Team:SDU-Denmark/labnotes7
From 2010.igem.org
(Difference between revisions)
(New page: {{:Team:SDU-Denmark/css2}} {{:Team:SDU-Denmark/navi2}} <div id="subnavi"> <div id="leftcolumn"> = Lab notes (16/9 - 22/9) = __TOC__) |
(→Restriction Digest) |
||
| (14 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
<div id="leftcolumn"> | <div id="leftcolumn"> | ||
| - | = Lab notes ( | + | = Lab notes (23/8 - 29/8) = |
__TOC__ | __TOC__ | ||
| + | |||
| + | == Photosensor == | ||
| + | === Insertion of B0015 in pSB3C5 and pSB3T5 === | ||
| + | |||
| + | '''Date:''' 8/23 - 8/29 2010<Br> | ||
| + | '''Done By:''' Maria and Lc<Br> | ||
| + | '''Protocol:''' [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#DE1.3 DE1.3][https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#LG1.2 LG1.2][https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.3 CP1.3]<Br> | ||
| + | ==== Pfu PCR amplification and purification of B0015 ==== | ||
| + | '''Date:''' 8/23 2010<Br> | ||
| + | '''Done By:''' Maria and Lc<Br> | ||
| + | '''Protocol:'''[https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1]<br> | ||
| + | '''Notes:'''<br> | ||
| + | 4 PCR reactions are prepared. 2uL Miniprep of pSB1AK3 (white 43) are distrubuted in each tube. PCR tubes are marked B00153.A-D.<br> | ||
| + | Premix x5:<br> | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>pfu buffer + MgSO<small>4</small></td> | ||
| + | <td>25uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>dNTP mix</td> | ||
| + | <td>7.5uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>VF2 primer</td> | ||
| + | <td>7.5uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>VR primer</td> | ||
| + | <td>7.5uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>H<small>2</small>0</td> | ||
| + | <td>190uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>pfu polymerase </td> | ||
| + | <td>2uL</td> | ||
| + | </tr> | ||
| + | </table><br><br> | ||
| + | PCR program:<br> | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>start</td> | ||
| + | <td>94C</td> | ||
| + | <td>3min</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>denaturating</td> | ||
| + | <td>94C</td> | ||
| + | <td>2min</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>annealing</td> | ||
| + | <td>55C</td> | ||
| + | <td>30s</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>elongation</td> | ||
| + | <td>72C</td> | ||
| + | <td>30s</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>go to</td> | ||
| + | <td>2</td> | ||
| + | <td>rep.29x</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>end</td> | ||
| + | <td>72C</td> | ||
| + | <td>5min</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>hold</td> | ||
| + | <td>4C</td> | ||
| + | <td></td> | ||
| + | </tr> | ||
| + | </table><br> | ||
| + | 5uL of the PCR product was loaded onto a 2% agarose gel. Gene ruler 100bp DNA ladder was used ad marker.<br><br> | ||
| + | '''Results:'''<br> | ||
| + | '''Analysis:'''<br> | ||
| + | A strong band was observed at app. 450bp, and the rest of the PCR product was purified according to protocol using the GFX purification kit.<br> | ||
| + | End conc.: 48ng/uL <br><br> | ||
| + | |||
| + | ==== Restriction digest of B0015 ==== | ||
| + | '''Date:''' 8/24 2010<Br> | ||
| + | '''Done By:''' Maria and Lc<Br> | ||
| + | '''Protocol:'''[https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#DE1.3 DE1.3]<br> | ||
| + | '''Notes:'''<br> | ||
| + | Restriction mixture B0015:<br> | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>H<small>2</small>O</td> | ||
| + | <td>38uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>FD green buffer</td> | ||
| + | <td>8uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>EcoRI</td> | ||
| + | <td>4uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>PstI</td> | ||
| + | <td>4uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>B0015</td> | ||
| + | <td>30uL</td> | ||
| + | </tr> | ||
| + | </table><br><br> | ||
| + | |||
| + | The digested sample was loaded onto a 2% agarose extraction gel. Uncut B0015 was used as controle. Gene ruler 100bp DNA ladder was used as marker.<br> | ||
| + | DNA was extracted from gel according to protocol.<br><br> | ||
| + | '''Results:'''<br> | ||
| + | DNA conc: | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>sample</td> | ||
| + | <td>conc. (ng/uL)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>B0015</td> | ||
| + | <td>6.5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>pSB3C5</td> | ||
| + | <td>50.8</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>pSB3T5</td> | ||
| + | <td>9.9</td> | ||
| + | </tr> | ||
| + | </table><br><br> | ||
| + | '''Analysis:''' | ||
| + | the purified DNA was used for ligation.<br><br> | ||
| + | |||
| + | ==== Ligation of PS and pSB1C3 and pCB1AK3 ==== | ||
| + | '''Date:''' 8/24 2010<Br> | ||
| + | '''Done By:''' Maria and Lc<Br> | ||
| + | '''Protocol:'''[https://2010.igem.org/Team:SDU-Denmark/protocols#LG1.2 LG1.2]<br> | ||
| + | '''Notes:'''<br> | ||
| + | For each of the ligations three ligation mixtures were prepared. vector concentrations of 10n0g/uL (pSB3C5) and 50ng/uL (pSB3T5) respectively was used for each mixture. Appropiate amount of insert was added to reach vector:insert ratios of 1:1, 1:3 and 1:6 respectively. <br> | ||
| + | Ligation mixtures (B0015 in pSB3C5):<br> | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td></td> | ||
| + | <td>L1</td> | ||
| + | <td>L2</td> | ||
| + | <td>L3</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>T4 ligase buffer</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>T4 ligase</td> | ||
| + | <td>1uL</td> | ||
| + | <td>1uL</td> | ||
| + | <td>1uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>pSB3C5</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>B0015 </td> | ||
| + | <td>0.7uL</td> | ||
| + | <td>2uL</td> | ||
| + | <td>4.5uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>H<small>2</small>0</td> | ||
| + | <td>14.3uL</td> | ||
| + | <td>13uL</td> | ||
| + | <td>10.5uL</td> | ||
| + | </tr> | ||
| + | </table><br> | ||
| + | Ligation mixtures (B0015 in pSB3T5):<br> | ||
| + | <table style="text-align: left;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td></td> | ||
| + | <td>L1</td> | ||
| + | <td>L2</td> | ||
| + | <td>L3</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>T4 ligase buffer</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | <td>2uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>T4 ligase</td> | ||
| + | <td>1uL</td> | ||
| + | <td>1uL</td> | ||
| + | <td>1uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>pSB3T5</td> | ||
| + | <td>5uL</td> | ||
| + | <td>5uL</td> | ||
| + | <td>5uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>B0015 </td> | ||
| + | <td>0.5uL</td> | ||
| + | <td>1uL</td> | ||
| + | <td>2uL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>H<small>2</small>0</td> | ||
| + | <td>11.5uL</td> | ||
| + | <td>11uL</td> | ||
| + | <td>10uL</td> | ||
| + | </tr> | ||
| + | </table><br><br> | ||
| + | The samples was incubated at 17C ON at used for transformation<br><br> | ||
| + | |||
| + | ==== Transfomation of ligated plasmid in Top 10 E.coli ==== | ||
| + | |||
| + | '''Date:''' 8/25 2010<Br> | ||
| + | '''Done By:''' Maria and Lc<Br> | ||
| + | '''Protocol:'''[https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1][https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1]<br> | ||
| + | '''Notes:'''<br> | ||
| + | The compotent cells and transformation was carried out according to protocol.<br><br> | ||
| + | '''Results:'''<br> | ||
| + | the controle plates were okay, and there were many colonies on plates with cells transformed with either of the ligation mixtures..<br><br> | ||
| + | '''Analysis:'''<br> | ||
| + | The transformation was successfull and colonies was selected and used in coloni PCR.<br> | ||
| + | --[[User:Tipi|Tipi]] 13:41, 26 September 2010 (UTC)<br><br> | ||
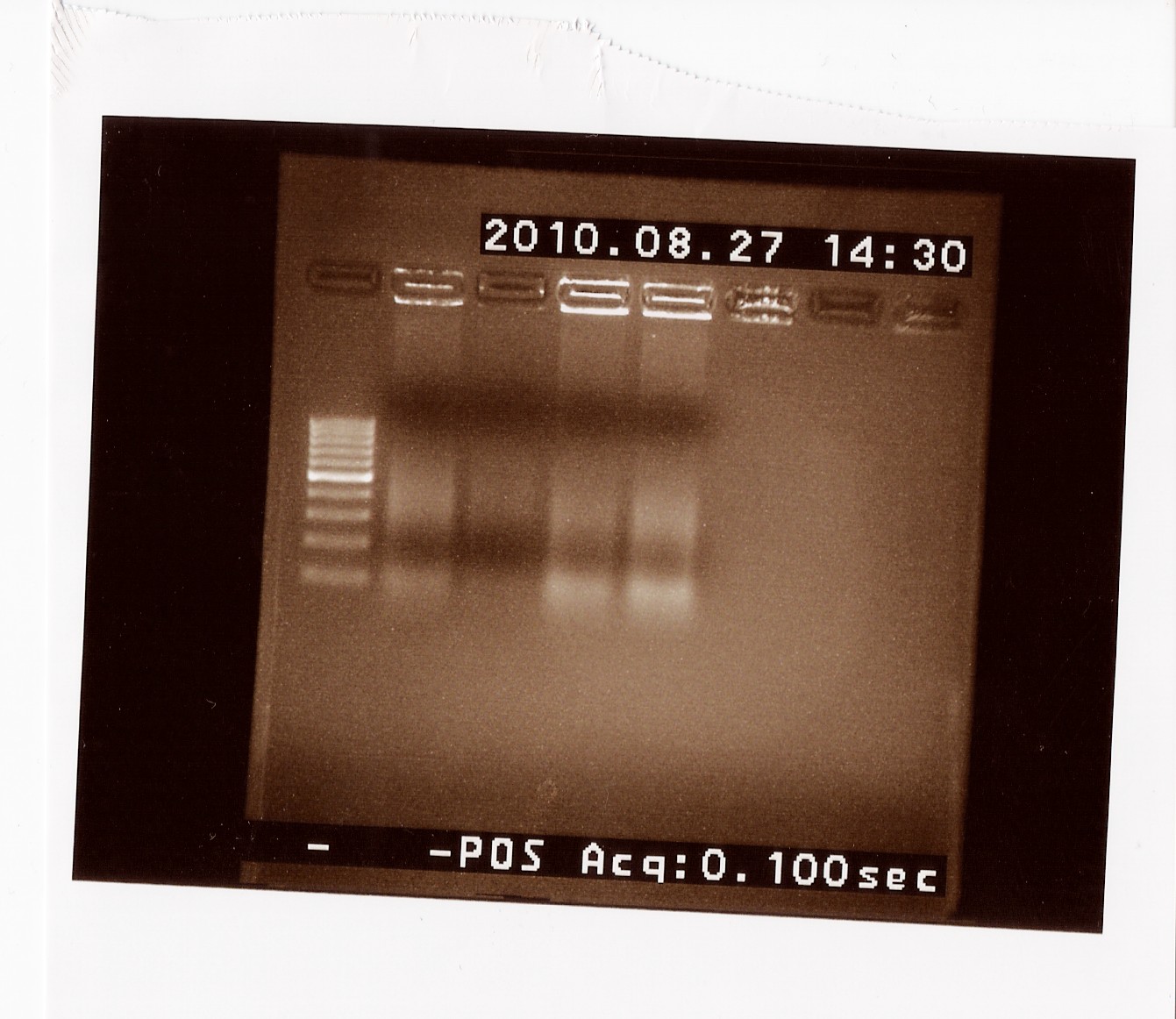
| + | === Colony PCR on B0017 === | ||
| + | Date: 27/8<br> | ||
| + | Done by: LC<br> | ||
| + | Methods: PCR<br> | ||
| + | Protocols: [https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.3 CP1.3]<br> | ||
| + | Notes: <br> | ||
| + | Premix:<br> | ||
| + | 12,5 µl 10xTAQ Buffer<br> | ||
| + | 5 µl MgCl2 <br> | ||
| + | 5 µl VF2 <br> | ||
| + | 5 µl VR <br> | ||
| + | 2,5 µl dNTP<br> | ||
| + | 17,5 µl H2O<br> | ||
| + | 5/8 µl TAQ Polymerase<br> | ||
| + | <br> | ||
| + | 9,5 µl Premix were added to 15 µl of H2O containing the lysed cells.<br> | ||
| + | <br> | ||
| + | PCR Program:<br> | ||
| + | <table style="text-align: left;" border="1" cellpadding="2" | ||
| + | cellspacing="2"> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Start<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">94 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">2 min<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Denaturing<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">94 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">1 min<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Annealing<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">55 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">1 min<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Elongation<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">72 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">30 sec<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Goto2<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">rep<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">29x<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">End<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">72 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">3 min<br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="vertical-align: top;">Hold<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;">4 C<br> | ||
| + | </td> | ||
| + | <td style="vertical-align: top;"><br> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | <br> | ||
| + | <br> | ||
| + | Results: <br> | ||
| + | [[Image:Team-SDU-Denmark-cPCRB0017.jpg|300px]] <br> | ||
| + | No useable results, only unclear bands around 120 BP, which seem to be the result of mispriming with VR on B0010. | ||
| + | |||
| + | == Retinal == | ||
| + | |||
| + | ==== Futher PCR on POT2 with NinaB (New Primers NO. 5) ==== | ||
| + | Date: 23/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: PCR<br> | ||
| + | protocos:[https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1] | ||
| + | Notes:<br> | ||
| + | NinB2fw and NinaB2rv was used.<br> | ||
| + | PCR were run programed as:<br> | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <table style="text-align: left; width: 300px;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>PCR</td> | ||
| + | <td>Temp. (C)</td> | ||
| + | <td>Time (min)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Start</td> | ||
| + | <td>95</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Denaturing</td> | ||
| + | <td>95</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Anneling</td> | ||
| + | <td>67,9</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Elongation</td> | ||
| + | <td>72</td> | ||
| + | <td>4</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>End</td> | ||
| + | <td>72</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Hold</td> | ||
| + | <td>4</td> | ||
| + | <td>indef.</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | <br> | ||
| + | </body> | ||
| + | </html> | ||
| + | <br> | ||
| + | PCR product from gradient PCR (d. 20/8-10), tube no. 5, was used as template.<br> | ||
| + | The other tubes were pooled.<br> | ||
| + | |||
| + | === PCR on POT2 with NinaB (New Primers) === | ||
| + | Start date: 24/8<br> | ||
| + | Methods: Ligation, Competent cells, Transformation<br> | ||
| + | Protocols: [https://2010.igem.org/Team:SDU-Denmark/protocols#LG1.1 LG1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#CC1.1 CC1.1], [https://2010.igem.org/Team:SDU-Denmark/protocols#TR1.1 TR1.1] | ||
| + | |||
| + | ==== DNA purification from PCR ==== | ||
| + | Date: 20/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: DNA purification from PCR<br> | ||
| + | protocos:GFX purification from PCR - kit | ||
| + | <br><br> | ||
| + | One of the pooled tubes was eluted in 200µL, the others was eluted in 20µL<br> | ||
| + | 200µL nanodrop: 16,4 ng/µL<br> | ||
| + | 20µL nanodrop: 133,9ng/µL<br> | ||
| + | |||
| + | ==== Restriction Digest ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: Restriction Digest<br> | ||
| + | protocos:RD1.1[https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1] | ||
| + | Notes:<br> | ||
| + | Restriction mixture:<br> | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <table style="text-align: left; width: 100px;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>38 µL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>8 µL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>4 µL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>4 µL</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>30 µL</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | <br> | ||
| + | </body> | ||
| + | </html> | ||
| + | Gel was run with uncut controles:<br> | ||
| + | |||
| + | ==== Gel purification ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: gel purifikation<br> | ||
| + | protocos:GFX gel purifikation kit | ||
| + | Notes:<br> | ||
| + | Purifide products was Nanodroped:<br> | ||
| + | NinaB 1: 4,5 ng/µL<br> | ||
| + | NinaB 2: 1,15 ng/µL<br> | ||
| + | NinaB 3: 7,74 ng/µL<br> | ||
| + | PSB1C3: 25,66 ng/µL<br> | ||
| + | Nina B pooled: 4,5 ng/µL<br> | ||
| + | |||
| + | ==== Ligation ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: Ligation<br> | ||
| + | protocos:[https://2010.igem.org/Team:SDU-Denmark/protocols#LG1.3 L1.3] | ||
| + | Notes:<br> | ||
| + | 3 ligatons mixtures was made:<br> | ||
| + | 1:1 volumens 1 plasmid:5 insert | ||
| + | 1:3 volumens 1 plasmid:15 insert | ||
| + | 1:6 volumens 1 plasmid:30 insert | ||
| + | |||
| + | ==== Colony PCR on ligation from 24/8 ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: Colony PCR<br> | ||
| + | protocos:[https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1] | ||
| + | Notes:<br> | ||
| + | 15 colonies were picked form different plates, with differnt plasmid to insert ratio: colonie's 1,2,3,13 were picked from plates with 1:1 plasmid to insert ratio, colonie's 4,5,6 were picked from plates with 1:3 plasmid to insert ratio and colonie's 7,8,9,10,11,12,14,15 were picked from plates with 1:6 plasmid to insert ratio.<br> | ||
| + | The PCR program was:<br> | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <table style="text-align: left; width: 300px;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>PCR</td> | ||
| + | <td>Temp. (C)</td> | ||
| + | <td>Time (min)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Start</td> | ||
| + | <td>95</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Denaturing</td> | ||
| + | <td>95</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Anneling</td> | ||
| + | <td>68,0</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Elongation</td> | ||
| + | <td>72</td> | ||
| + | <td>4</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>End</td> | ||
| + | <td>72</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Hold</td> | ||
| + | <td>4</td> | ||
| + | <td>indef.</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | <br> | ||
| + | </body> | ||
| + | </html> | ||
| + | A gel was run on the PCR products:<br> | ||
| + | Futher experiments and PCR was run on tubes: 5,6,10,11 because they have the greatest yeild.<br> | ||
| + | |||
| + | ==== Restriction Digest ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: Restriction Digest<br> | ||
| + | protocos:[https://2010.igem.org/Team:SDU-Denmark/protocols#RD1.1 RD1.1] | ||
| + | Notes:<br> | ||
| + | Restriction digest was performed on tubes 5,6,10 and 11 to test for insertion of ninaB (Correct orientation) XbaI and SPEI was used and a gel was run:<br> | ||
| + | |||
| + | ==== Colony PCR on ligation from 24/8 (colonies 5,6,10 and 11 + new colonies) ==== | ||
| + | Date: 24/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: Colony PCR<br> | ||
| + | protocos:[https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1 CP1.1] | ||
| + | Notes:<br> | ||
| + | 8 new colonies (16-23) were chosen in addition to colonies 5,6,10 and 11 form 25I8-10 colonie PCR.<br> | ||
| + | PCR were run with TAQ polymerase and VF2 + VR primers according to the following program:<br> | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <table style="text-align: left; width: 300px;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>PCR</td> | ||
| + | <td>Temp. (C)</td> | ||
| + | <td>Time (min)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Start</td> | ||
| + | <td>94</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Denaturing</td> | ||
| + | <td>94</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Anneling</td> | ||
| + | <td>55,0</td> | ||
| + | <td>0.5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Elongation</td> | ||
| + | <td>72</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>End</td> | ||
| + | <td>72</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Hold</td> | ||
| + | <td>4</td> | ||
| + | <td>indef.</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | A gel was run on the products:<br> | ||
| + | ==== PCR on NinaB with old primers (NinaBfw and NinaBrv) ==== | ||
| + | Date: 27/8<br> | ||
| + | Done by: Marie & Tommy<br> | ||
| + | Methods: PCR<br> | ||
| + | protocos:CP1.1[https://2010.igem.org/Team:SDU-Denmark/protocols#CP1.1] | ||
| + | Notes:<br> | ||
| + | PCR was run on NinaB (from PCR with NinaBfw and NinaBrv), this time with the old primers (NinaBfw and NinaBrv) in an attempt to add the E and P restriction sites to the NinaB fracment.<br> | ||
| + | The PCR program was set to:<br> | ||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <table style="text-align: left; width: 300px;" border="1" | ||
| + | cellpadding="2" cellspacing="2"> | ||
| + | <tr> | ||
| + | <td>PCR</td> | ||
| + | <td>Temp. (C)</td> | ||
| + | <td>Time (min)</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Start</td> | ||
| + | <td>95</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Denaturing</td> | ||
| + | <td>95</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Anneling</td> | ||
| + | <td>55,0</td> | ||
| + | <td>0.75</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Elongation</td> | ||
| + | <td>72</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Denaturing</td> | ||
| + | <td>94</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Anneling</td> | ||
| + | <td>73,0</td> | ||
| + | <td>0.75</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Elongation</td> | ||
| + | <td>72</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>End</td> | ||
| + | <td>72</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Hold</td> | ||
| + | <td>4</td> | ||
| + | <td>indef.</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | NinaB From second new primer PCR (23/8, purifid 24/8) was used as templated.<br> | ||
Latest revision as of 22:31, 23 October 2010
 "
"