|
SEPTEMBER: WEEK 3
September, 13th
Unfortunately we discovered we tried to ligate I32 (E-S) to I37 (X-P) instead of I37 (E-X) ;-(
So this step will be repeated.
Trasformation of ligations
into E. coli DH5-alpha.
Inoculum of
into 5 ml LB+Amp for ligations of the following day.
Inoculum from single colony of MG42 and MC43 in 5 ml LB+Cm12.5, than ON at 37°C.
September, 14th
Finally Mr.Gene sent us our YEAST: a little difficult to pick up the package but at the end we succeeded!
I52, I54, I55, I56, I57, I58 plates showed in general few colonies; I52 and I55 showed very few colonies (<=5). Very strange (mumble mumble...), they will be screened ASAP (stored at +4°C).
Miniprep and quanfification of:
- I26: 69,7 ng/ul
- I31: 89,5 ng/ul
- I34: 147,6 ng/ul
3-hours digestion:
- I26: XbaI-PstI (Insert)
- I31: EcoRI-SpeI (Insert)
- I34: E-X (Vector)
Gel run/cut of samples (I26 and I31 insert bands were very very soft)
 Gel run/cut for digested I26, I31 and I34. and gel extraction:
- I26 (X-P): 2,6 ng/ul
- I31 (E-S): 1,4 ng/ul
- I34 (E-X): ng/ul
We already had digested DNA so we could perform new ON ligations
- I59: I21 (E-S) + I34 (E-X)
- I60: I20 (S-P) + I26 (X-P)
- I61: I31 (E-S) + I37 (E-X)
and repeat
- I53: I32 (E-S) + I37 (E-X)
Screening PCR on 3 colonies picked from MC42 and MC43 respectively.
Two method were used: our standard PCR picking a colony from the plate and using it for the PCR and a different protocol picking the colony and treating it with a 95°C 5 min step before the PCR.
The results were the follow:
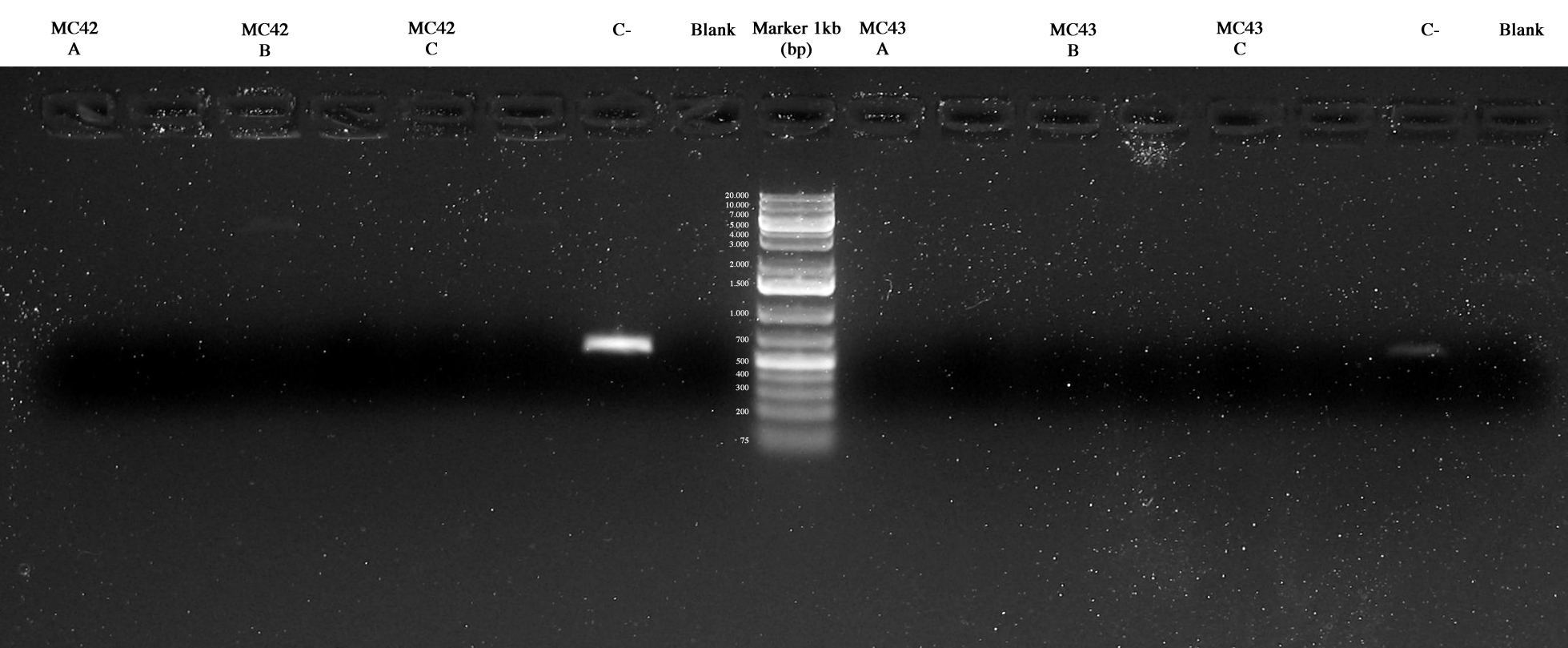 MC42A/B/C, Cneg, blank, MC43A/B/C, Cneg, blank. Negative controls were ok and maybe MC42_C and MC43_C were ok too but further investigations were necessary.
5 ul of MG42 and MG43 were streaked on LB+Cm12.5 agar plates and incubated ON at 43°C.
Tecan Test was performed on prepared samples, after the usual protocol (dilution, medium change and dilution 1:1000).
September, 15th
Transformation of I 53, I59, I60, I61 ligations into E. coli DH5-alpha. They were plated on LB+Amp agar plates and let grow ON at 37°C.
3 colonies were picked from each MG plates and screening PCR was performed also with MC42 and MC43 samples. Two methods with different DNA polymerases were used in order to identify best experimental conditions (Taq polymerase and Pfx accuprime polymerase). The amplicons length (about 4Kb) probably was the reason of our problem in all the PCRs.
 Screening PCR. In order: MC42ABC, MG42ABC, Cneg, BLANK, MG42ABC, MG43ABC, Cneg, BLANK (Taq samples over and Pfx samples under) September, 16th
All plates showed colonies, so we could perform screening through "colony PCR" for ligations:
- I52
- I53
- I54
- I55
- I56
- I57
- I58
- I59
- I60
- I61
For each plate we picked 3 colonies that were also inoculated into 1 ml LB+Amp in order to be ready to make glycerol stock of positive ones.
Agarose gel was prepared and samples loaded and run:
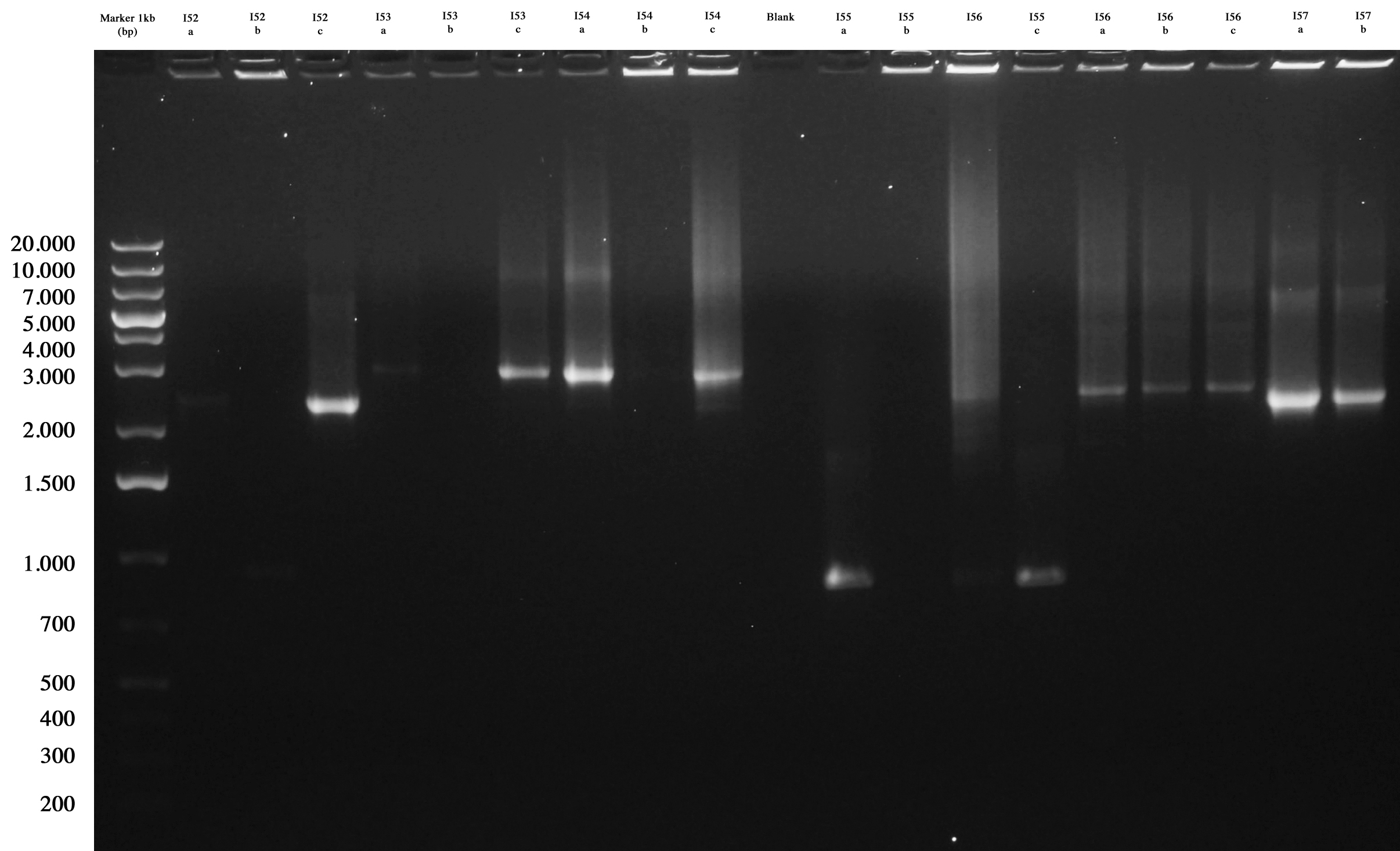 Screening gel run for ligations I52..I57. |  Screening gel run for ligations I58..I61. |
As you can see I52c, I53a/c, I54a, I56a/b/c, I57a/b are positive. So we decided to make glycerol stocks for I52c, I53c, I54a, I56a and I57a. I55 was negative. We would have performed colony PCR again. Probably all colonies I58..I60 were positive but their length was a little longer than expected. So we decided to further screen them through digestion.
I58a/b/c, I59a/b/c, I60a/b/c and I61a/b/c were let grow ON at 37°C in order to perform miniprep the following day.
MC and MG cultures OD was referred to the lowest one in order to perform Tecan Test (note that MG42-A didn't grow so we didn't perform test on this sample). Samples were then centrifuged: the resulting pellets were resuspended on 1 ml of PBS.
Tecan Test was performed on prepared samples, after the usual protocol (dilution, medium change and dilution 1:1000).
September, 17th
Miniprep and Nanodrop quantification for:
- I58a: 127,5 ng/ul
- I58b: 108,9 ng/ul
- I58c: 100,1 ng/ul
- I59a: 161,6 ng/ul
- I59b: 268,8 ng/ul
- I59c: 214,3 ng/ul
- I60a: 151,6 ng/ul
- I60b: 149,7 ng/ul
- I60c: 157,6 ng/ul
- I61a: 147,1 ng/ul
- I61b: 138,3 ng/ul
- I61c: 125,3 ng/ul
Digestion (for screening) and gel extraction of the positives (for next ligation step) for:
- I58a/b/c: EcoRI-SpeI
- I59a/b/c: XbaI-PstI
- I60a/b/c: E-S
Digestion (only screening) for
Agarose gel was prepared and samples loaded and run (and cut were necessary)
| |  Screening and gel extraction for positive I59, I60. Screening for I61. |
As you can see they are all positive (I58 was run a little longer to better separate bands). So we stocked and gel extracted I58c, I59b, I60a and only stocked I61a.
September, 18th
Gel extraction and quantification of:
- I58 (E-S): 13.8 ng/ul
- I59 (X-P): 24.6 ng/ul
- I60 (E-S): 12.8 ng/ul
DNA was than stored at -20°C.
September, 19th
Since there were no more colonies in I55 agar plate, we had to transform it again to perform screening through colony PCR. So I55 was transformed again into E. coli DH5-alpha and plated on LB+Amp agar plate, that was let grow ON at 37°C.
|
|