Team:MIT mammalian Switch
From 2010.igem.org
| Line 60: | Line 60: | ||
<div id="unique" style="padding:0px; font-size: 14px; border: 1px solid black; margin:0px; background-color:transparent;"> | <div id="unique" style="padding:0px; font-size: 14px; border: 1px solid black; margin:0px; background-color:transparent;"> | ||
| - | <table width=650px style="background-color: white; margin-top:5px; padding: 10px;"><tr><td><div class="bodybaby">Synthetic Switch: Implementation of Bistable Toggles in Mammalian Cells</div></td> | + | <table width=650px style="background-color: white; margin-top:5px; padding: 10px;"><tr><td><div class="bodybaby">Synthetic Switch: Implementation of Bistable Toggles in Mammalian Cells</div></td></tr> |
<tr><td>It is useful for a synthetic system capable of sensing mechanical stimuli to be able to convert a transient signal into a constitutive response. During the summer we designed both a complete and a simplified bistable toggle with a low and high output state using positive feedback loops. | <tr><td>It is useful for a synthetic system capable of sensing mechanical stimuli to be able to convert a transient signal into a constitutive response. During the summer we designed both a complete and a simplified bistable toggle with a low and high output state using positive feedback loops. | ||
| Line 71: | Line 71: | ||
<br>4. rtTA3 is transcribed after teh activation of EGSH. | <br>4. rtTA3 is transcribed after teh activation of EGSH. | ||
<br>5. Each rtTA3 binds to two of the six tetO sites in the TREt promoter and induces the promoter. | <br>5. Each rtTA3 binds to two of the six tetO sites in the TREt promoter and induces the promoter. | ||
| - | <br>6. rtTA, another version of rtTA3, further induces activation of the TREt promoter. This establishes a positive feedback loop, which propels the system from a low to a stable high output state. In the high output state, EYFP is constitutively expressed at a high level. | + | <br>6. rtTA, another version of rtTA3, further induces activation of the TREt promoter. This establishes a positive feedback loop, which propels the system from a low to a stable high output state (the switch is "flipped"). In the high output state, EYFP is constitutively expressed at a high level. |
| + | <br> | ||
| + | <li>Note 1: DOX in this system would be already present in the media. rtTA3 combines with DOX to form an activated complex in order to induce TREt. The outside signal being added is PonS. Without PonS addition, the presence of DOX is unable to flip the switch because there is very low level transcription of rtTA3. | ||
| + | <li>Note 2: 2A sites are cis elements that enable polycistronic translation in mammalian cells. Proteins before and after the 2A site are translated at roughly equal frequency. | ||
| + | <br> The three components of this system has been fully constructed and are ready for transfection into mammalian cells. Unfortunately at the present, due to time constraints, we are unable to provide conclusive data supporting the functionality of the toggle. The figure below show a preliminary experiment during which PonS was added post transient calcium phosphate transfection of a HEK293FT cell line containing the Hef1a_VgEcR_RxR construct with the other two components of the toggle. CMV_EGFP was transfected in other wells to control for transfection efficiency. | ||
| + | </tr> | ||
| + | <tr><td><img src=https://static.igem.org/mediawiki/2010/0/07/Simplified_Toggle_Micrographs.png> | ||
| + | <p>There is significant increase in fluorescence in the cells transfected with the toggle within 7 hours after the addition of ponS. We did not have enough DNA to perform a parallel transfection without PonS addition; however, we argue that the increase in fluorescence we saw 7 hours after PonS addition is much greater than in the previous 41 hours post co-transfection. Thus it can be concluded that the co-tranfescted construct shows EYFP fluorescence with, but not without, PonS induction. | ||
</td> | </td> | ||
Revision as of 10:38, 27 October 2010
Synthetic Switch: Implementation of Bistable Toggles in Mammalian Cells |
It is useful for a synthetic system capable of sensing mechanical stimuli to be able to convert a transient signal into a constitutive response. During the summer we designed both a complete and a simplified bistable toggle with a low and high output state using positive feedback loops.
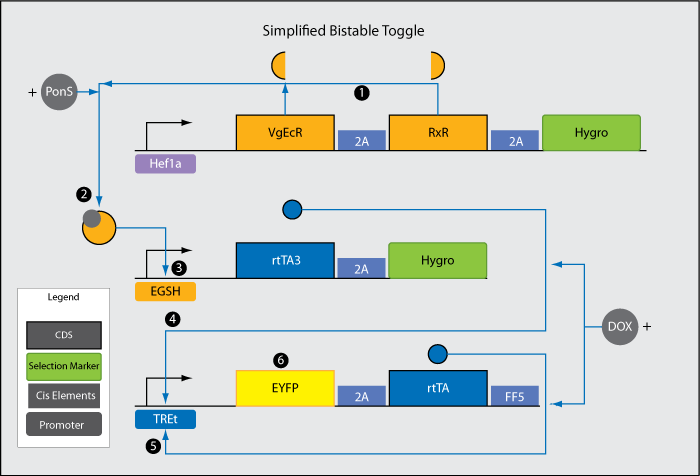
The three components of this system has been fully constructed and are ready for transfection into mammalian cells. Unfortunately at the present, due to time constraints, we are unable to provide conclusive data supporting the functionality of the toggle. The figure below show a preliminary experiment during which PonS was added post transient calcium phosphate transfection of a HEK293FT cell line containing the Hef1a_VgEcR_RxR construct with the other two components of the toggle. CMV_EGFP was transfected in other wells to control for transfection efficiency. |

There is significant increase in fluorescence in the cells transfected with the toggle within 7 hours after the addition of ponS. We did not have enough DNA to perform a parallel transfection without PonS addition; however, we argue that the increase in fluorescence we saw 7 hours after PonS addition is much greater than in the previous 41 hours post co-transfection. Thus it can be concluded that the co-tranfescted construct shows EYFP fluorescence with, but not without, PonS induction. |
 "
"
 iGEM 2010
iGEM 2010