Team:SDU-Denmark/labnotes
From 2010.igem.org
(Difference between revisions)
(→Amplification of FlhDC with Taq) |
(→Restriction digest of BBa_E0040 miniprep product) |
||
| (22 intermediate revisions not shown) | |||
| Line 385: | Line 385: | ||
===Amplification of pSB3K3 with Taq=== | ===Amplification of pSB3K3 with Taq=== | ||
<br> | <br> | ||
| - | ''Start date | + | ''Start date'': July 14th 2010<br><br> |
''Methods'': [https://2010.igem.org/Team:SDU-Denmark/labnotes#Exp._3 The purified pSB3K3 plasmids] were amplified by PCR using Taq, as a test, before using the more expensive Pfu. <br><br> | ''Methods'': [https://2010.igem.org/Team:SDU-Denmark/labnotes#Exp._3 The purified pSB3K3 plasmids] were amplified by PCR using Taq, as a test, before using the more expensive Pfu. <br><br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ||
| - | |||
''Notes'': The plasmids were collected from tubes green 10 and green 11 in the freezer. VR and VF2 primers were used.<br> | ''Notes'': The plasmids were collected from tubes green 10 and green 11 in the freezer. VR and VF2 primers were used.<br> | ||
| - | The experiment were performed simultaneously with the Taq control version of [https://2010.igem.org/Team:SDU-Denmark/labnotes# | + | The experiment were performed simultaneously with the Taq control version of [https://2010.igem.org/Team:SDU-Denmark/labnotes#Extraction_of_B0034_from_pSB1A2_plasmid_using_PFU the extraction of B0034 from pSB1A2 plasmid] done by the phototaxis goup <br><br> |
--[[User:Sheila|Sheila]] 09:08, 14 July 2010 (UTC) | --[[User:Sheila|Sheila]] 09:08, 14 July 2010 (UTC) | ||
| Line 397: | Line 396: | ||
''Date:'' July 14th <br><br> | ''Date:'' July 14th <br><br> | ||
''Experiment done by:'' Pernille and Louise <br><br> | ''Experiment done by:'' Pernille and Louise <br><br> | ||
| - | ''Methods:'' We used a | + | ''Methods:'' We used a overnight culture of ''E. coli'' MG1655 cells <br><br> |
''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#GP1.1 GP1.1]<br><br> | ''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#GP1.1 GP1.1]<br><br> | ||
| - | ''Notes:'' We made four | + | ''Notes:'' We made four tubes with 300 microlitres of ''E. coli'' MG1655 cell media instead of 200 microlitres. In tube 1 and 2 we unfortunately added 800 microlitres un-diluted precipitation solution, in tube 3 and 4 we diluted the precipitation solution according to the GP1.1 protocol. <br><br> |
| - | ''Results:'' The concentration of the purified DNA was measured by | + | ''Results:'' The concentration of the purified DNA was measured by NanoDrop. Tube 1 and 2 did not contain any DNA which we had expected because of the high concentration of precipitation soultion. The DNA concentration in tube 3 and 4 were 28,38ng/uL and 19,4ng/uL, respectively. Tube 3 and 4 were afterwards pooled and saved in the freezer (labeled with white tag 1). The purified DNA was run on a agarose gel to check if it actual contains DNA. |
<br><br> | <br><br> | ||
--[[User:Louch07|Louise]] 10:44, 14 July 2010 (UTC) | --[[User:Louch07|Louise]] 10:44, 14 July 2010 (UTC) | ||
| Line 410: | Line 409: | ||
''Date'': July 15th<br><br> | ''Date'': July 15th<br><br> | ||
''Experiment done by'': Sheila<br><br> | ''Experiment done by'': Sheila<br><br> | ||
| - | ''Methods'': The DNA purification was performed on an | + | ''Methods'': The DNA purification was performed on an overnight culture of the ''E. coli'' strain MG1655 . Three samples of 400 microlitres of culture were extracted, centrifuged, the supernatant removed and the pellet was redissolved in 200 microlitres water<br><br> |
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#GP1.1 GP1.1]<br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#GP1.1 GP1.1]<br><br> | ||
''Notes'': <br><br> | ''Notes'': <br><br> | ||
| - | ''Results'': Not a lot of DNA was detected | + | ''Results'': Not a lot of DNA was detected with the NanoDrop. The samples yielded concentrations of 9,88ng/µL, 13,5ng/µL and 5,3ng/µL respectively. The gel showed 4 bands of DNA in each sample. The samples were located at 200bp, 950bp, 1500bp and >10000bp. The bands less visible in lane 3, which were loaded with sample 3, which also yielded the least concentration.<br><br> |
--[[User:Sheila|Sheila]] 09:20, 15 July 2010 (UTC)<br><br> | --[[User:Sheila|Sheila]] 09:20, 15 July 2010 (UTC)<br><br> | ||
| Line 419: | Line 418: | ||
<br> | <br> | ||
''Experiment done by:'' Christian<br><br> | ''Experiment done by:'' Christian<br><br> | ||
| - | ''Date:'' | + | ''Date:'' July 14th<br><br> |
| - | ''Methods:'' PCR on purified | + | ''Methods:'' PCR on purified chromosomal DNA <br><br> |
''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ||
''Notes:'' 4 tubes loaded with DNA from freezer: 2x(8 green) and 2x(9 green). Both tubes were discarded as they were empty.<br> | ''Notes:'' 4 tubes loaded with DNA from freezer: 2x(8 green) and 2x(9 green). Both tubes were discarded as they were empty.<br> | ||
| Line 426: | Line 425: | ||
Primers used were FlhD fw and FlhC rw. <br><br> | Primers used were FlhD fw and FlhC rw. <br><br> | ||
Anealing temperature was set to 48°C and elongation was set to 2 minutes. <br><br> | Anealing temperature was set to 48°C and elongation was set to 2 minutes. <br><br> | ||
| - | 1,5% | + | 1,5% agarose gel was loaded with Generuler DNA ladder mix. <br><br> |
''Results:''Band showed at around 3kb in both wells for (8 green) and one well for (9 green) <br><br> | ''Results:''Band showed at around 3kb in both wells for (8 green) and one well for (9 green) <br><br> | ||
''Analysis:'' The FlhDC regulon was not extracted. <br><br> | ''Analysis:'' The FlhDC regulon was not extracted. <br><br> | ||
| Line 438: | Line 437: | ||
''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2]<br><br> | ''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2]<br><br> | ||
''Note:'' Polymerase used: TAQ <br><br> | ''Note:'' Polymerase used: TAQ <br><br> | ||
| - | ''Methods:'' PCR on purified MG1655 chromosomal DNA | + | ''Methods:'' PCR on purified ''E. coli'' MG1655 chromosomal DNA from Gel electrophoresis.<br><br> We used the four new primers we designed in the order: FlhDC fw, FlhDC rev, FlhDCmut fw and FlhDCmut rev. <br> We ran a gradient PCR with six different annealing temperatures for three primer combinations (Tables below).<br><br> |
'''PCR-mixes:''' | '''PCR-mixes:''' | ||
<br> | <br> | ||
| Line 552: | Line 551: | ||
</td> | </td> | ||
<td style="vertical-align: top;">Elongation<br> | <td style="vertical-align: top;">Elongation<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">x 29<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">-<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">GOTO step 2<br> | ||
</td> | </td> | ||
</tr> | </tr> | ||
| Line 557: | Line 564: | ||
<td style="vertical-align: top;">5 min<br> | <td style="vertical-align: top;">5 min<br> | ||
</td> | </td> | ||
| - | <td style="vertical-align: top;"> | + | <td style="vertical-align: top;">72<br> |
</td> | </td> | ||
<td style="vertical-align: top;">End<br> | <td style="vertical-align: top;">End<br> | ||
| Line 577: | Line 584: | ||
cellpadding="2" cellspacing="2"> | cellpadding="2" cellspacing="2"> | ||
<tr> | <tr> | ||
| - | <td style="vertical-align: top;"> | + | <td style="vertical-align: top;">Column<br> |
</td> | </td> | ||
<td style="vertical-align: top;">Temperature (Celcius)<br> | <td style="vertical-align: top;">Temperature (Celcius)<br> | ||
| Line 623: | Line 630: | ||
'''''Gel electrophoresis''''' | '''''Gel electrophoresis''''' | ||
<br> | <br> | ||
| - | ''Method:'' We used a 1.5% gel and two | + | ''Method:'' We used a 1.5% agarose gel and two ladders (GeneRuler DNA ladder mix), 8 microlitres PCR product was loaded together with 2 microlitres agarose loading dye<br><br> |
'''Loading Table''' | '''Loading Table''' | ||
<table style="text-align: left; width: 100%;" border="1" cellpadding="2" | <table style="text-align: left; width: 100%;" border="1" cellpadding="2" | ||
| Line 638: | Line 645: | ||
<td style="vertical-align: top;">1<br> | <td style="vertical-align: top;">1<br> | ||
</td> | </td> | ||
| - | <td style="vertical-align: top;"> | + | <td style="vertical-align: top;">GeneRuler DNA ladder mix<br> |
</td> | </td> | ||
<td style="vertical-align: top;">-<br> | <td style="vertical-align: top;">-<br> | ||
| Line 646: | Line 653: | ||
<td style="vertical-align: top;">2<br> | <td style="vertical-align: top;">2<br> | ||
</td> | </td> | ||
| - | <td style="vertical-align: top;">3.4 (PCR-mix 3. | + | <td style="vertical-align: top;">3.4 (PCR-mix 3. Column 4)<br> |
</td> | </td> | ||
<td style="vertical-align: top;">FlhDC fw + FlhDC rev<br> | <td style="vertical-align: top;">FlhDC fw + FlhDC rev<br> | ||
| Line 776: | Line 783: | ||
<td style="vertical-align: top;">20<br> | <td style="vertical-align: top;">20<br> | ||
</td> | </td> | ||
| - | <td style="vertical-align: top;"> | + | <td style="vertical-align: top;">GeneRuler DNA ladder mix<br> |
</td> | </td> | ||
<td style="vertical-align: top;">-<br> | <td style="vertical-align: top;">-<br> | ||
| Line 787: | Line 794: | ||
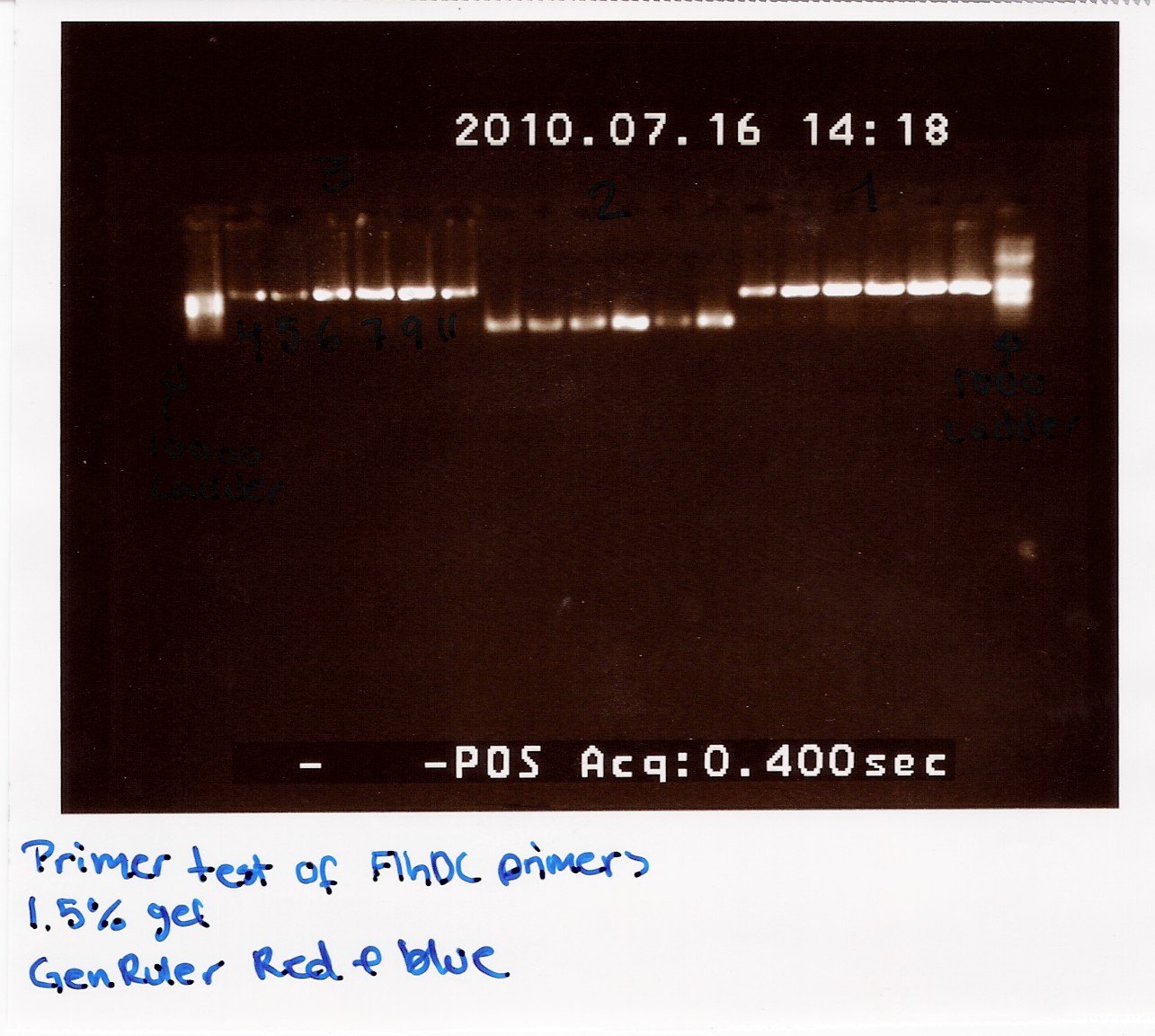
[[Image:Team-SDU_Denmark-Primer_test_of_FlhDC.jpg|300px]] | [[Image:Team-SDU_Denmark-Primer_test_of_FlhDC.jpg|300px]] | ||
<br> | <br> | ||
| - | The image shows PCR results with the right lengths. Strong expression for PCR with primers; FlhDC fw and FlhDCmut rev is seen at temperatures 53.4 | + | The image shows PCR results with the right lengths. Strong expression for PCR with primers; FlhDC fw and FlhDCmut rev is seen at temperatures 53.4 Celcius, 56.1 Celcius and 60.1 Celcius, FlhDCmut fv and FlhDC rev is seen at 56.1 celcius, and FlhDC fw and FlhDC rev is seen at all gradient temperatures. The Gradient shows a good result for all three PCR's at 56.1 Celcius.<br><br> |
--[[User:Louch07|Louise]] 12:50, 16 July 2010 (UTC) | --[[User:Louch07|Louise]] 12:50, 16 July 2010 (UTC) | ||
<br><br> | <br><br> | ||
| Line 797: | Line 804: | ||
''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1]<br><br> | ''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1]<br><br> | ||
''Note:'' Polymerase used: Pfu. Three different samples were prepared as was done,[https://2010.igem.org/Team:SDU-Denmark/labnotes#Checking_the_new_primers when the new primers were tested with Taq] (one, copied the entire operon, the second copied the gene from FlhD to the restriction site in FlhC and the third copied from the restriction site to the end of the FlhC gene), and run at three different temperatures: 50,8˚C, 56,1˚C and 64,5˚C respectively. The temperatures were chosen based on the clearest bands in the gel run in the before mentioned experiment. <br><br> | ''Note:'' Polymerase used: Pfu. Three different samples were prepared as was done,[https://2010.igem.org/Team:SDU-Denmark/labnotes#Checking_the_new_primers when the new primers were tested with Taq] (one, copied the entire operon, the second copied the gene from FlhD to the restriction site in FlhC and the third copied from the restriction site to the end of the FlhC gene), and run at three different temperatures: 50,8˚C, 56,1˚C and 64,5˚C respectively. The temperatures were chosen based on the clearest bands in the gel run in the before mentioned experiment. <br><br> | ||
| - | ''Methods:'' PCR on purified MG1655 chromosomal DNA | + | ''Methods:'' PCR on purified ''E. coli'' MG1655 chromosomal DNA from Gel electrophoresis.<br><br> Primers used: FlhDC fw, FlhDC rev, FlhDCmut fw and FlhDCmut rev. <br><br> |
| - | ''Results:'' At last, we have PCR results. The gel showed clear bands of both mutated pieces of the gene, as well as the entire operon. And because this PCR was run with Pfu, we can actually use these results and move on with the | + | ''Results:'' At last, we have PCR results. The gel showed clear bands of both mutated pieces of the gene, as well as the entire operon. And because this PCR was run with Pfu, we can actually use these results and move on with the project.<br><br> |
--[[User:Sheila|Sheila]] 15:31, 16 July 2010 (UTC) | --[[User:Sheila|Sheila]] 15:31, 16 July 2010 (UTC) | ||
| Line 806: | Line 813: | ||
== Group: Retinal == | == Group: Retinal == | ||
| - | === Transformation of TOP10 e. coli with [http://partsregistry.org/Part:BBa_K274210 | + | === Transformation of TOP10 ''e. coli'' with [http://partsregistry.org/Part:BBa_K274210 K274210]=== |
<br> | <br> | ||
Experiment continued from last week. | Experiment continued from last week. | ||
<br><br> | <br><br> | ||
'''Colony PCR of K274210 in pSB1A2'''<br><br> | '''Colony PCR of K274210 in pSB1A2'''<br><br> | ||
| - | Start date: July | + | Start date: July 12th<br><br> |
Experiment done by: Christian, Lars Christian<br><br> | Experiment done by: Christian, Lars Christian<br><br> | ||
''Methods:'' Colony PCR (modified) <br><br> | ''Methods:'' Colony PCR (modified) <br><br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ||
| - | ''Notes:'' MgCl2 was added as a gradient, samples 1-2 contain 2 | + | ''Notes:'' MgCl2 was added as a gradient, samples 1-2 contain 2 microlitres (0,04255), samples 3-5 2,25 microlitres (0,04787). We mistakenly added 30µl (instead of 17µl) premix to samples 6 - 9, so the concentration of MgCl2 is reduced. Sample 6 contains 2,25 microlitres (0,0375), samples 7-8 2,5 microlitres (0,04167), sample 9 2,75 microlitres (0,04583). <br><br> |
Gel was loaded with DNA-ladder plus, with the upper marker at 5000 bp.<br><br> | Gel was loaded with DNA-ladder plus, with the upper marker at 5000 bp.<br><br> | ||
| - | ''Results:'' We found plasmids at the expected 5 | + | ''Results:'' We found plasmids at the expected 5 kb marker in colonies 1-8. Successfull plates were kept in incubator over night. They will be made into overnight cultures and thereafter frozen.<br><br> |
| - | ''Analysis:'' It seems we have transformed our cells with | + | ''Analysis:'' It seems we have transformed our cells with K274210. Confirmation will come when we run experiments to demonstrate presence of beta-carotene. <br><br> |
| - | === Transformation of [http://partsregistry.org/Part:BBa_R0011 | + | === Transformation of [http://partsregistry.org/Part:BBa_R0011 R0011 ]and [http://partsregistry.org/Part:BBa_I0500 I0500] === |
| - | Start date: | + | Start date: July 12th End date:July 14th<br> |
| - | ''Methods:'' | + | ''Methods:'' Overnight culture, competent cells, transformation.<br> |
''Protocols:''[https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1]. | ''Protocols:''[https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1]. | ||
<br><br> | <br><br> | ||
| - | '''Making competent E.Coli TOP 10'''<br> | + | '''Making competent ''E.Coli'' TOP 10'''<br> |
Experiment done by: Christian, Maria and LC<br> | Experiment done by: Christian, Maria and LC<br> | ||
| - | Date: | + | Date: July 12th<br> |
Notes: Everything was done according to protocol.<br> | Notes: Everything was done according to protocol.<br> | ||
Results: We made competent cells for use within 48 hours.<br> | Results: We made competent cells for use within 48 hours.<br> | ||
| Line 834: | Line 841: | ||
'''Transformation of pBad and lacl promoter'''<br> | '''Transformation of pBad and lacl promoter'''<br> | ||
Experiment done by: Christian, Maria and LC<br> | Experiment done by: Christian, Maria and LC<br> | ||
| - | Date: | + | Date: July 12th - 13th<br> |
| - | Notes: Added | + | Notes: Added 200 microlitres of the un-pelleted cells, instead of 150µl to the agar plates.<br>Some of the agar dishes were damaged during drying and plating. We tried to use them anyway.<br><br> |
Results: No plates showed colonies, apart from the positive control<br> | Results: No plates showed colonies, apart from the positive control<br> | ||
Analysis: We suspect the agar dishes might have been the cause of this.<br><br> | Analysis: We suspect the agar dishes might have been the cause of this.<br><br> | ||
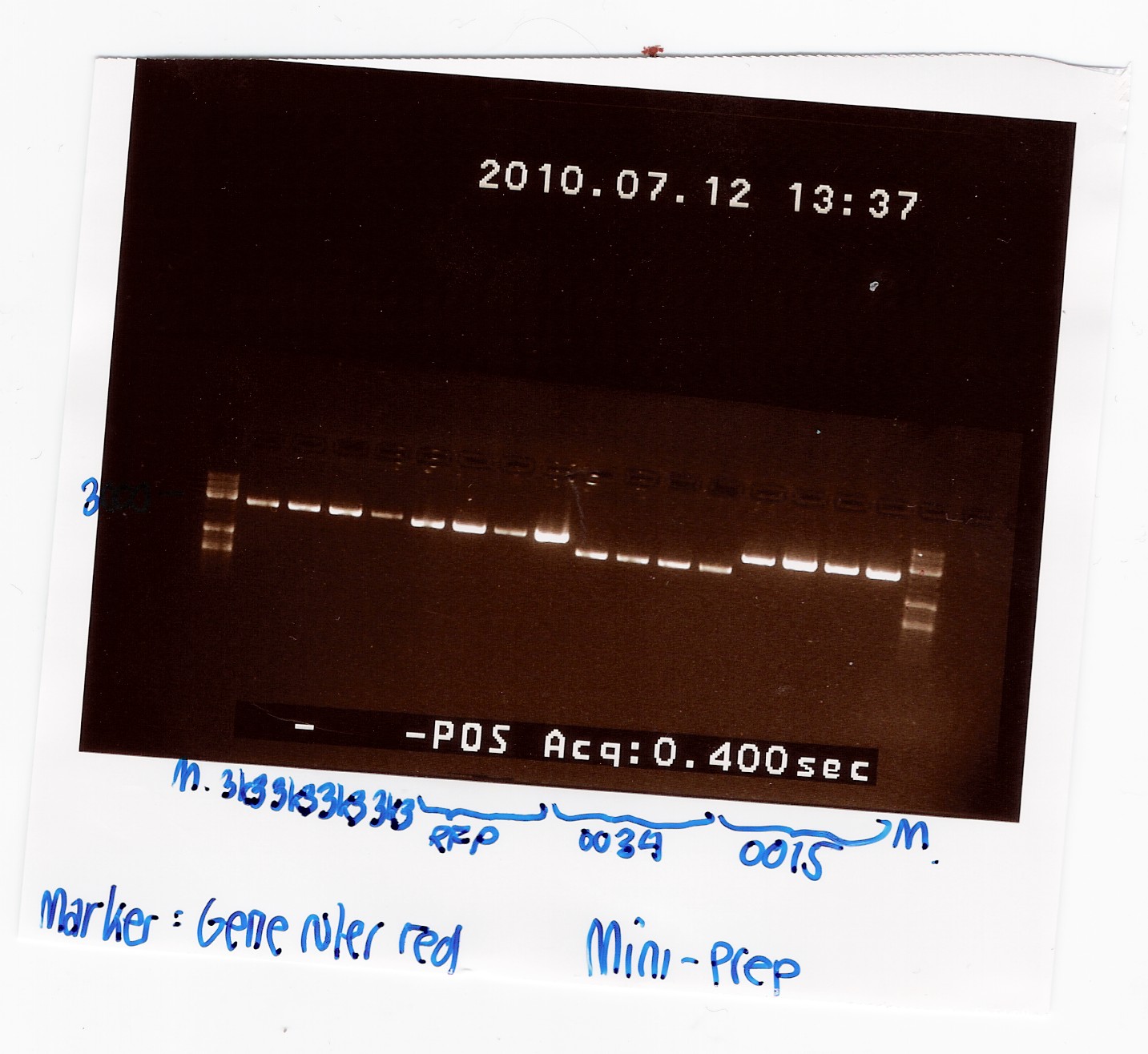
| - | === Mini-prep of pSB1A2 w. [http://partsregistry.org/Part:BBa_B0034 | + | === Mini-prep of pSB1A2 w. [http://partsregistry.org/Part:BBa_B0034 B0034], pSB1AK3 w. [http://partsregistry.org/Part:BBa_B0015 B0015] and pSB3K3 w. [http://partsregistry.org/Part:BBa_J04450 J04450] (transformation from 08/07) === |
| - | Start date: | + | Start date: July 12th<br> |
''Methods:'' Mini-prep <br><br> | ''Methods:'' Mini-prep <br><br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.2 MP1.2] <br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.2 MP1.2] <br><br> | ||
''Notes:'' | ''Notes:'' | ||
| - | Samples were loaded into a 1% gel with | + | Samples were loaded into a 1% agarose gel with GeneRuler DNA ladder mix from Fermentas as marker.<br><br> |
''Results:''<br> | ''Results:''<br> | ||
Gel electrophoresis: <br> | Gel electrophoresis: <br> | ||
| Line 1,028: | Line 1,035: | ||
All bands appeared to be about 1000 kb smaller than the expected size. This could be due to the supercoiling of the plasmids. To verify this, the plasmids were cut with PstI: [https://2010.igem.org/Team:SDU-Denmark/labnotes#Digestion_of_pSB1A2_w._B0034.2C_pSB1AK3_w._B0015_and_pSB3K3_w._J04450_and_pSB1A2_using_pstI.28miniprep_from_12.2F07.29 Digestion of plasmids] | All bands appeared to be about 1000 kb smaller than the expected size. This could be due to the supercoiling of the plasmids. To verify this, the plasmids were cut with PstI: [https://2010.igem.org/Team:SDU-Denmark/labnotes#Digestion_of_pSB1A2_w._B0034.2C_pSB1AK3_w._B0015_and_pSB3K3_w._J04450_and_pSB1A2_using_pstI.28miniprep_from_12.2F07.29 Digestion of plasmids] | ||
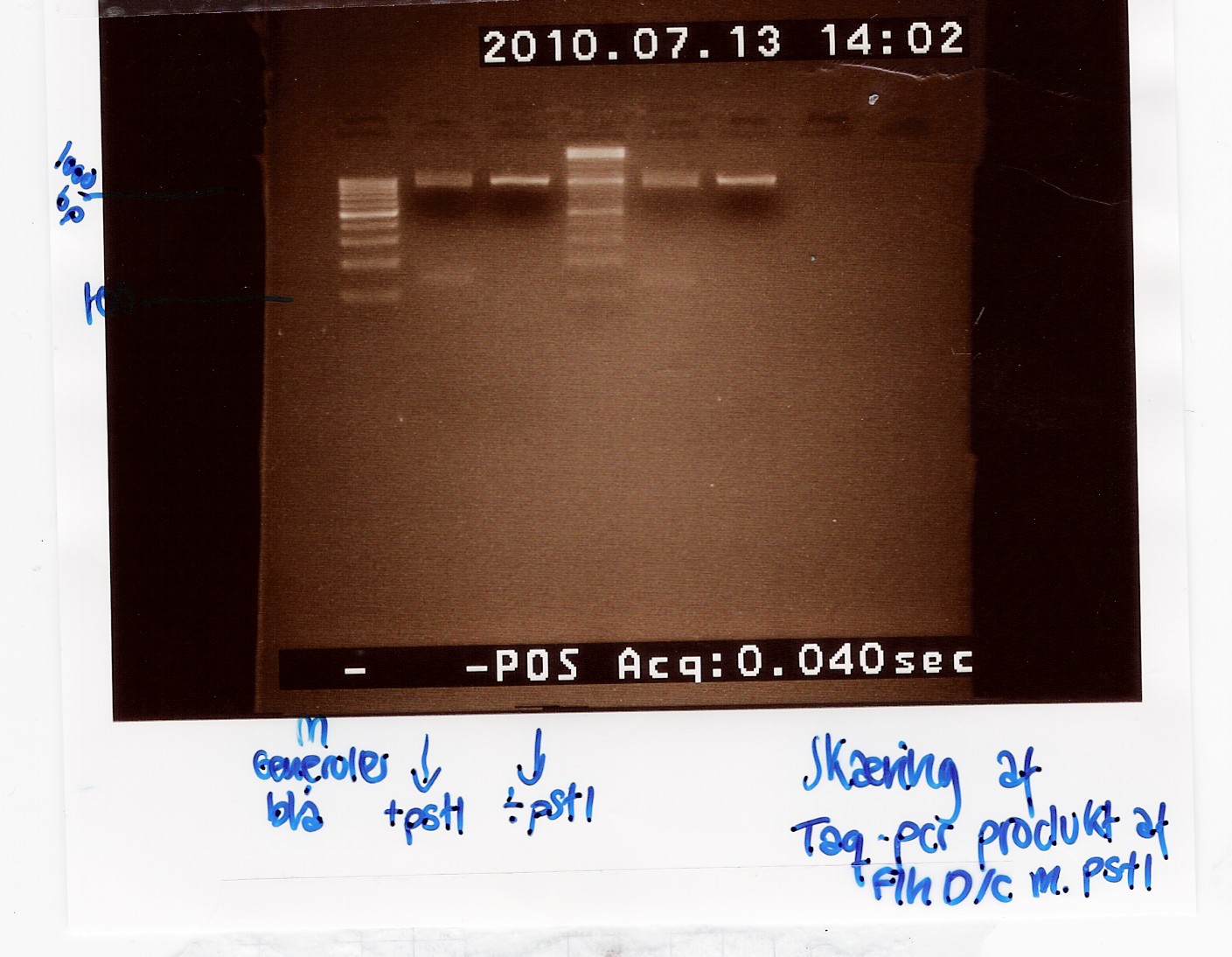
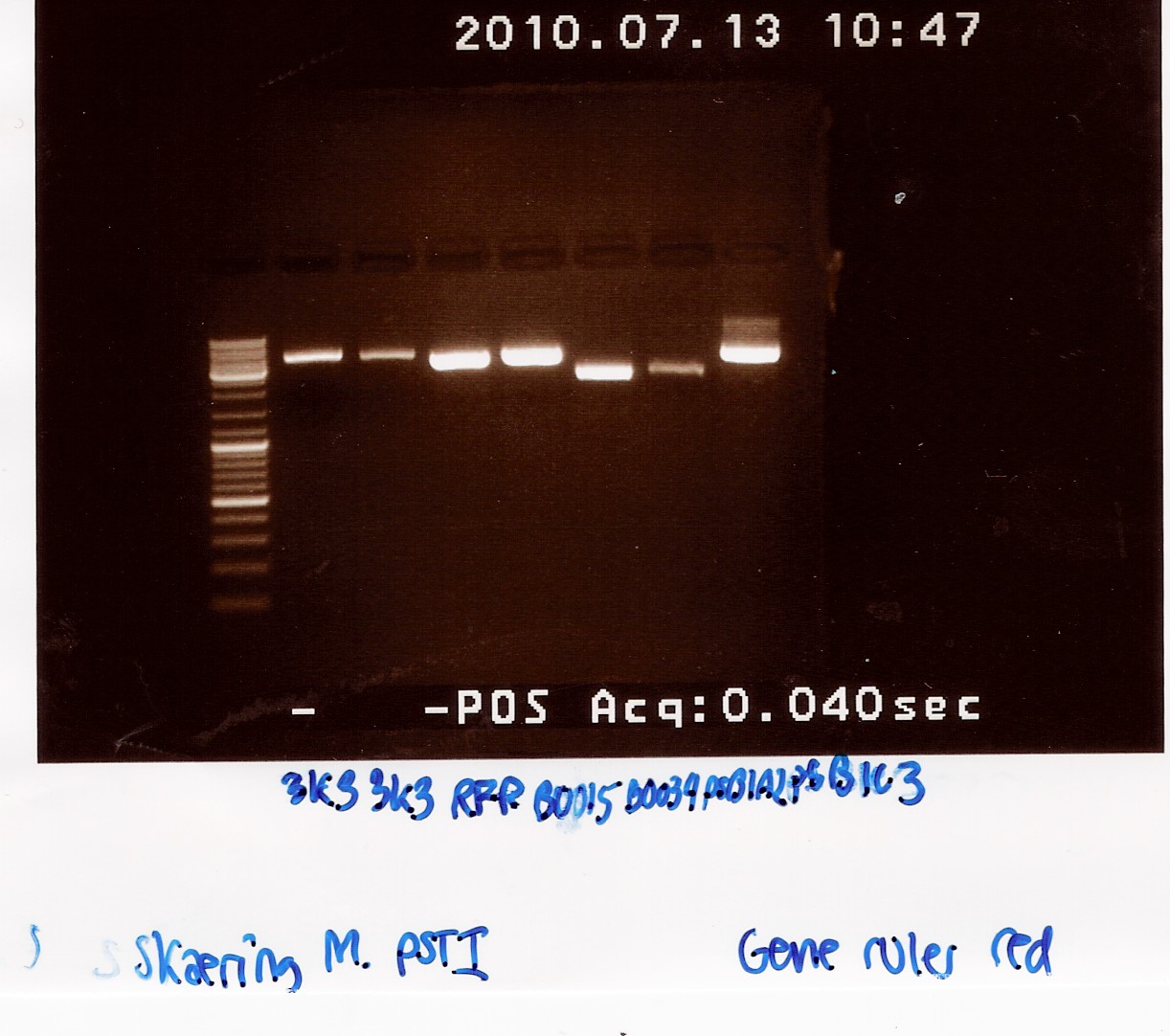
| - | === Digestion of pSB1A2 w. [http://partsregistry.org/Part:BBa_B0034 | + | === Digestion of pSB1A2 w. [http://partsregistry.org/Part:BBa_B0034 B0034], pSB1AK3 w. [http://partsregistry.org/Part:BBa_B0015 B0015] and pSB3K3 w. [http://partsregistry.org/Part:BBa_J04450 J04450]and pSB1A2 using pstI(miniprep from 12/07) === |
| - | Start date: | + | Start date: July 13th<br> |
''Methods:'' Digestion <br> | ''Methods:'' Digestion <br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1]<br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1]<br> | ||
''Experiment done by:'' Maria and Sheila<br><br> | ''Experiment done by:'' Maria and Sheila<br><br> | ||
''Notes'': Digestion of the plasmids with a single restriction enzyme was done to ensure that the plasmids extracted from [https://2010.igem.org/Team:SDU-Denmark/labnotes#Mini-prep_of_pSB1A2_w._B0034.2C_pSB1AK3_w._B0015_and_pSB3K3_w._J04450.28transformation_from_08.2F07.29 mini-prep] has the correct size. Digestion was done with half the amount of restriction mixture. <br><br> | ''Notes'': Digestion of the plasmids with a single restriction enzyme was done to ensure that the plasmids extracted from [https://2010.igem.org/Team:SDU-Denmark/labnotes#Mini-prep_of_pSB1A2_w._B0034.2C_pSB1AK3_w._B0015_and_pSB3K3_w._J04450.28transformation_from_08.2F07.29 mini-prep] has the correct size. Digestion was done with half the amount of restriction mixture. <br><br> | ||
| - | Samples were loaded into a 1.5% gel with | + | Samples were loaded into a 1.5% agarose gel with GeneRuler DNA ladder mix from Fermentas as marker. <br><br> |
''Results:''<br> | ''Results:''<br> | ||
Gel electrophoresis: <br> | Gel electrophoresis: <br> | ||
| Line 1,044: | Line 1,051: | ||
--[[User:Tipi|Tipi]] 15:53, 19 July 2010 (UTC) | --[[User:Tipi|Tipi]] 15:53, 19 July 2010 (UTC) | ||
| - | === Transformation of | + | === Transformation of ''E. coli'' MG1655 with [http://partsregistry.org/Part:BBa_K274210 K274210], [http://partsregistry.org/Part:BBa_E0040 E0040] and [http://partsregistry.org/Part:BBa_I0500 I0500] === |
| - | Start Date: | + | Start Date: July 13th <br> |
| - | ''Methods:'' | + | ''Methods:'' overnight cultures, Competent Cells, Transformation of competent cells. <br> |
''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1]<br><br> | ''Protocols:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1]<br><br> | ||
'''Experiment:''' Making competent ''E. coli'' MG1655<br> | '''Experiment:''' Making competent ''E. coli'' MG1655<br> | ||
''Experiment done by:'' Christian<br> | ''Experiment done by:'' Christian<br> | ||
| - | ''Date:'' | + | ''Date:'' July 13th<br><br> |
| - | + | ''Notes:'' Growth was started at 09.30 with an OD550 of 0.19A. The cells were harvested at 10.26 where OD550 was 0.48.<br><br> | |
| - | ''Notes:'' | + | ''Results:'' Competent cells were made according to protocol. Apparently ''E. coli'' MG1655 reaches OD550 0.5 at least an hour faster than ''E. coli'' TOP10.<br><br> |
| - | + | '''Experiment:''' Transforming ''E.coli'' MG1655 strain with E0040 (GFP coding sequence) and I0500 (pBad arabinose inducible promoter) | |
| - | ''Results:'' Competent cells were made according to protocol. Apparently MG1655 | + | |
| - | '' | + | |
| - | '''Experiment:''' Transforming MG1655 strain with | + | |
<br> | <br> | ||
| - | Date: | + | Date: July 13th<br><br> |
''Notes:'' | ''Notes:'' | ||
| - | I0500 is located in pSB2K3. E0040 is in pSB1A2. | + | I0500 is located in pSB2K3. E0040 is in pSB1A2. 200 microlitres cells were added to each tube. Positive control is RFP generator as usual. Negative control is just cells.<br><br> |
| - | Only | + | Only 1 microliter DNA material was transferred per tube, since it was taken from the iGEM 2010 distribution plates.<br><br> |
Plates were dried at 42°C for 15 minutes, just prior to plating. Upon plating we discovered a problem with many of the ampicilin plates we made a couple of days ago. The agar breaks on the slightest contact, and are therfore impossible to use.<br><br> | Plates were dried at 42°C for 15 minutes, just prior to plating. Upon plating we discovered a problem with many of the ampicilin plates we made a couple of days ago. The agar breaks on the slightest contact, and are therfore impossible to use.<br><br> | ||
| - | As a result of the defective agar plates, most of our cells carrying | + | As a result of the defective agar plates, most of our cells carrying E0040 were ruined. We managed to save and plate out two batches, one from each tube, so we are hoping for the best tomorrow.<br><br> |
| - | Results: Only cells with | + | Results: Only cells with E0040 grew colonies by overnight incubation. |
| - | === Miniprep of [http://partsregistry.org/Part:BBa_K098995 | + | === Miniprep of [http://partsregistry.org/Part:BBa_K098995 K098995] in pSB1A2 === |
| - | Start date: | + | Start date: July 13th <br> |
''Methods:'' Fermentas GeneJET plasmid miniprep kit <br><br> | ''Methods:'' Fermentas GeneJET plasmid miniprep kit <br><br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ||
''Experiment done by:'' LC <br><br> | ''Experiment done by:'' LC <br><br> | ||
| - | ''Notes:'' Centrifuged the | + | ''Notes:'' Centrifuged the overnight culture for 15 mins. at 3000 rpm. Used 10 ml of culture in each tube and added 500 microlitres resuspension solution, then split it into two samples. <br><br> |
''Results:''<br> | ''Results:''<br> | ||
Results were as expected (correct), the plasmid was around 3000 bp long on the gel. <br><br> | Results were as expected (correct), the plasmid was around 3000 bp long on the gel. <br><br> | ||
''Analysis: ''All four samples turned out fine, so they were pooled and frozen. (See results for details) | ''Analysis: ''All four samples turned out fine, so they were pooled and frozen. (See results for details) | ||
<br> | <br> | ||
| - | Nr. 28 in the freezer = | + | Nr. 28 in the freezer = K098995<br><br> |
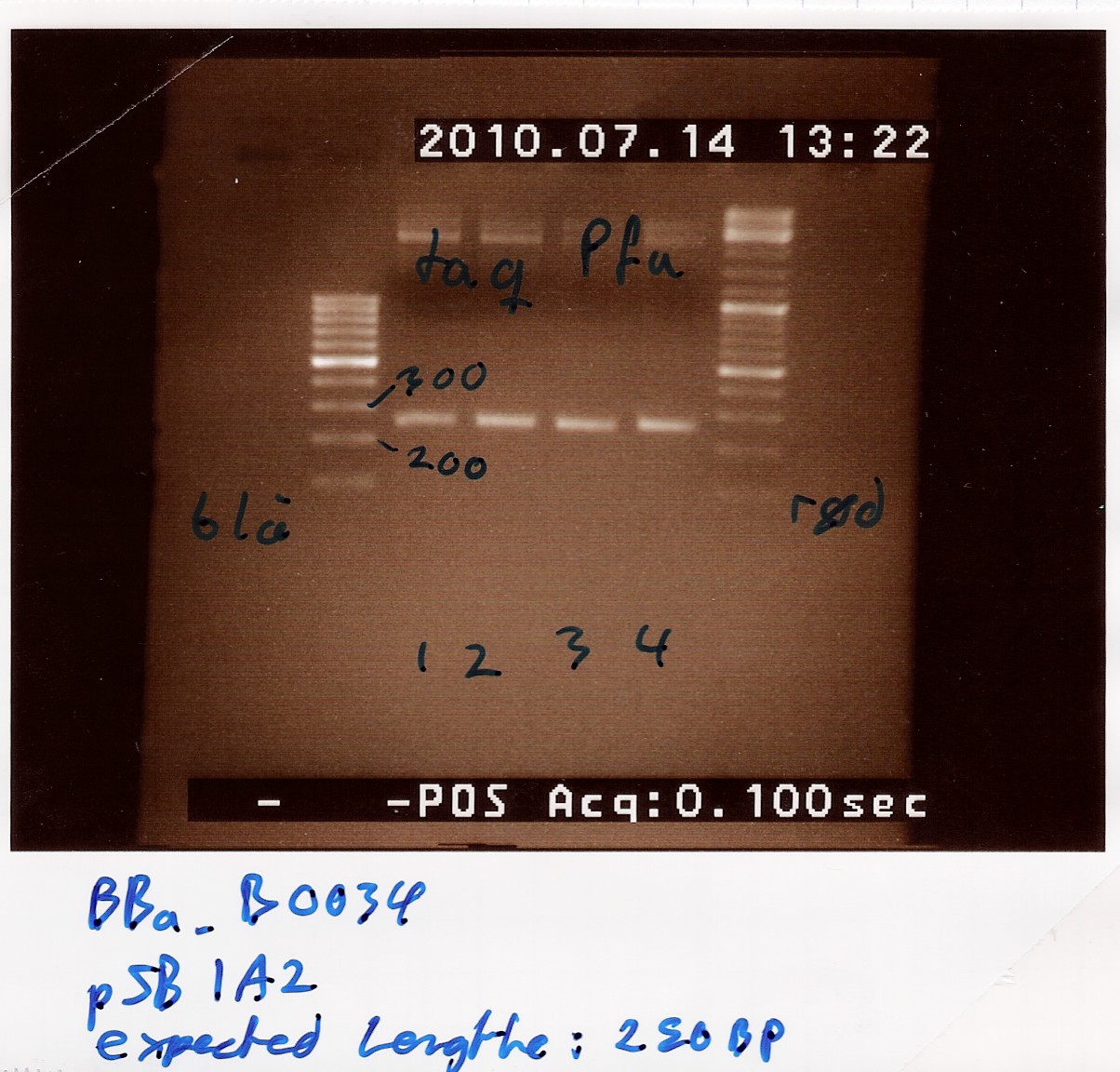
| - | === Extraction of [http://partsregistry.org/Part:BBa_B0034 | + | === Extraction of [http://partsregistry.org/Part:BBa_B0034 B0034] from pSB1A2 plasmid using PFU === |
==== Preliminary PCR to amplify VF2-VR piece containing part and restriction sites ==== | ==== Preliminary PCR to amplify VF2-VR piece containing part and restriction sites ==== | ||
| - | Date: | + | Date: July 14th <br> |
Methods: PCR of DNA in solutions<br> | Methods: PCR of DNA in solutions<br> | ||
Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1-PFU protocol]<br><br> | Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1-PFU protocol]<br><br> | ||
Experiment done by: Christian | Experiment done by: Christian | ||
<br><br> | <br><br> | ||
| - | Notes: DNA material from tube 27 was taken from the freezer. Primers were standard VF2 | + | Notes: DNA material from tube 27 was taken from the freezer. Primers were standard VF2 and VR of which we used 10 micromolars. PFU buffer with MgSO4 was used. No further MgSO4 was added. A TAQ control was done in a parallel experiment.<br><br> |
| - | Protocol was followed to point, with the | + | Protocol was followed to point, with the exception of 1 microliter additional H2O being added because of an error. It will yield 51 microlitres in each pcr reaction.<br><br> |
| - | Program was run according to protocol. | + | Program was run according to protocol. Elongation time was set to 1 minute and 30 seconds.<br><br> |
A control on the same DNA was run using TAQ polymerase. It was run on the same protocol and program as a parallel experiment in the flagella group: [https://2010.igem.org/Team:SDU-Denmark/labnotes#Amplification_of_pSB3K3_with_Taq Amplification of pSB3K3 with taq] | A control on the same DNA was run using TAQ polymerase. It was run on the same protocol and program as a parallel experiment in the flagella group: [https://2010.igem.org/Team:SDU-Denmark/labnotes#Amplification_of_pSB3K3_with_Taq Amplification of pSB3K3 with taq] | ||
<br><br> | <br><br> | ||
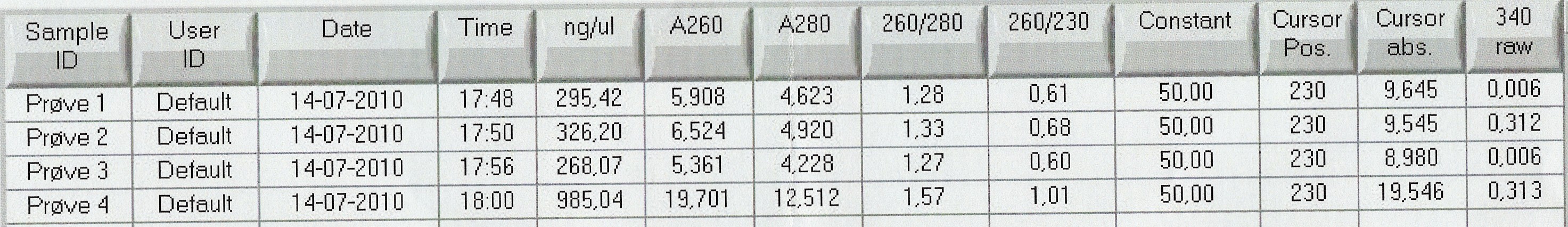
| - | Results: Bands showed in all four wells on the gel, located at the expected length of | + | Results: Bands showed in all four wells on the gel, located at the expected length of 250 bp. Nanodrop showed some contamination, but a good DNA concentration of 345ng/uL. The contamination might be the entire plasmid, since elongation time was a little in the high end.<br> |
[[Image:Team-SDU-Denmark-BBa_B0034-pSB1A2-PFU.jpg |300px]]<br><br> | [[Image:Team-SDU-Denmark-BBa_B0034-pSB1A2-PFU.jpg |300px]]<br><br> | ||
Analysis: The PCR product might be to contaminated still, and there's too little anyway. Therefore The PCR product will need to be further amplified.<br><br> | Analysis: The PCR product might be to contaminated still, and there's too little anyway. Therefore The PCR product will need to be further amplified.<br><br> | ||
==== Further amplification of PCR product ==== | ==== Further amplification of PCR product ==== | ||
| - | Date: | + | Date: July 14th<br> |
Methods: PCR of DNA in solutions<br> | Methods: PCR of DNA in solutions<br> | ||
Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1-PFU protocol]<br><br> | Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1-PFU protocol]<br><br> | ||
Experiment done by: Christian | Experiment done by: Christian | ||
<br><br> | <br><br> | ||
| - | Notes: 4 tubes were loaded, using one | + | Notes: 4 tubes were loaded, using one microliter of DNA template from the above experiment. Protocol was followed to point.<br><br> |
The same PCR program was used (PF1 on machine 2), elongation time once again set to 1 minute and 30 seconds.<br><br> | The same PCR program was used (PF1 on machine 2), elongation time once again set to 1 minute and 30 seconds.<br><br> | ||
Remaining PCR product was frozen as (White-2).<br><br> | Remaining PCR product was frozen as (White-2).<br><br> | ||
| - | + | A 1% agarose gel was loaded with 5 microlitres samples of each tube and GeneRuler 100bp ladder from Fermentas as marker. | |
<br><br> | <br><br> | ||
| - | Results: Bands showed at | + | Results: Bands showed at 250bp for all tubes. Tubes 1-3 showed a strange band that was larger than the ladder could show. Tube 4 was free of this band, and also showed a clearer band at 250bp.<br> |
[[Image:Team-SDU-Denmark-BBa_B0034-pSB1A2-PFU-run2.jpg |300px]]<br><br> | [[Image:Team-SDU-Denmark-BBa_B0034-pSB1A2-PFU-run2.jpg |300px]]<br><br> | ||
Nanodrop showed signatures for tubes 1-3 like in the samples of the above experiment. DNA was twice as concentrated in tube 4.<br> | Nanodrop showed signatures for tubes 1-3 like in the samples of the above experiment. DNA was twice as concentrated in tube 4.<br> | ||
| Line 1,114: | Line 1,118: | ||
==== Gel Extraction of PCR product ==== | ==== Gel Extraction of PCR product ==== | ||
| - | Date: | + | Date: July 15th<br> |
Methods: Gel Extraction Kit<br> | Methods: Gel Extraction Kit<br> | ||
Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#DNA_extraction_from_gel_.28fermentas.29 DE1.1]<br> | Protocol: [https://2010.igem.org/Team:SDU-Denmark/protocols#DNA_extraction_from_gel_.28fermentas.29 DE1.1]<br> | ||
Experiment done by: Christian, Lars Christian | Experiment done by: Christian, Lars Christian | ||
<br><br> | <br><br> | ||
| - | Notes: | + | Notes: Four 50 microliter wells were created in the gel to accomodate all the genetic material, which was loaded along with GeneRuler 100bp ladder from Fermentas as marker. We extracted DNA from the 250bp position according to protocol.<br><br> |
| - | + | After dissolving the gel slice, we added 100% isopropanol to solution in 1:2 ratio, as noted in the kit protocol for DNA pieces less than 500bp.<br><br> | |
Lars Christian eluded twice with 30ul elution buffer instead of once with 50ul for 4 of the eight tubes. Otherwise protcol was followed.<br><br> | Lars Christian eluded twice with 30ul elution buffer instead of once with 50ul for 4 of the eight tubes. Otherwise protcol was followed.<br><br> | ||
Extractions were pooled and frozen as (White 11) | Extractions were pooled and frozen as (White 11) | ||
<br><br> | <br><br> | ||
| - | Results: Nanodrop showed 5,00ng/ | + | Results: Nanodrop showed 5,00ng/uL DNA with 260/280 ratio at 2.29 and 260/230 ratio at 0.02. |
<br><br> | <br><br> | ||
| - | Analysis: This product is not optimal. We will need to decide | + | Analysis: This product is not optimal. We will need to decide whether to try again. |
<br><br> | <br><br> | ||
--[[User:CKurtzhals|CKurtzhals]] 16:07, 15 July 2010 (UTC) | --[[User:CKurtzhals|CKurtzhals]] 16:07, 15 July 2010 (UTC) | ||
| - | === Miniprep and transformation of [http://partsregistry.org/Part:BBa_K274210 | + | === Miniprep and transformation of [http://partsregistry.org/Part:BBa_K274210 K274210] in pSB1A2 === |
| - | Start date: 14 | + | Start date: July 14.th <br> |
''Methods:'' Fermentas GeneJET plasmid miniprep kit <br><br> | ''Methods:'' Fermentas GeneJET plasmid miniprep kit <br><br> | ||
''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ''Protocol'': [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ||
| Line 1,137: | Line 1,141: | ||
''Notes:'' No pellet after step 4, but still continued. Later we got a small pellet at step 6. <br><br> | ''Notes:'' No pellet after step 4, but still continued. Later we got a small pellet at step 6. <br><br> | ||
''Results:''<br> | ''Results:''<br> | ||
| - | The miniprep failed as expected, since there was no pellet after the second centrifugation. | + | The miniprep failed as expected, since there was no pellet after the second centrifugation.<br><br> |
''Analysis:''The experiment will be repeated in the afternoon.<br> | ''Analysis:''The experiment will be repeated in the afternoon.<br> | ||
| Line 1,143: | Line 1,147: | ||
Methods and protocol as in the first try.<br> | Methods and protocol as in the first try.<br> | ||
| - | ''Notes:'' We only had 3,5 ml of | + | ''Notes:'' We only had 3,5 ml of overnight culture for the miniprep, so we had to manage with that.<br> |
''Results:'' Insert gel picture and nanodrop table. <br> | ''Results:'' Insert gel picture and nanodrop table. <br> | ||
''Analysis:'' The second try worked, so we went on to make transformations with the plasmid. It is in the freezer as nr. 3 (white). | ''Analysis:'' The second try worked, so we went on to make transformations with the plasmid. It is in the freezer as nr. 3 (white). | ||
| Line 1,149: | Line 1,153: | ||
'''Transformation of the miniprepped plasmid:'''<br><br> | '''Transformation of the miniprepped plasmid:'''<br><br> | ||
''Experiment done by:'' LC, Maria <br><br> ''Protocol:'' TR 1.1 <br><br> | ''Experiment done by:'' LC, Maria <br><br> ''Protocol:'' TR 1.1 <br><br> | ||
| - | ''Notes:'' | + | ''Notes:'' Transformed K274210 in pSB1A2 on LA plates with 100 microgram/ml ampicillin into ''E. coli'' MG1655. We used 2 microliter from the miniprep product for each sample.<br> |
Our suspicion that the agar plates we made are faulty got confirmed, three more plates were destroyed while spreading the bacteria on them (Sample 1a, 2b, 2pellet and the positive control)<br><br> | Our suspicion that the agar plates we made are faulty got confirmed, three more plates were destroyed while spreading the bacteria on them (Sample 1a, 2b, 2pellet and the positive control)<br><br> | ||
| - | ''Results:'' Cells grew just fine and the negative control was as expected. After a night at 37° the cells seemed a little more yellowish | + | ''Results:'' Cells grew just fine and the negative control was as expected. After a night at 37° the cells seemed a little more yellowish to the eye, than the usual. <br><br> |
''Analysis:'' The transformations succeeded. <br><br> | ''Analysis:'' The transformations succeeded. <br><br> | ||
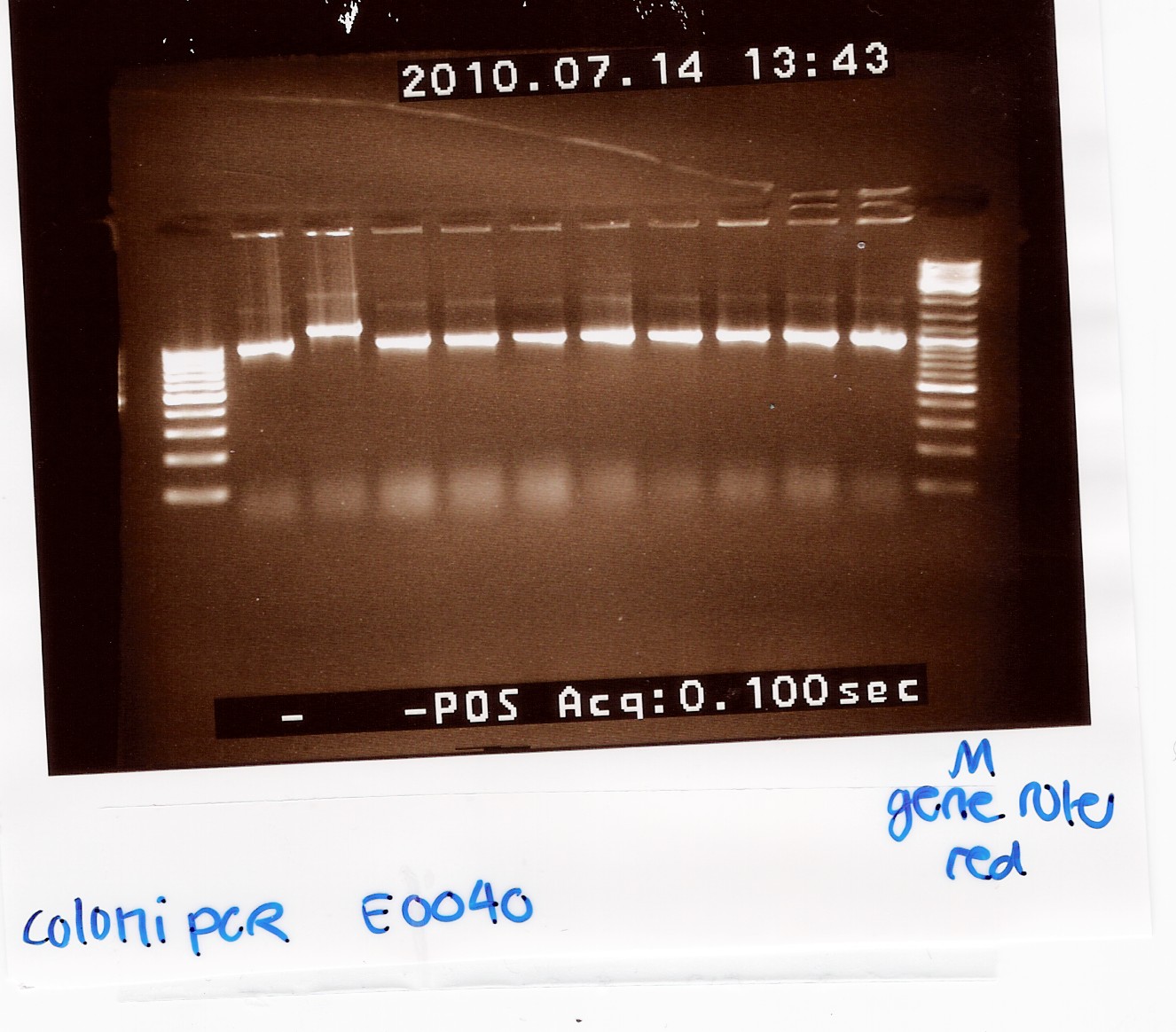
| - | === Coloni PCR of pSB1A2 w. [http://partsregistry.org/Part:BBa_E0040 | + | === Coloni PCR of pSB1A2 w. [http://partsregistry.org/Part:BBa_E0040 E0040] (transformation 13/7) === |
| - | Start date: | + | Start date: July 14th <br> |
| - | ''Methods:'' | + | ''Methods:'' Taq coloni PCR and gel electrophoresis <br><br> |
''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.2 CP1.2] <br><br> | ||
''Experiment done by:'' Maria <br><br> | ''Experiment done by:'' Maria <br><br> | ||
| - | ''Notes:''10 colonies from the [https://2010.igem.org/Team:SDU-Denmark/labnotes#Transformation_of_MG1655_e._coli_with_BBa_274210.2C_BBa_E0040_and_BBa_I0500 Transformation 13/7] by taq PCR using VF2 and VR | + | ''Notes:''10 colonies from the [https://2010.igem.org/Team:SDU-Denmark/labnotes#Transformation_of_MG1655_e._coli_with_BBa_274210.2C_BBa_E0040_and_BBa_I0500 Transformation 13/7] by taq PCR using 10 micromolar of VF2 and VR.<br> |
In PCR tube no. 6 most of the premix was lost. <br><br> | In PCR tube no. 6 most of the premix was lost. <br><br> | ||
| Line 1,170: | Line 1,174: | ||
--[[User:Tipi|Tipi]] 16:09, 19 July 2010 (UTC) | --[[User:Tipi|Tipi]] 16:09, 19 July 2010 (UTC) | ||
| - | === Transformation of [http://partsregistry.org/Part:BBa_K081005 | + | === Transformation of [http://partsregistry.org/Part:BBa_K081005 K081005] in pSB1A2 (constitutive promoter and RBS combined) and [http://partsregistry.org/Part:BBa_R0011 R0011] in pSB1A2 in Top 10 E.Coli === |
| - | Start date: | + | Start date: July 15th - 16th <br> |
| - | ''Methods:'' | + | ''Methods:'' Overnight culture, making competent cells, transformation <br><br> |
''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1] <br><br> | ''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1] <br><br> | ||
''Experiment done by:'' LC <br><br> | ''Experiment done by:'' LC <br><br> | ||
| - | ''Notes:'' | + | ''Notes:''Overnight culture was made of 110 ml LB medium and a frozen top 10 culture. The cells took quite a while to reach an OD550 > 0,5, but it was consistent with our prior experience with ''E.coli'' TOP10: |
<html> | <html> | ||
<table style="text-align: left; width: 100px;" border="1" | <table style="text-align: left; width: 100px;" border="1" | ||
| Line 1,229: | Line 1,233: | ||
''Results:''Both transformations failed, even though positive and negative control were correct. | ''Results:''Both transformations failed, even though positive and negative control were correct. | ||
<br><br> | <br><br> | ||
| - | ''Analysis:''We are unsure as | + | ''Analysis:''We are unsure as to the reason of the experiment failing, on monday we will try to transform the same brick on different plates to check if it is in the right plasmid ([https://2010.igem.org/Team:SDU-Denmark/labnotes2#Transformation_of_K081005_in_pSB1A2_.28constitutive_promoter_and_RBS_combined.29.2CR0011_in_pSB1A2.2C_pSB3C5_w._J04450_and_pSB3T5_w._J04450_in_Top_10_E.Coli transformation]).<br> |
--[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC)<br><br> | --[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC)<br><br> | ||
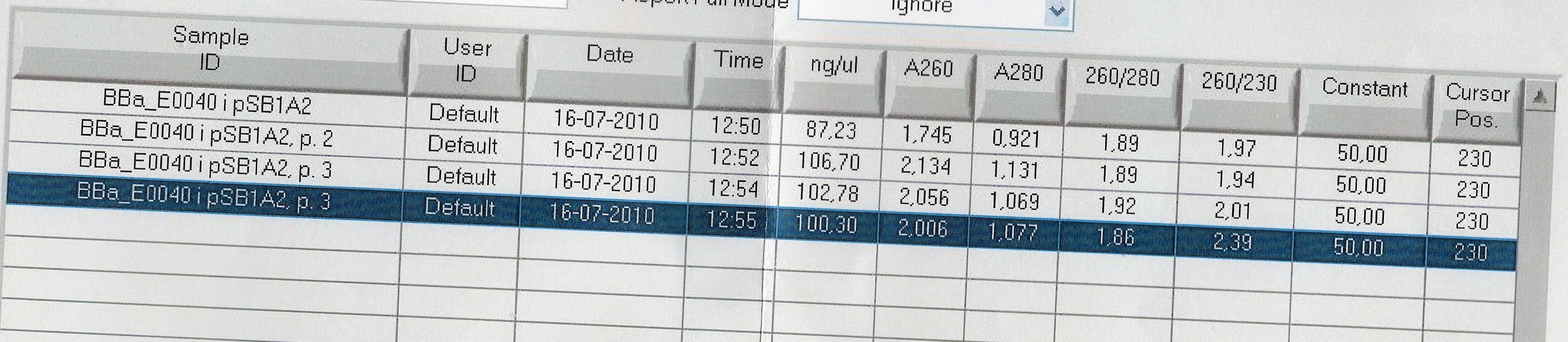
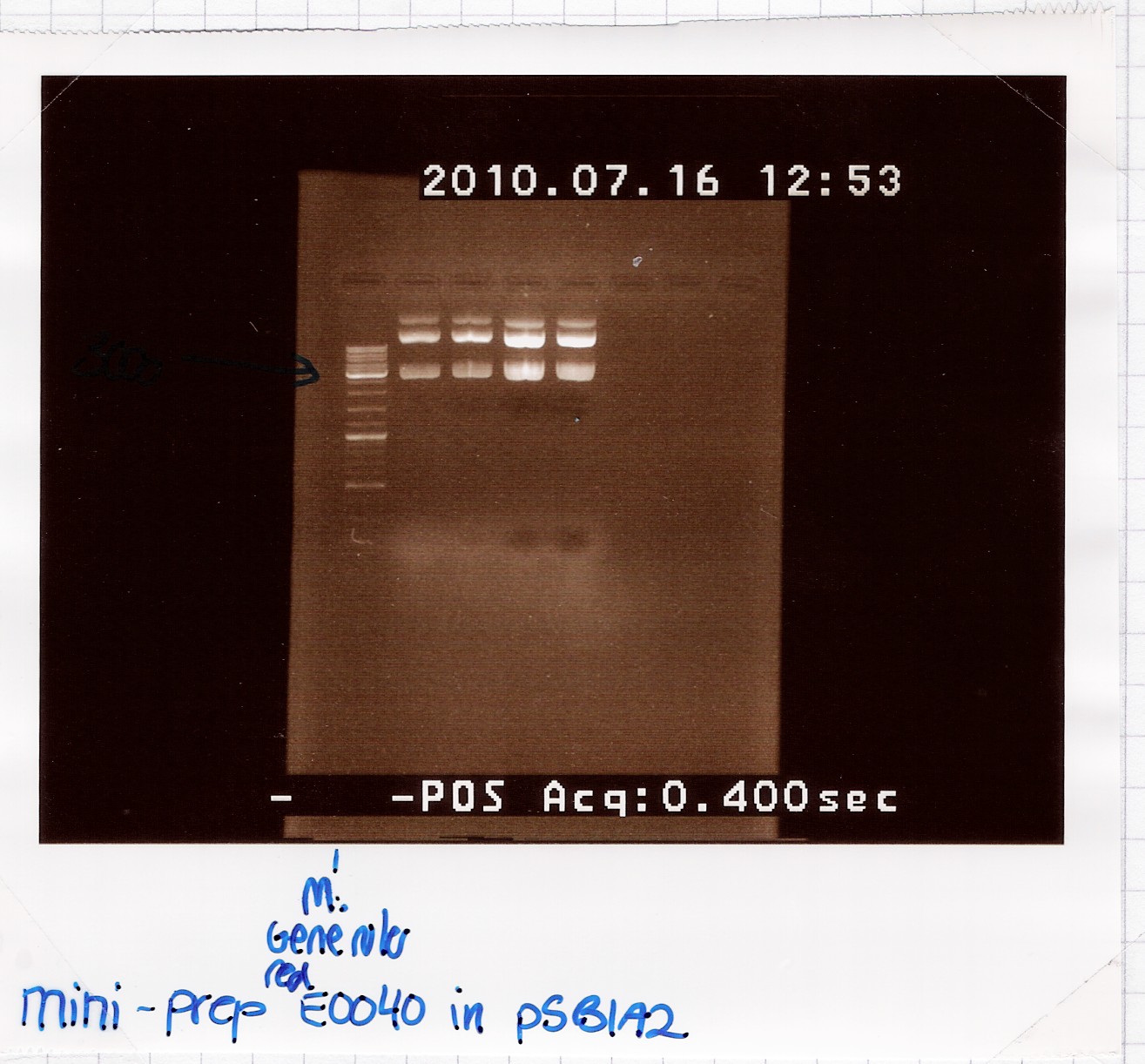
=== Miniprep of [http://partsregistry.org/Part:BBa_E0040 BBa_E0040] === | === Miniprep of [http://partsregistry.org/Part:BBa_E0040 BBa_E0040] === | ||
| - | Start date: | + | Start date: July 16th<br> |
| - | ''Methods:'' | + | ''Methods:'' Overnight culture, miniprep <br><br> |
''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#MP1.1 MP1.1] <br><br> | ||
''Experiment done by:'' LC <br><br> | ''Experiment done by:'' LC <br><br> | ||
| Line 1,245: | Line 1,249: | ||
--[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC) | --[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC) | ||
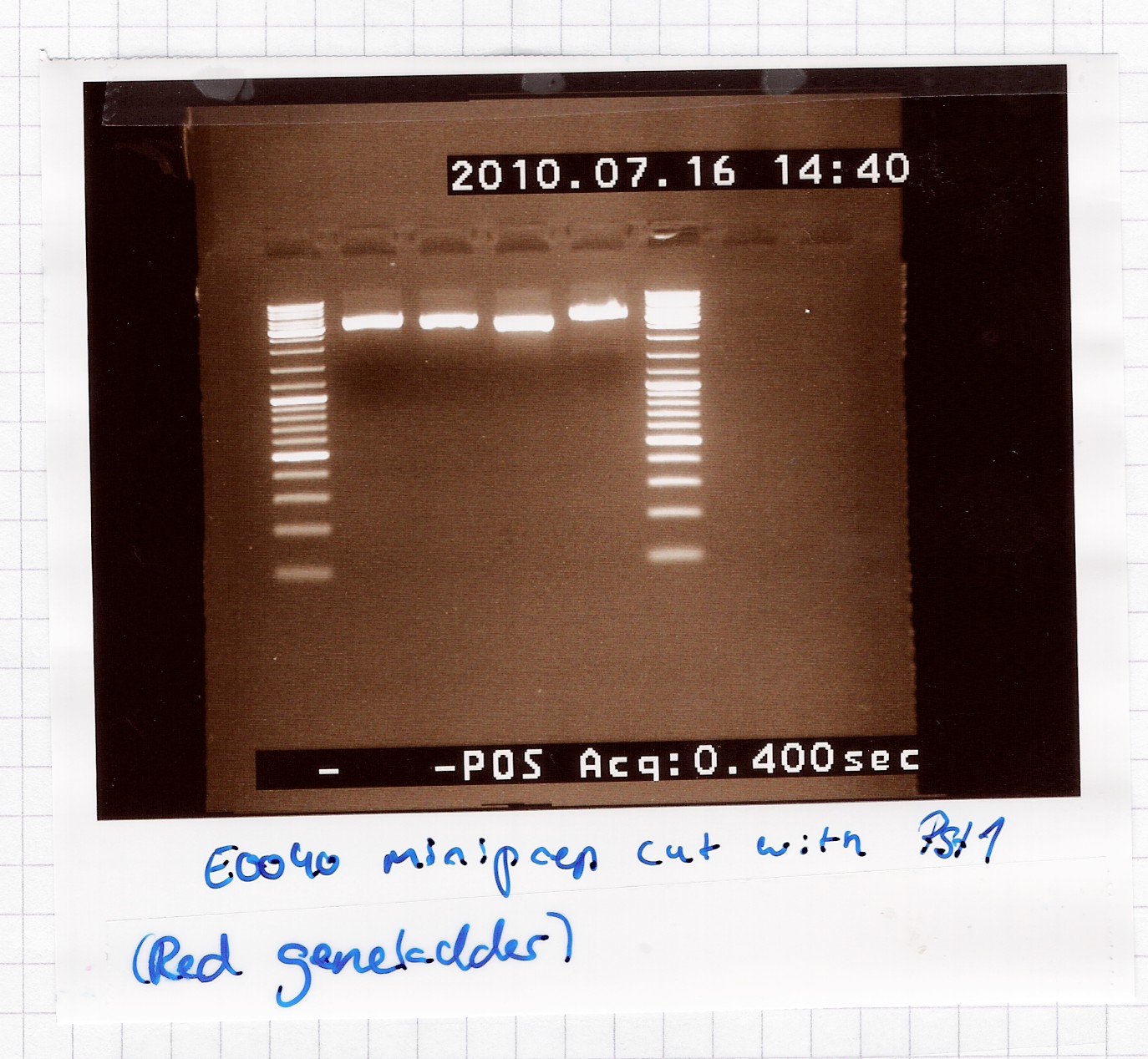
| - | ==== Restriction digest of [http://partsregistry.org/Part:BBa_E0040 | + | ==== Restriction digest of [http://partsregistry.org/Part:BBa_E0040 E0040] miniprep product ==== |
| - | Start date: | + | Start date: July 16th<br> |
''Methods:'' Restriction digest <br><br> | ''Methods:'' Restriction digest <br><br> | ||
''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1] <br><br> | ''Protocol:'' [https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1] <br><br> | ||
| Line 1,255: | Line 1,259: | ||
<br> | <br> | ||
[[Image:Team-SDU-Denmark-rd1607.jpg|300px]]<br> | [[Image:Team-SDU-Denmark-rd1607.jpg|300px]]<br> | ||
| - | A single band on the gel, which | + | A single band is seen on the gel, which means that the multiple bands in the miniprep gel were only because of supercoiling. |
<br><br> | <br><br> | ||
''Analysis:'' Sample 2 and 3 got pooled and frozen, 1 and 4 were discarded, since their length didn't seem exactly right.<br><br> | ''Analysis:'' Sample 2 and 3 got pooled and frozen, 1 and 4 were discarded, since their length didn't seem exactly right.<br><br> | ||
| - | Nr. 12 in the freezer = | + | Nr. 12 in the freezer = E0040 miniprep product in pSB1A2.<br> |
--[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC) | --[[User:Lclund|Lclund]] 10:00, 19 July 2010 (UTC) | ||
Latest revision as of 19:16, 24 October 2010
 "
"