Team:Imperial College London/Modelling
From 2010.igem.org
m |
m |
||
| Line 64: | Line 64: | ||
|<b style="font-size:12px">Output Amplification Model</b><br/> | |<b style="font-size:12px">Output Amplification Model</b><br/> | ||
Goals: | Goals: | ||
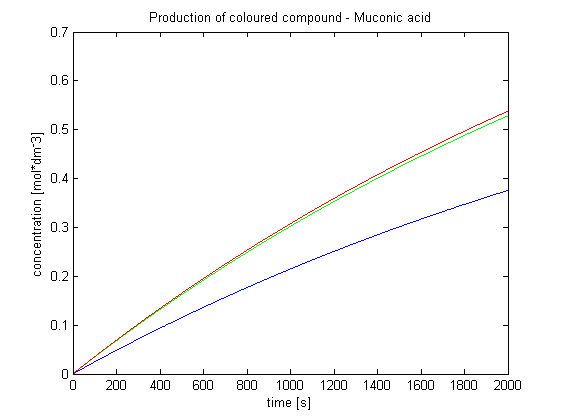
| - | <ol><p>This model was mainly developed in order to determine whether simple production is better than 1- or 2-step amplification.</p> | + | <ol><p>This model was mainly developed in order to determine whether simple production is better than 1- or 2-step amplification.</p>Furthermore, an estimation of the speed of the response was desirable. |
</ol> | </ol> | ||
Elements of the system: | Elements of the system: | ||
| - | <ol><li>Dioxygenase (<i>blue on diagrams below</i>) is an enzyme that acts on catechol to produce yellow output. In most of our models dioxygenase was treated as an output | + | <ol><li>Dioxygenase (<i>blue on the diagrams below</i>) is an enzyme that acts on catechol to produce a yellow output. In most of our models dioxygenase was treated as an output because it was found that active dioxygenase acting on catechol produces the coloured output within a split second.</li> |
| - | <li>GFP-Dioxygenase fusion protein (<i>GFP is shown green on diagrams</i>). Dioxygenase joined by the linker to GFP was assumed to be inactive.</li> | + | <li>GFP-Dioxygenase fusion protein (<i>GFP is shown green on the diagrams</i>). Dioxygenase joined by the linker to GFP was assumed to be inactive.</li> |
| - | <li>TEV protease (<i>yellow on diagrams below</i>) has ability to cleave GFP-Dioxygenase fusion protein, hence, | + | <li>TEV protease (<i>yellow on the diagrams below</i>) has the ability to cleave the GFP-Dioxygenase fusion protein, hence, it activates dioxygenase</li> |
| - | <li>Split TEV protease (<i>orange on diagrams below</i>) | + | <li>Split TEV protease (<i>orange on the diagrams below</i>) is an inactive split form of TEV mounted on coiled coils. It can be activated again by coiled coils being cleaved by another active TEV.</li> |
</ol> | </ol> | ||
| Line 93: | Line 93: | ||
</html> | </html> | ||
|- | |- | ||
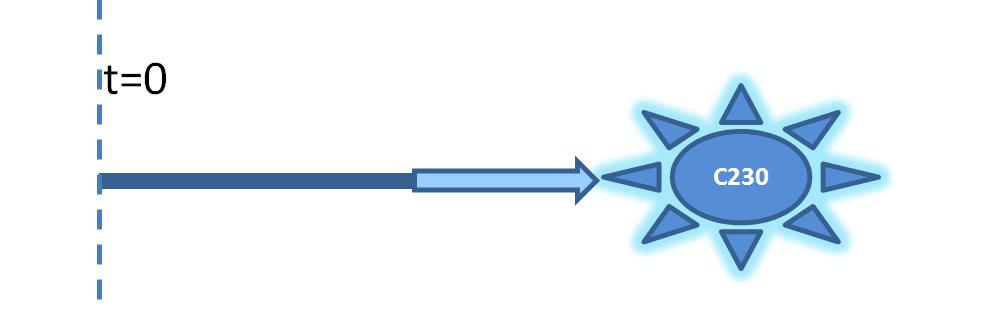
| - | |Diagram showing 1 step amplification. TEV enzyme is | + | |Diagram showing 1-step amplification. TEV enzyme is produced (by simple production) to activate pre-produced inactive dioxygenase. |
|} | |} | ||
</div> | </div> | ||
| Line 100: | Line 100: | ||
|- | |- | ||
|<html> | |<html> | ||
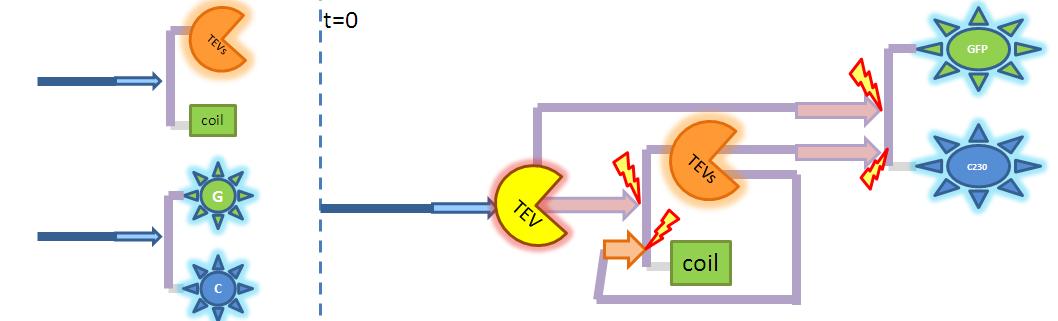
| - | <img src="http://www.openwetware.org/images/0/02/2-step_amplification.JPG" width="275" alt="Diagram illustrating 2 step amplification. Enzyme | + | <img src="http://www.openwetware.org/images/0/02/2-step_amplification.JPG" width="275" alt="Diagram illustrating 2-step amplification. Enzyme activates another enzyme which activates dioxygenase. Both pre-products have the same TEV-site, so simply produced TEV is allowed to act directly on dioxygenase too." /> |
</html> | </html> | ||
|- | |- | ||
| - | |Diagram illustrating 2 step amplification. Enzyme | + | |Diagram illustrating 2-step amplification. Enzyme activates another enzyme which activates dioxygenase. Both pre-products have the same TEV-site, so simply produced TEV is allowed to act directly on dioxygenase too. |
|} | |} | ||
</div> | </div> | ||
| Line 112: | Line 112: | ||
<ol> | <ol> | ||
<li>The chemical and enzymatic reactions are modelled according to the Law of Mass Action.</li> | <li>The chemical and enzymatic reactions are modelled according to the Law of Mass Action.</li> | ||
| - | <li> | + | <li>Our model assumes that the modelled system is inert within the bacterial body or that reactions with other species within the bacterium is negligible. For example, the TEV protease is not supposed to cleave other molecules due to its specifity.</li> |
</ol> | </ol> | ||
| Line 118: | Line 118: | ||
<b style="font-size:12px">Surface Protein Model</b><br/> | <b style="font-size:12px">Surface Protein Model</b><br/> | ||
Goals: | Goals: | ||
| - | <ol><p>The aim of this model is to determine the concentration of Schistosoma elastase or TEV protease that should be added to bacteria to trigger | + | <ol><p>The aim of this model is to determine the concentration of Schistosoma elastase or TEV protease that should be added to the bacteria in order to trigger a response. This would allow us to correlate the required concentration for the activation with the concentration of Schistosoma elastase in the lake.<</p> |
| - | <p>It | + | <p>It was also attempted to model how long it takes for the protease or elastase to cleave the required amount of peptides.</p> |
</ol> | </ol> | ||
Elements of the system: | Elements of the system: | ||
| - | <ol><li> | + | <ol><li>The surface protein consists of a cell wall binding domain, linker, AIP (Auto Inducing Peptide)</li> |
| - | <li>Schistosoma elastase (enzyme released by the parasite) cleaves AIP from cell wall binding domain at the linker site. In laboratory we used TEV protease as we could not | + | <li>Schistosoma elastase (this is the enzyme released by the parasite) cleaves AIP from the cell wall binding domain at the linker site. In the laboratory we used TEV protease as we could not obtain the Schistosoma elastase.</li> |
| - | <li>ComD receptor is | + | <li>The ComD receptor is activated (i.e. AIP concentration is high enough).</li> |
</ol> | </ol> | ||
| Line 131: | Line 131: | ||
<ol> | <ol> | ||
<li>The chemical and enzymatic reactions are modelled according to the Law of Mass Action.</li> | <li>The chemical and enzymatic reactions are modelled according to the Law of Mass Action.</li> | ||
| - | <li> | + | <li>Our model assumes that the modelled system is inert within the bacterial body or that reactions with other species within bacterium is negligible. For example, the TEV protease is not supposed to cleave other molecules due to its specifity.</li> |
| - | <li>Due to carefully chosen cell concentrations, the diffusion of free AIPs could be neglected. However, | + | <li>Due to our carefully chosen cell concentrations, the diffusion of free AIPs could be neglected. However, this restricts the model to the considered cell concentrations only.</li> |
| - | <li> | + | <li>The threshold for receptor activation was defined by one specific value as opposed to considering intermediate states between fully "off" and "on".</li> |
</ol> | </ol> | ||
Revision as of 08:55, 15 October 2010
| Temporary sub-menu: Output Amplification Model; Surface Protein Model; Wet-Dry Lab Interaction; Dry Lab Diary; |
| Introduction to modelling |
In the process of designing our construct two major questions arose which could be answered by computer modelling:
|
| Results & Conclusions | ||||
Output Amplification Model
|
| Quick overview of models | |||||||||
| Output Amplification Model Goals: This model was mainly developed in order to determine whether simple production is better than 1- or 2-step amplification. Furthermore, an estimation of the speed of the response was desirable.Elements of the system:
The aim of this model is to determine the concentration of Schistosoma elastase or TEV protease that should be added to the bacteria in order to trigger a response. This would allow us to correlate the required concentration for the activation with the concentration of Schistosoma elastase in the lake.< It was also attempted to model how long it takes for the protease or elastase to cleave the required amount of peptides. Elements of the system:
Major assumptions:
|
 "
"