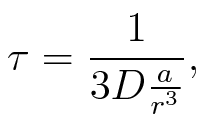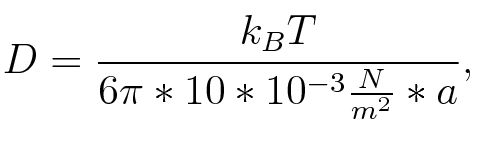Team:TU Munich/Modeling
From 2010.igem.org
(→His-terminator) |
|||
| Line 70: | Line 70: | ||
But, as, for us, the fact whether the terminator is folding or not is crucial and the videos below show that for appropriate signal length the terminator is not folding. Concerning this fact, for all linker lengths we tested there was only a dependence on the signal length but the results were independent from the linker length. Hence, the linker length dependence can be neglected.<br> | But, as, for us, the fact whether the terminator is folding or not is crucial and the videos below show that for appropriate signal length the terminator is not folding. Concerning this fact, for all linker lengths we tested there was only a dependence on the signal length but the results were independent from the linker length. Hence, the linker length dependence can be neglected.<br> | ||
| + | |||
| + | {| | ||
| + | |2 nt | ||
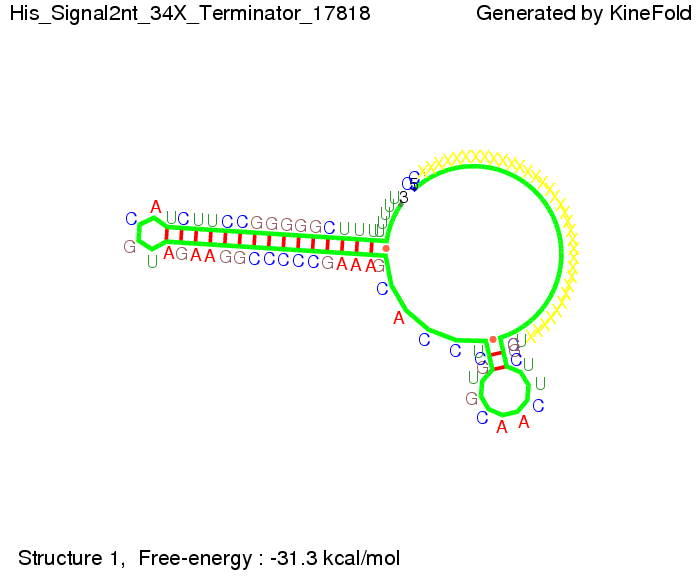
| + | |[[Image: TU_Munich_iGEM2010_sstraub_His_Signal2nt_34X_Terminator-71539_00001.png |thumb|none|450 px |signal length 2 nt]] | ||
| + | |- | ||
| + | |16 nt | ||
| + | |[[Image: TU_Munich_iGEM2010_sstraub_His_Signal16nt_34X_Terminator-54218_00001.png |thumb|none|450 px |signal length 16 nt]] | ||
| + | |- | ||
| + | |32 nt | ||
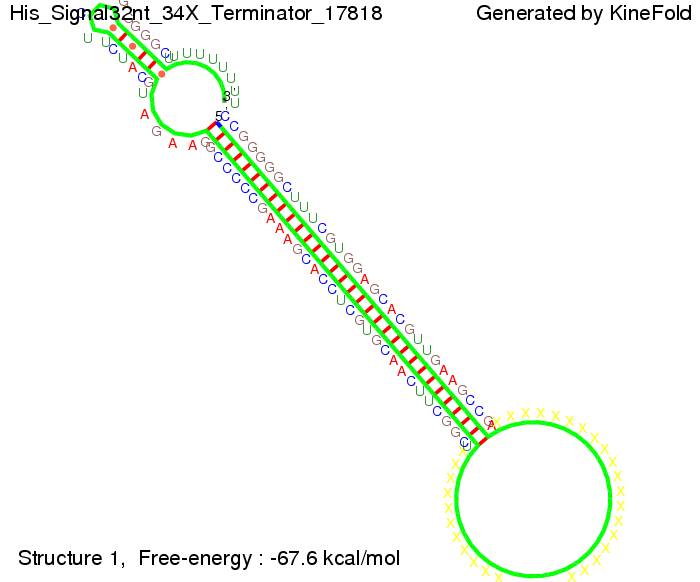
| + | |[[Image: TU_Munich_iGEM2010_sstraub_His_Signal32nt_34X_Terminator-52581_00001.png |thumb|none|450 px |signal length 32 nt]] | ||
| + | |} | ||
| + | |||
| - | |||
| - | |||
| - | |||
===RNA folding path videos:=== | ===RNA folding path videos:=== | ||
Revision as of 13:04, 22 October 2010
|
||||||||
|
|
OverviewWe simulated the termination and anti-termination properties of our signal-terminator constructs with the Kinefold web server and used some standard estimations for diffusive terms. Our main goal was to prove that our constructs work and that termination is stopped efficiently, that is that the signal molecule binds and anti-terminations occurs before the RNA polymerase falls off.
DiffusionThe question whether anti-termination occurs is not only guided by the folding process of the signal-terminator pair, but also by how long the signal takes to diffuse to the terminator sequence.
To account for the diffusion time, we estimated the hit rate τ (following 6.), which is the time until the signal meets the terminator sequence for the first time:
where D is the diffusion constant, a the radius of gyration of the signal molecule and r the radius of the cell.
where n is the length of the signal which is 0,3 nm/monomer, l is the persistency length which is following (5.) 2nm for single-stranded RNA. Thus, for a signal of length 32 nt, a = 6,4 nm. The diffusion constant D was obtained by
where kB is the Boltzmann constant and T is the absolute temperature. Thus, for a cell containing 100 signal molecules, the signal needs 0,1518 s until it first hits the terminator sequence. CloseAs the folding time is significantly larger than the diffusion and thus much less relevant for modeling our signal-terminator constructs, we didn't employ more elaborate techniques to model diffusion. SwitchHis-terminatorTerminator sequence: Both terminator and signal sequence comprise two parts:
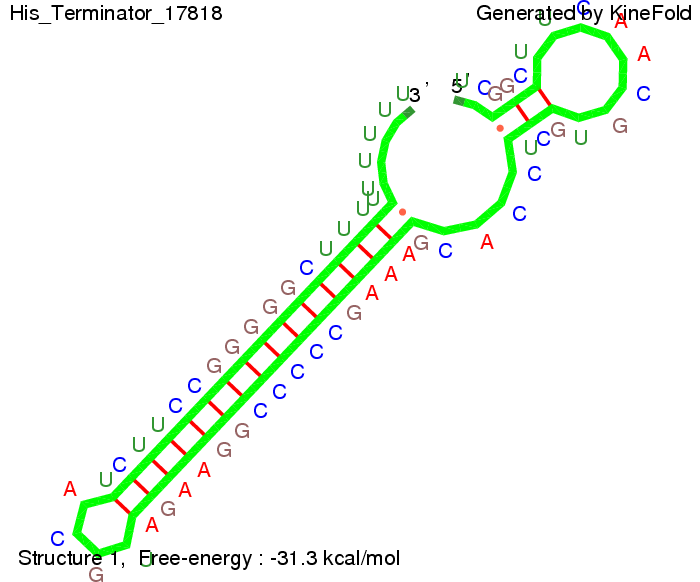
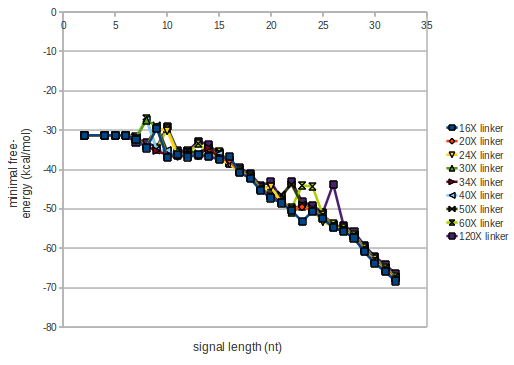
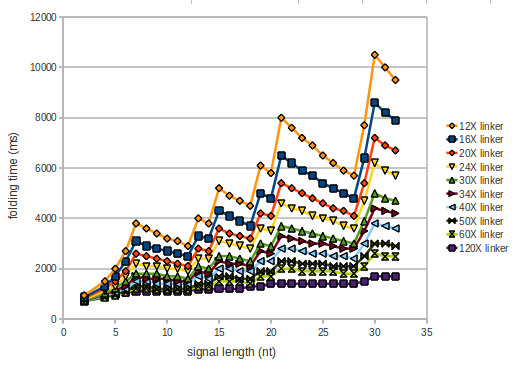
The minimal free-energy structure of the folded terminator including the random signaling part is shown in the figure "His-terminator" below: We modeled the signal-terminator interaction for 2, 4-32 nt signal length and proved that the signal sequence binds to the terminator sequence and impedes the terminator folding, thus the polymerase doesn't fall off and the output signal is produced. As we observed a dependence of the folding time and minimal energy respectively from the linker length we used linkers of length 12, 16, 20, 24, 30, 34, 40, 50, 60, 120 'X' bases. For the minimal energy there is only a very small linker length dependence as one can see in the figure below. The figure also shows that the minimal free-energy structure for the signal sequence bound to the terminator sequence is much more stable, around 35 kcal/mol, than the minimal free-energy structure for the terminator sequence alone. However, for the folding time linker length dependence is significant as one can see in the figure below. But, as, for us, the fact whether the terminator is folding or not is crucial and the videos below show that for appropriate signal length the terminator is not folding. Concerning this fact, for all linker lengths we tested there was only a dependence on the signal length but the results were independent from the linker length. Hence, the linker length dependence can be neglected.
RNA folding path videos:For this video the signal is only 2 nt long. One can see that the terminator is folding and that the signal cannot bind to the terminator sequence due to its short length. This video shows the folding of a terminator and a signal of length 20 nt. which shows that the terminator is already hindered from folding completely. This video is done for full signal length of 32 nt. One can see that the terminator does not fold at all as the signal immediately binds to the terminator sequence as it is synthesized. Trp-terminatorResultsNetworkModelingResultsOutlook |
|||||||
 "
"