22:38, 26 August 2010

Tuesday July 20, 2010
Raida
Today I set up and ran two 1% gels: one for my PCR products from yesterday and the second one for Emily's PCR products- both of which were done to test whether our primers are working or not. Please see the gel to the side: As we can see in the gel electrophoresis image, the expected results has been obtained. For example, Lane 8 contains only the Lux0047E gene where as Lane 6 has J23002 + Lux0047E, hence the band in Lane 8 goes slightly further relative to Lane 6 because it is a shorter band. Lane 7 shows no band because the primers did not and was not supposed to anneal to the site because R0040 + I3502 was in a standard plasmid from this year and has a different sequence than all the 4 other plasmids that were used for this particular PCR. The primers are not complimentary to the sequence of the plasmids being used this year. So the fact that Lane 7 shows no band means there was no amplification- so the result is positive. Lanes 4 and 5 show exactly the same band because they are the same genes in the same plasmid backbone. Furthermore, Lane 9 shows no band because it is the Master Mix and our negative control. Therefore, it can be concluded that the primers are functional.
Apart from the Wet Lab, I've also helped Jeremey with the summary of our Lethbridge Workshop.
Jeremy
Today I completed the ligation and transformation of the I0500+E0034 plasmid switch. I also ran the PCR products and the results came back negative. So I set up another PCR and Gel.
Patrick
Today I helped plasmid prep the degP promoter and cpxR promoter on a GFP and RFP reporter device each. I also continued looking to literature regarding high and low-copy plasmids for the modelling project.
Himika
Today I plated the construction that I did last night. I also overnight cultured the construction from last night by inoculating from the cells to be plated. Tomorrow I will plasmid prep the cultures. I also looked into the literature regarding the plasmid which will be presented to Paul on Thursday.
Chris
Today, I did a colony PCR of the CpxP promoter that was transformed into both ampicillin-kanamycin and ampicillin-chloramphenicol plasmids. The results of the colony PCR were then run on a 1.2% gel and the bands were very irregular as shown on the right. The gel was run for approximately 1 hour and the bands did not move after 35 minutes. Another colony PCR was set up and let to run again overnight. As well, overnight culures were made of all 11 samples so they could be plasmid prepped appropriately if the Colony PCR showed up possible correct bands. As well, I helped Emily test the plate reader with several cultures that had arabinose inducible promoters induced overnight of GFP and RFP.
Alex
Today I did a plasmid prep of the overnight colonies of CpxR and DegP devices. I also made a gel and ran the colony PCR product. Three of the colonies appear to be the anticipated size, others appear to be failed constructions.
Emily
Today I retransformed the psB1AC3 and psB1AK3 vectors with the P1010 ccdb insert in them and I transformed them into d53.30 competant cells and plated on A,C and K plates. I also did a plasmid switch of B0034 from an Amp vector into AC and AK vectors. I antarctic phosphotased the vector and then ligated and transformed into TOP10 E coli cells. I then plated on A,C and K and left for overnight growth. I also spent a large portion of my day testing out the plate reader. I made serial dilutions of the ovenright cultures that I had made of a constitutive GFP circuit, a constitutive RFP circuit and B0015. I tested these all as well as a negative control of just LB broth and our 4 constructs of I0500-B0034-E0040 that have been induced with arabinose. Today I also filled more pipette tip boxes and autoclaved a bunch of necessary lab supplies like pipette tip boxes and 1.5 mL centrifuge tubes.
Himika
Today I did a colony PCR o
 "
"