Team:Heidelberg/Notebook/Material Methods
From 2010.igem.org
(→Buffers) |
Laura Nadine (Talk | contribs) (→Microscopy) |
||
| Line 162: | Line 162: | ||
Single images were obtained using the Leica TCS SP5 confocal microscope and camera with the Leica AF6000 imaging software. GFP fluorescence was excited by Argon 488nm laser and measured at 520-560nm, BFP fluorescence was excited by UV laser at 405nm and measured at 440-460nm. Pictures were taken sequentially line by line in three different channels for GFP, BFP and bright field. | Single images were obtained using the Leica TCS SP5 confocal microscope and camera with the Leica AF6000 imaging software. GFP fluorescence was excited by Argon 488nm laser and measured at 520-560nm, BFP fluorescence was excited by UV laser at 405nm and measured at 440-460nm. Pictures were taken sequentially line by line in three different channels for GFP, BFP and bright field. | ||
| + | |||
| + | |||
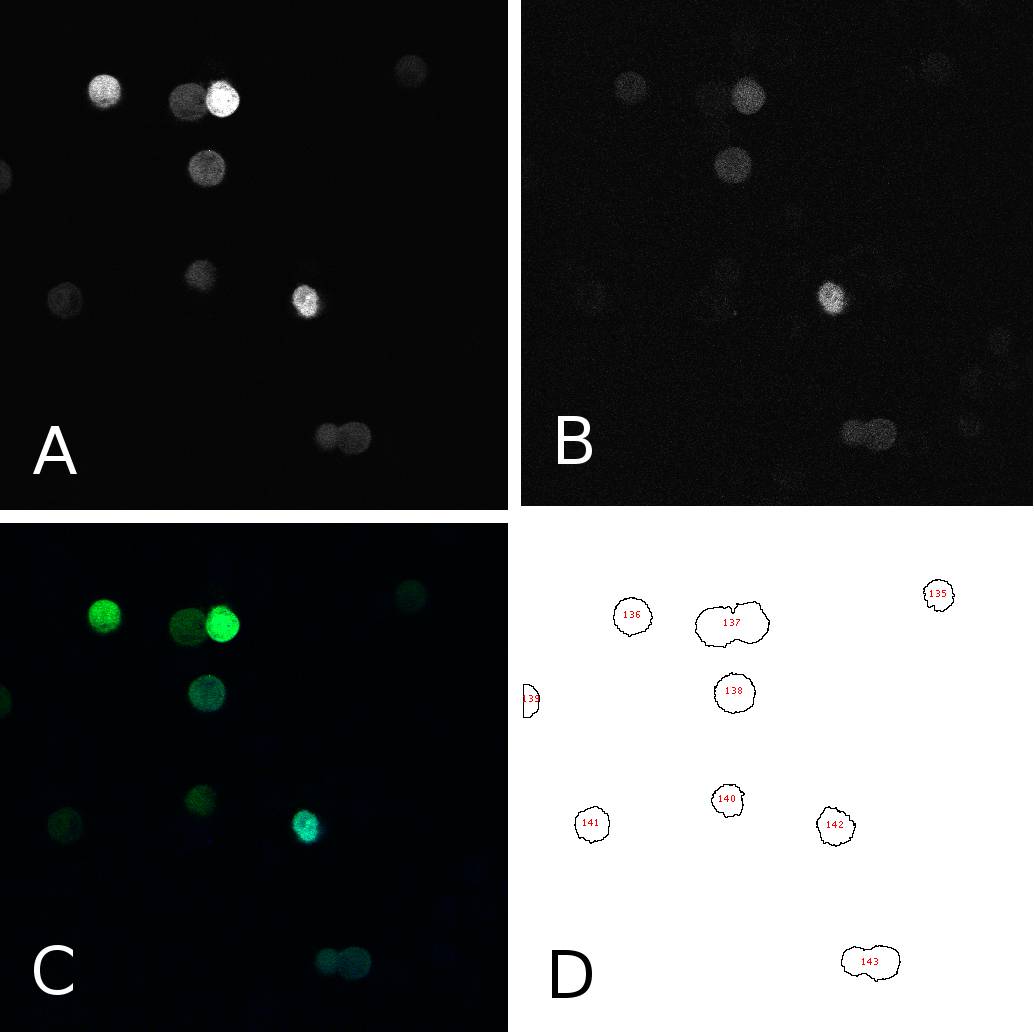
| + | [[Image:panel.jpg|thumb|500px|center|'''HeLa cells two days after transfection with miMeasure''' (A) fluorescence signal GFP channel, 8bit; (B) fluorescence signal BFP channel, 8bit; (C) merge of channels A and B, RGB (D) cells after segmentation and automated cell counting and annotation]] | ||
To analyze the fluorescence of single cells, we segmented the images using ImageJ. In 8bit pictures, we set the threshold for each channel to 50, thereby filtering the background. This allows us to annotate cells automatically using the “analyze particles” tool. We could now get the fluorescence intensity for each single cell on each channel (GFP or BFP) as an 8bit output, i.e. a value between 50 and 255. Panel 1 shows an example of one such image in different channels and after segmentation. From the data thus obtained, we calculated the GFP:BFP ratios for each cell using a simple algorithm. We could then visualize the mean of these rations in a bar plot or use all the data to calculate a linear regression curve. | To analyze the fluorescence of single cells, we segmented the images using ImageJ. In 8bit pictures, we set the threshold for each channel to 50, thereby filtering the background. This allows us to annotate cells automatically using the “analyze particles” tool. We could now get the fluorescence intensity for each single cell on each channel (GFP or BFP) as an 8bit output, i.e. a value between 50 and 255. Panel 1 shows an example of one such image in different channels and after segmentation. From the data thus obtained, we calculated the GFP:BFP ratios for each cell using a simple algorithm. We could then visualize the mean of these rations in a bar plot or use all the data to calculate a linear regression curve. | ||
Revision as of 13:32, 24 October 2010

|
|
|||||||||||||||||||||||||||||||||
 "
"