Team:BCCS-Bristol/Wetlab/K381001 Construction
From 2010.igem.org
| Line 3: | Line 3: | ||
| - | + | =Constructing Our Biobrick - K381001= | |
| + | |||
| + | ==Overview== | ||
| + | |||
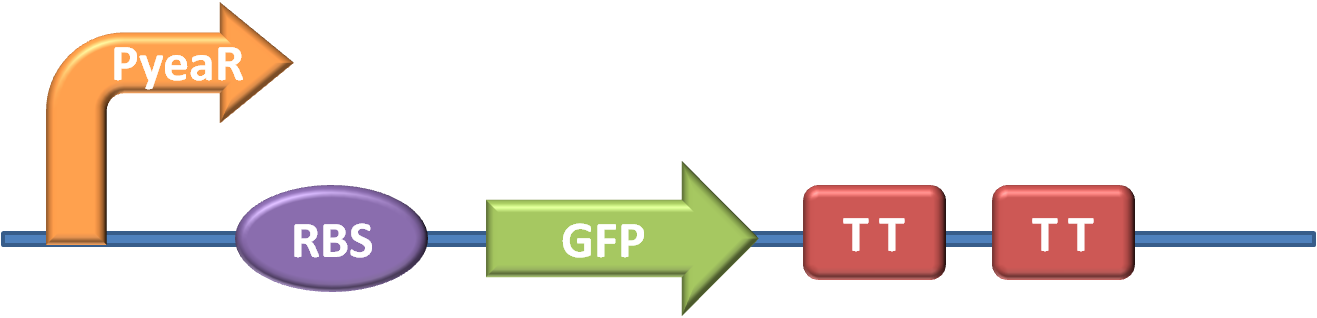
| + | The first stage in the project was to aquire the necessary parts and construct our key biobrick - one that would trigger GFP production in response to Nitrates or Nitrites. The design of the part can be seen in fig. 1 (for more detailed information about our new brick BBa_K381001 look on our part design page) | ||
| + | |||
| + | [[Image:Part.png|centre|300px]] | ||
| + | |||
| + | The promoter is BBa_K216005, and was requested from the parts registry | ||
| + | The GFP generator along with strong RBS and transcription terminators is BBa_E0840, and was taken from well 12O kitplate 1 of the 2010 distribution. | ||
| + | |||
| + | These two parts were cut from their respective plasmids and ligated together. The resulting part was then transformed into commercial NovaBlue cells, then miniprepped to generate large amounts. | ||
| - | |||
| - | |||
| - | |||
Construction of the biobrick can be broadly broken down into | Construction of the biobrick can be broadly broken down into | ||
| Line 18: | Line 26: | ||
| - | Generating large quantities of DNA | + | ==Generating large quantities of DNA== |
| - | + | ||
| + | Before using the biobricks from the parts registry, we first had to obtain large amounts of these plasmids as the amount provided by the registry was is not sufficient. This is done as follows: | ||
| + | |||
| + | * Commercial NovaBlue competent cells were transformed with the DNA from the parts registry, typically transforming 50μL of cells with 2μL of DNA | ||
* Colonies from these transformations were then selected and used to grow overnights - using a wire loop to inoculate 5mL of LB medium (5μL of Ampicillin also added) | * Colonies from these transformations were then selected and used to grow overnights - using a wire loop to inoculate 5mL of LB medium (5μL of Ampicillin also added) | ||
* Combined 3mL of BBa_K216005, BBa_E0840 and BBa_E0240 cell cultures in 1.5mL eppendorf tubes twice - total of 6mL combined cell culture for each biobrick. Performed a miniprep on the combined cultures using a Qiagen "QIAprep Spin Miniprep Kit". Eluted 100μL of solution containing BBa_K216005, BBa_E0840 and BBa_E0240 biobricks in high concentration. | * Combined 3mL of BBa_K216005, BBa_E0840 and BBa_E0240 cell cultures in 1.5mL eppendorf tubes twice - total of 6mL combined cell culture for each biobrick. Performed a miniprep on the combined cultures using a Qiagen "QIAprep Spin Miniprep Kit". Eluted 100μL of solution containing BBa_K216005, BBa_E0840 and BBa_E0240 biobricks in high concentration. | ||
| - | Digesting Component Biobricks: | + | |
| + | |||
| + | ==Digesting Component Biobricks== | ||
| + | |||
| + | Once we possessed large quantities of each of the biobricks we wanted to use, we then needed to digest them with appropriate enzymes to leave sticky ends that would match correctly, this was done as follows: | ||
* BBa_E0840 and BBa_E0240 were digested with Xba1 and Pst1 using the following mix: | * BBa_E0840 and BBa_E0240 were digested with Xba1 and Pst1 using the following mix: | ||
Revision as of 13:04, 25 October 2010
iGEM 2010
Contents |
Constructing Our Biobrick - K381001
Overview
The first stage in the project was to aquire the necessary parts and construct our key biobrick - one that would trigger GFP production in response to Nitrates or Nitrites. The design of the part can be seen in fig. 1 (for more detailed information about our new brick BBa_K381001 look on our part design page)
The promoter is BBa_K216005, and was requested from the parts registry The GFP generator along with strong RBS and transcription terminators is BBa_E0840, and was taken from well 12O kitplate 1 of the 2010 distribution.
These two parts were cut from their respective plasmids and ligated together. The resulting part was then transformed into commercial NovaBlue cells, then miniprepped to generate large amounts.
Construction of the biobrick can be broadly broken down into
- Generating larger quantities of both our component biobricks
- Digesting each of these biobricks
- Phosphatase treatment of the vector and gel extraction of the digests
- Ligating together the desired parts to create a new biobrick
- Generating larger quantities of this biobrick, checking and sequencing
Generating large quantities of DNA
Before using the biobricks from the parts registry, we first had to obtain large amounts of these plasmids as the amount provided by the registry was is not sufficient. This is done as follows:
- Commercial NovaBlue competent cells were transformed with the DNA from the parts registry, typically transforming 50μL of cells with 2μL of DNA
- Colonies from these transformations were then selected and used to grow overnights - using a wire loop to inoculate 5mL of LB medium (5μL of Ampicillin also added)
- Combined 3mL of BBa_K216005, BBa_E0840 and BBa_E0240 cell cultures in 1.5mL eppendorf tubes twice - total of 6mL combined cell culture for each biobrick. Performed a miniprep on the combined cultures using a Qiagen "QIAprep Spin Miniprep Kit". Eluted 100μL of solution containing BBa_K216005, BBa_E0840 and BBa_E0240 biobricks in high concentration.
Digesting Component Biobricks
Once we possessed large quantities of each of the biobricks we wanted to use, we then needed to digest them with appropriate enzymes to leave sticky ends that would match correctly, this was done as follows:
- BBa_E0840 and BBa_E0240 were digested with Xba1 and Pst1 using the following mix:
- 30μL DNA
- 0.5μL BSA 100x
- 5μL NEB Buffer 3
- 1μL Pst1
- 1.3μL Xba1
- 12.2μL ddH2O
- BBa_K216005 was digested with Spe1 and Pst1 using the following mix:
- 30μL DNA
- 0.5μL BSA 100x
- 5μL NEB Buffer 2
- 1.3μL Pst1
- 1μL Spe1
- 12.2μL ddH2O
- Each digest was left for 2 hours at 37°C
 "
"