Team:TU Delft/21 July 2010 content
From 2010.igem.org
(Difference between revisions)
(→Alkane degradation) |
(→Alkane degradation) |
||
| Line 1: | Line 1: | ||
==Alkane degradation== | ==Alkane degradation== | ||
Unfortunately there were no transformants on [https://2010.igem.org/Team:TU_Delft#/blog?blog=20_July_2010 yesterday's] plates. This is most likely due to the fact that the ligation didn't work with the small pieces of DNA of the RBSs. Next plan is to cut open the RBS plasmid without removing the RBS (with SpeI and PstI) and insert the gene in this plasmid. We will try this tomorrow. | Unfortunately there were no transformants on [https://2010.igem.org/Team:TU_Delft#/blog?blog=20_July_2010 yesterday's] plates. This is most likely due to the fact that the ligation didn't work with the small pieces of DNA of the RBSs. Next plan is to cut open the RBS plasmid without removing the RBS (with SpeI and PstI) and insert the gene in this plasmid. We will try this tomorrow. | ||
| + | |||
| + | ==Emulsifier== | ||
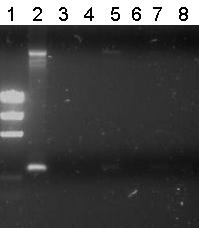
| + | [[Image:TU_Delft_PCR_Pieter_21_07_01.jpg|thumb|right|2% agarose gel]]There were small colonies on the plates. Pieter picked seven and performed Colony PCR on them. | ||
| + | |||
| + | '''Lane Description''' | ||
| + | {| style="color:black; background-color:white;" cellpadding="5" cellspacing="0" border="1" | ||
| + | |'''#''' | ||
| + | |'''Description''' | ||
| + | |'''Expected lenght (bp)''' | ||
| + | |- | ||
| + | |1 | ||
| + | |EZ Ladder (5 μL) | ||
| + | | | ||
| + | |- | ||
| + | |2 | ||
| + | |B0032 | ||
| + | |220 | ||
| + | |- | ||
| + | |3 | ||
| + | |Colony 1 | ||
| + | |300 | ||
| + | |- | ||
| + | |4 | ||
| + | |Colony 2 | ||
| + | |300 | ||
| + | |- | ||
| + | |5 | ||
| + | |Colony 3 | ||
| + | |300 | ||
| + | |- | ||
| + | |6 | ||
| + | |Colony 4 | ||
| + | |300 | ||
| + | |- | ||
| + | |7 | ||
| + | |Colony 5 | ||
| + | |300 | ||
| + | |- | ||
| + | |8 | ||
| + | |Colony 6 | ||
| + | |300 | ||
| + | |- | ||
| + | |9 | ||
| + | |Colony 7 | ||
| + | |300 | ||
| + | |} | ||
| + | |||
| + | In lane 2 we loaded the negative control (PCR Product of B0032). Unfortunately all other lanes were empty or the same as the negative control. So from this gel we conclude that the ligation has failed. | ||
Revision as of 09:50, 23 July 2010
Alkane degradation
Unfortunately there were no transformants on yesterday's plates. This is most likely due to the fact that the ligation didn't work with the small pieces of DNA of the RBSs. Next plan is to cut open the RBS plasmid without removing the RBS (with SpeI and PstI) and insert the gene in this plasmid. We will try this tomorrow.
Emulsifier
There were small colonies on the plates. Pieter picked seven and performed Colony PCR on them.Lane Description
| # | Description | Expected lenght (bp) |
| 1 | EZ Ladder (5 μL) | |
| 2 | B0032 | 220 |
| 3 | Colony 1 | 300 |
| 4 | Colony 2 | 300 |
| 5 | Colony 3 | 300 |
| 6 | Colony 4 | 300 |
| 7 | Colony 5 | 300 |
| 8 | Colony 6 | 300 |
| 9 | Colony 7 | 300 |
In lane 2 we loaded the negative control (PCR Product of B0032). Unfortunately all other lanes were empty or the same as the negative control. So from this gel we conclude that the ligation has failed.
 "
"