Team:NYU/Parts
From 2010.igem.org

When choosing what simple parts to make for our project we had two main goals: 1) to improve the quality of parts already in the registry by making them more accessible to all users and 2) to augment the current set of yeast-compatible biobricks to harness the power of this synthetic biology-friendly model organism. Incorporating these goals with the 'Quality not Quantity' mantra, we decided to focus our efforts on certain areas of advancement that we believe future researchers will like to see in the registry.
1. To improve parts already in the registry by lessening the constrictions on their use and making them more adaptable.
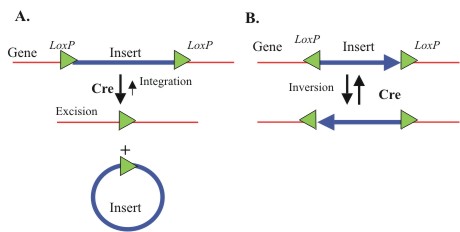
- 36bp LoxP site: the LoxP site is the naturally-occurring substrate for the Cre recombinase. In nature (and in previous biobricks) it is a palindromic sequence 34 base pairs long. Because this is not a multiple of three, when used inside a transcribed ORF the 34bp LoxP site can throw off the frame of the translated parts. To get around this, previous teams had to either add or take away bases from surrounding parts to keep their systems in frame (painstakingly and sometimes unsuccessfully). To get around this restriction, we designed and Biobricked a 36bp LoxP site that Registry users can include in transcribed regions without worrying about keeping everything in frame.
- pGAL-RBS: This promoter is induced by the presence of galactose and repressed by the presence of glucose. Because most future projects will involve fusing a Ribosome Binding Site to the promoter we decided to simplify things and submit this construct to the Registry.
- pTEF-RBS: This promoter is a medium-strength constitutive promoter. We submitted the part fused to the RBS.
- lexOpCYC-RBS: This pCYC promoter has a very limited amount of constitutive transcription. Just upstream is the lex operator to which the LexA DNA binding domain will bind. This can be useful for a number of constructs including our yeast two-hybrid screen. Again, we submitted this part with the RBS attached.
2. Yeast strains are more rarely used in iGEM projects but contain certain parameters that are ideal for certain projects. Many teams are beginning to use yeast this year and are submitting useful biobricks, so we decided to make a collection of biobricks parts aimed at protein localization within the cell (known as 'yeast functional tags'):
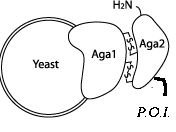
- Aga2: The Aga2 protein is screted from the yeast cell and attaches itself through disulfide bonds to the Aga1 protein on the cellular surface. If the Aga2 protein fused to a part of interest, the fusion is secreted, Aga2 complexes with Aga1 on the cellular surface, and the part of interest is displayed on the surface of the cell. While we did not end up using this part in our official construct, biobricking it from a plasmid we already had in our lab will surely benefit future teams.
- Aga2+Linker: Most everyone will want a linker attached to the Aga2 protein, so we went ahead and submitted this construct as well. COLLABORATION: We also sent this part along with the secretion tag to iGEM Osaka to aid them in the wetwork for their own project.
- Secretion Tag: Another 'yeast functional tag', the secretion tag does just what you think when fused to the C terminus of a protein of interest - directs secretion of the fused protein. Cleverly, this tag is cleaved from the fusion along the secretion pathway, resulting secretion of the protein of interest without the tag attached.
- Ura3: This is one of the proteins in the pathway for Uracil biosynthesis. It is incredibly useful as a selectable marker in yeast genetics because it can be selected for and against. We use it in our system as the nutritional marker in our yeast two-hybrid screen.
3. To construct and submit certain ubiquitously useful biobricks that we would be using.
- Flexible (Gly4Ser)3 peptide linker: for use with most any peptide fusions
4. To provide reporter constructs as positive controls of commonly used devices
- pGal-Aga2-eCFP: This reporter secretes Aga2:eCFP when the yeast is grown in galactose media. It is used as a positive control for the function of the surface display system. This construct was used by our team to test whether the Aga2 protein could direct secretion of a fusion and if the the Aga1 protein was present on the extracellular surface of our strain.
- pGal-LexBD-NLS-VP16:This construct should produce a nucleus-localized LexBD:VP16 fusion protein when grown in galactose media. This can be used as a positive control for any yeast two-hybrid system that uses the Lex operator and Lex binding domain.
Parts
New for iGEM 2010 is the groupparts tag. This tag will generate a table with all of the parts that your team adds to your team sandbox. Note that if you want to document a part you need to document it on the [http://partsregistry.org Registry], not on your team wiki.
<groupparts>iGEM010 NYU</groupparts>
 "
"