Team:TU Munich/Project
From 2010.igem.org
Hartlmueller (Talk | contribs) (→Implementation) |
Hartlmueller (Talk | contribs) (→Implementation) |
||
| Line 46: | Line 46: | ||
*RNA production is fast and energy saving for a cell containing the adapter. | *RNA production is fast and energy saving for a cell containing the adapter. | ||
*As the half-time of RNA can be rather short, transmitter RNA will not accumulate within the cell and it is therefore less likely for the system to become saturated. | *As the half-time of RNA can be rather short, transmitter RNA will not accumulate within the cell and it is therefore less likely for the system to become saturated. | ||
| - | * | + | |
| + | ==Design and functionality of logic gates== | ||
| + | The concept introduced above provides a framework that can potentials serve as an universal adapter between different BioBricks. However, the logic gates have not been specified more precisely so far. Generally speaking, the logic gates have to posses the following characteritics: | ||
| + | *Logic gates, such as AND, OR and NOT, have to be implemented. | ||
| + | *All logic gates have to recognize the corresponding transmitter RNA and, in responds, produce an output transmitter molecule. | ||
| + | *Logic gates should follow a basic design rule, in such a way, that their creation can automated in silico. | ||
| + | *The responds function of all logic gates should be comparable to each other. | ||
| + | |||
| + | In the following, a concept is introduced that will meet all requirements | ||
| + | |||
| + | |||
| + | |||
| - | |||
| - | |||
| - | |||
==How is it really working - The smallest locial unit - establishing an equipollent to an electronic == | ==How is it really working - The smallest locial unit - establishing an equipollent to an electronic == | ||
Revision as of 21:27, 23 October 2010
|
|
VisionUntil today, 13.628 biobrick sequences[1] have been submitted to partsregistry, thereof 102 reporter units, 12 signaling bricks and xx sensing parts.
Since there, people are trying to arrange these single biological building blocks in such a manner that allows producing special biotechnological products (metabolic engineering), developing biological sensory circuits (biosensors) and even giving microorganisms the ability to react on multiple environmental factors and serve both as disease indicator and drug. These examples and further promising ideas were implemented on previous iGEM-competitions.[2][3][4]
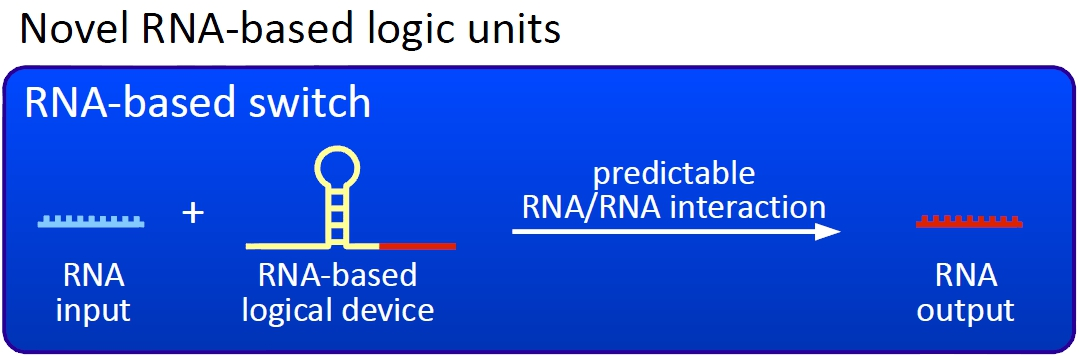
ImplementationTo functionally connect BioBricks there are several possibilities including genetic switches, riboswitches and direct protein-protein interactions. We investigated several hypothetically principles, and decided to focus our practical work on the development of a RNA-RNA interaction-based switch. These switches are capable of changing between two states, a state of antitermination and termination, and make use of highly-specific RNA-RNA interaction. In principle such a switch can fulfill all requirements mentioned previously. The following text clarifies how these switches work in detail.
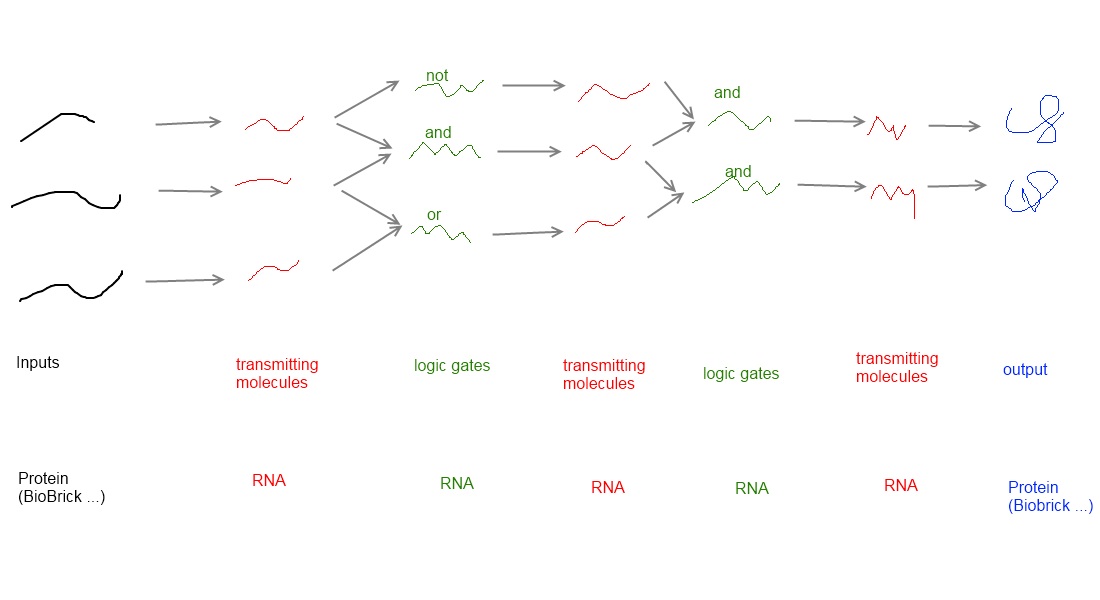
How to connect BioBricksOur adapter is a system, that activates or disables BioBricks (output BioBricks) in response to the presence of other Biobricks (input Biobricks). Our appraoch uses a molecular network to put this into practise and consists of four major elements:
These elements can be combiined to build up a molecular network (see illustration). Each input molecule (such as a BioBrick) produces a transmitter molecule that is unique for this input molecule. All transmitters belong to the same type of molecule and share a common design. However each transmitter molecule can only interact and activate a certain subset of logic gates. In other words, logic gates have to recognize and bind the corresponding transmitter molecules. Depending on the type of the logic gate (AND, OR or NOT[6]), an output molecule is only created if both transmitter molecules are present (AND), at least one of two transmitter molecules is present (OR) or if no transmitter is present at all (NOT). Consequently a certain subset of output molecules are generated accoring to the presence of transmitter molecules, thus also accorind to the corresponding BioBricks. Computer vs. molecular netowrk
Logic gates in a molecular network can be compared to transistors used in a computer where billions of transistors are incorporated. on a computer chip, these transistors are connected by wires in such a way that the output of one transistor only acts on a subset of other transistors. However, spatial connection are not possible in a living cell. The wiring within a cell relies on specific interaction between the transmitter molecule and the correcponding logic gates. Again, these output molecules are transmitter molecules and can in turn interact with another subset ("layer") of logic gates. In theory many layers of logic gates can be connected by transmitters. The last layer of logic gates has to generate transmitter molecules, that do not interact with logic gates but induce output molecules, such as Biobricks, instead. Summarizing, the network established a connection between input BioBrick and output BioBrick in a functional manner.
Design and functionality of logic gatesThe concept introduced above provides a framework that can potentials serve as an universal adapter between different BioBricks. However, the logic gates have not been specified more precisely so far. Generally speaking, the logic gates have to posses the following characteritics:
In the following, a concept is introduced that will meet all requirements
How is it really working - The smallest locial unit - establishing an equipollent to an electronictransistor unit THe main goal is to provide universality, so input and output on same basis, RNA, transcribiton, sin THe main goal is to provide
switch
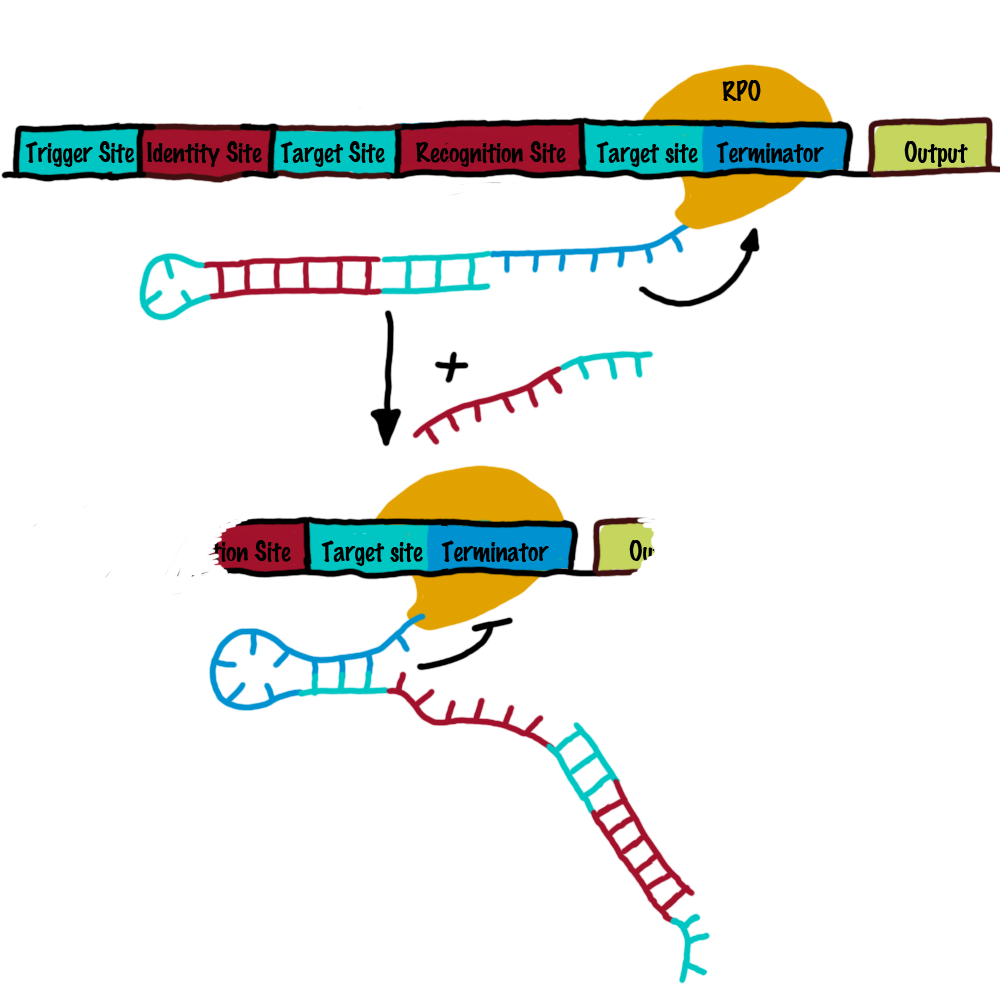
The switching unit is the functional core element of our switches, allowing a shift between an "on" and "off" state. Since we work on the level of transcription, a "switchable" transcriptional terminator is suitalbe for this purpose. The principle idea of our switching unit relies on such systems occuring in nature. We focus on two system: - Attenuation-Operon - tiny abortive RNA´s CloseThe special thing about it is all of our switches consist of THE SAME switching unit, so having found one functional "switchable" terminator will allow almost unlimited upscaling. This is the main difference to previous works on this field, which always required to develop a new shifting principle for each switch. Beside the extendablity furhter advantages are we can provide a comparable on/off shifting rate. This makes our switches more similar to transistors than ... Practically, the shift between the two states is induced by complementrary RNA-sequences, influencing the terminators secoundary structure. To seperate this switching element from the specific accessiblity, we designed a synthetic switching unit relying on NUPACK simulations, which is not able to change the terminators state on its own, but only in combination with a specific recognition site:
It defines the specific access of one of our switches by an input molecule. Therefore, a unique recognition element is assigned to each switch. This allows to to arrange and interconnect numerous of these switches in a specif logical order, comparable to wires connecting different transistors.
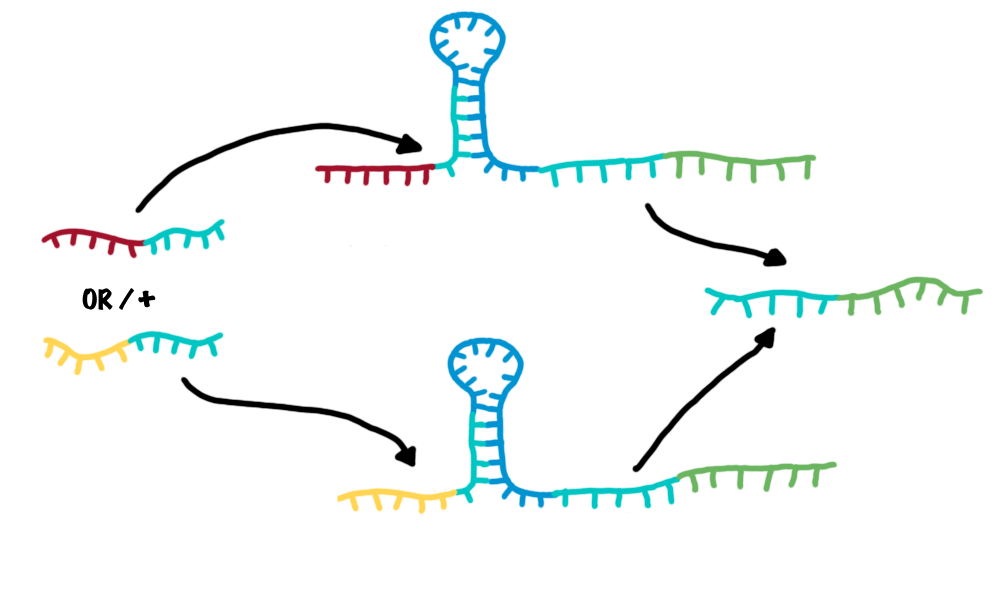
It is implemented by simply putting a random sequence with arbitrary lenght (has to be optimised) in front of the switching unit. Christoph: thermodynamisch kinetisch usw reicht dass um zu schalten?? xxx possbilites to build unique switches. (blacklist, etc) Closesignals, the inputs and outputs of the bioLOGICS switchSignals present the "trigger" to shift switches between on and off. It requires the ability to change the terminators secondary structure BUT, only if a special recognition site is detected. Thus, each signal consists of a trigger unit, interacting with the swichtes´ switching unit and a specifity site, interacting with the terminators recognition site. The challenge is to arrange and optimise these elemantary building blocks, that a trigger unit is only able to switch in combination with its respective specifity site. CloseThe bioLOGIC switch with regard of logical operationsAs described, each switch can be accessed by a specific RNA-signal molecule, illustrating the input. In turn, another RNA-signal molecule will be produced if the switch shifts its state, now beeing the output of one switch and at the same time, a possible input for the following switch or several ones. This easily allows arranging several switches in specific sequences and faulty wiring - the corner stone of a logical network. To easen the building of logical networks, applying mathematical logcis, e.g. Boolean logics like in computaltion science would be worthwile. It is possible to establish general Boolean operators with our switches and thus build "logical modules".
Since AND/OR/NOT are the most easy logic operations modularisable with our switches and can substitute all remaining operations, we exemplarly designed them.
EvaluationTo evaluate functionality of our molecular switches, we developed several in vivo and in vitro assays and relied on existing assays. Thus we don't want to develop additional biobricks, we rather want to establish a platform to realize the full opportunity of the biobrick system. This would enable people easily setting up sensor - reporter circuits AND interconnect them to complete biological chips... the way to real artificial cells and synthetic biology. (zu krass?) Network constructionDesigning complex biological networks based on either traditional protein engineering or our new bioLOGICS is still a complex task. We developed a software which allows the fast construction of a bioLOGICS based networks. ConceptThe basic principle of our switches are short RNA sequences, and the scientific idea shares similiarities with the principle of antitermination, but also inherits a completely new way of RNA based transcription regulation. We used three-dimensional structure predictions and thermodynamic calculation to develop a set of switches - about 50 nucleotides - and signals - about 20 nucleotides. The switches form a stem loop causing transcription termination, which can be resolved upon binding of the specific signal. On/off switching can therefore be easily controlled by signal avaiability and provides a new concept to control gene transcription.
Our switches are based on the principle of transcriptional antitermination. Transcription can be cancelled by the formation of a RNA stem loop in the nascent RNA-chain, which then causes the RNA polymerase to stop transcription and fall off the DNA. We use sequences as transcriptional switches, that are calculated to be capable of stem loop formation. We plan to control the termination at these switches with a small RNA molecule (our RNA "signal") that is complementary to a part of the stem loop forming sequence. This small functional RNA inhibits the stem loop formation by complementary base-pairing and hence avoids transcription termination. The signal is composed of two parts: While the first part provides specifity (recognition site), the second part causing stem loop disintegration (functional core) can in principle be the same for all bioLOGICS. Therefore variation of the first part allows the construction of an endless number of switches. The functional core causing stem loop disintegration is based on a working system (see attenuation) established by nature. Different stem loops were tested in this effort: Regulatory parts from the E. coli trp-operon, his-operon and one based on previous iGEM-work.
ResultsEvery network starts with a basic unit. While our declared aim is to enable networks allowing fine-tuning of gene expression beyond the regular on/off, exploring such an on/off switch/signal pair is the first step towards a functional network. We constructed several units and tested their efficiency, robustness and reproducibility in vivo, in vitro and in silico. Furthermore we developed a software which allows easy constructions of networks delivering a ready network. Conclusive elaboration of a few first RNA-based logic units is the major contribution of our iGEM team. References[1] http://partsregistry.org/cgi/partsdb/Statistics.cgi [2] https://2009.igem.org/Team:Imperial_College_London/M1 encapsulation [3] https://2009.igem.org/Team:TUDelft [4] https://2008.igem.org/Team:Heidelberg [5] Smolke and so on.... [6] http://en.wikipedia.org/wiki/Logic_gate#Symbols
|
|
 "
"