Team:SDU-Denmark/project-p
From 2010.igem.org
(Difference between revisions)
(→K343005) |
(→K343007) |
||
| Line 51: | Line 51: | ||
<br> | <br> | ||
'''1. Growth of bacterial culture on semi-solid agar plates (Experiment 1):'''<br> | '''1. Growth of bacterial culture on semi-solid agar plates (Experiment 1):'''<br> | ||
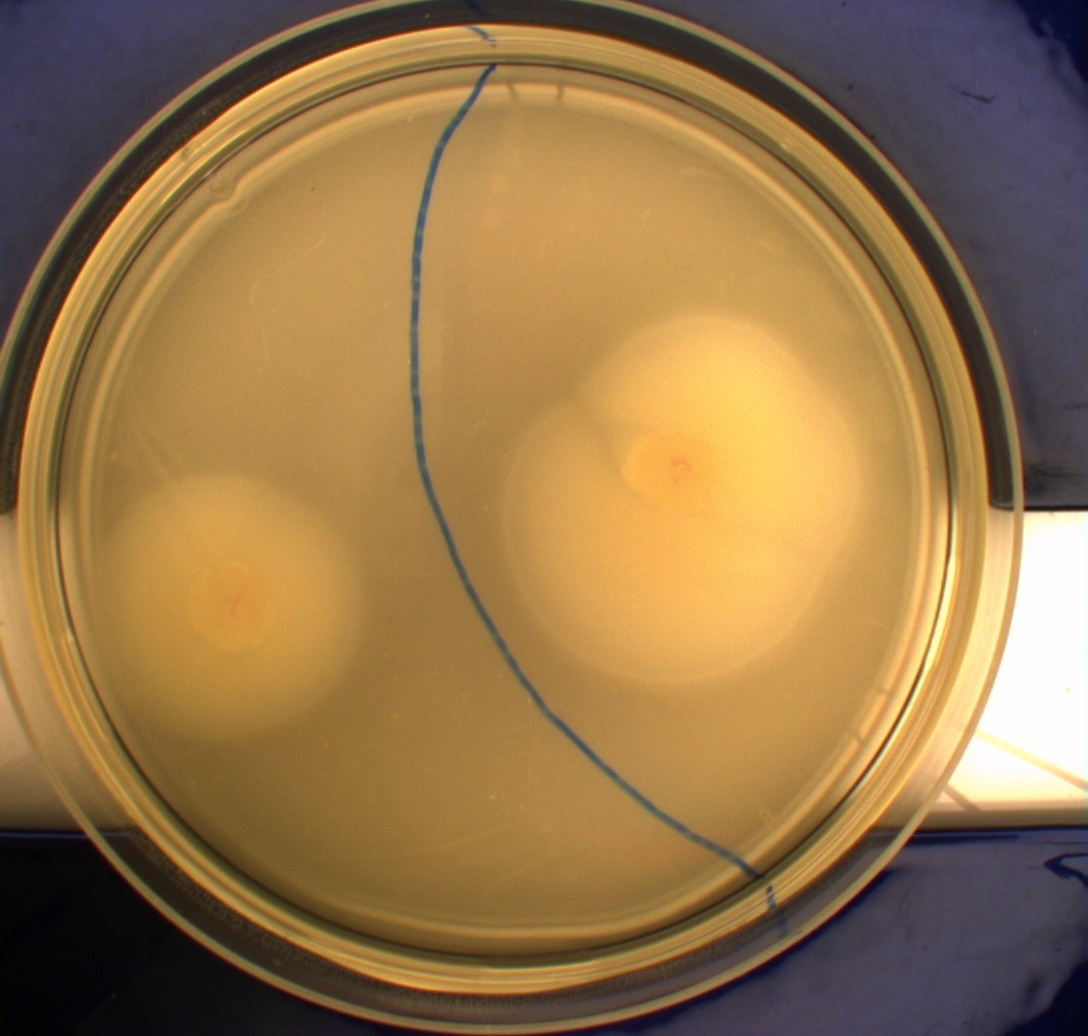
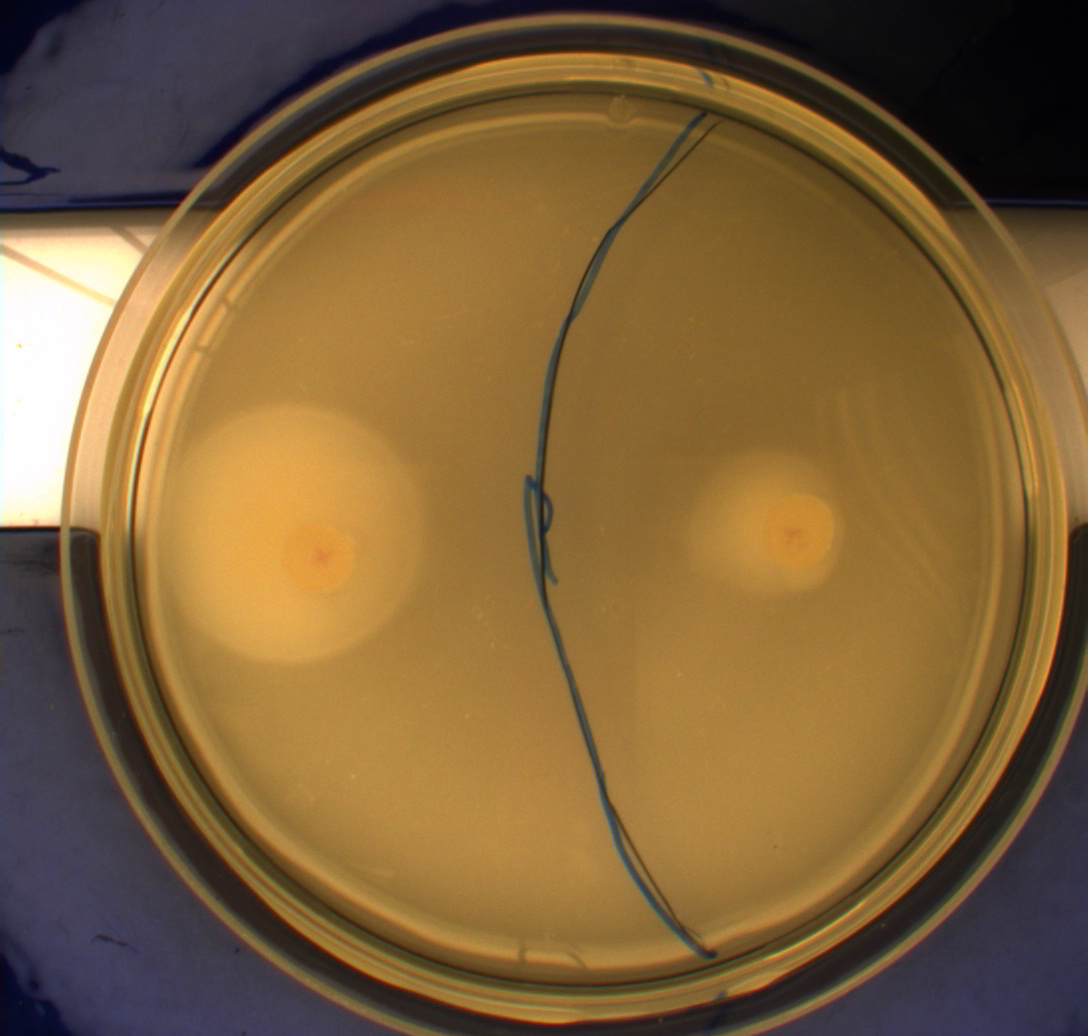
| - | The bacteria and plates were prepared after our own protocol, which you can find here: [https://2010.igem.org/Team:SDU-Denmark/protocols#Photosensor_characterisation] <br> [[Image:Ingeniør1.JPG|200px|thumb|left|Wildtype bacteria (Left half exposed to light, right half exposed to darkness)]][[Image:Ingeniør4.JPG|200px|thumb|left|Phototactic bacteria (Left half exposed to light, right half exposed to darkness)]] | + | The bacteria and plates were prepared after our own protocol, which you can find here: [https://2010.igem.org/Team:SDU-Denmark/protocols#Photosensor_characterisation PS1.1] <br> [[Image:Ingeniør1.JPG|200px|thumb|left|Wildtype bacteria (Left half exposed to light, right half exposed to darkness)]][[Image:Ingeniør4.JPG|200px|thumb|left|Phototactic bacteria (Left half exposed to light, right half exposed to darkness)]] |
Since exposure to blue light should decrease the phototaxic bacterias tumbling frequency, the expected result was that the colony which was placed between the light and dark half of the plate would spread out in the darkness and would not move further when it reached the light. This is counter-intuitive, since decreased tumbling should lead to a longer distance traveled. What happens at the microscopic scale in semisolid agar is that the agar creates a matrix like structure where there are channels through the agar, which the bacteria can swim through. The decrease in tumbling frequency of the bacteria will make it harder for them to find the channels in the agar to swim through, which leads to them being trapped where they were placed. The result is that a colony which shows an increased run time, will look as if it it was non-motile on these plates. Our results showed exactly this; the bacterial culture had spread out to on the dark half of the plate and did not get nearly as far on the half exposed to light. This experiment was done with a normal wildtype MG1655 and a non-motile strain of ''E.coli'', DH5alpha, as controls. As expected these cells did not show anything like the behavior described above, which indicates that the effect stems from the modification to our photosensor bacteria. These results were useable, but not fully conclusive, since there were some non-optimal conditions present in this experiment. We used ambient light instead of pure blue light, and the exposure to light for the multiple samples was not exactly even. Therefore we had to improve our experiment setup and see if we could reproduce these results with a more reliable setup.<br> | Since exposure to blue light should decrease the phototaxic bacterias tumbling frequency, the expected result was that the colony which was placed between the light and dark half of the plate would spread out in the darkness and would not move further when it reached the light. This is counter-intuitive, since decreased tumbling should lead to a longer distance traveled. What happens at the microscopic scale in semisolid agar is that the agar creates a matrix like structure where there are channels through the agar, which the bacteria can swim through. The decrease in tumbling frequency of the bacteria will make it harder for them to find the channels in the agar to swim through, which leads to them being trapped where they were placed. The result is that a colony which shows an increased run time, will look as if it it was non-motile on these plates. Our results showed exactly this; the bacterial culture had spread out to on the dark half of the plate and did not get nearly as far on the half exposed to light. This experiment was done with a normal wildtype MG1655 and a non-motile strain of ''E.coli'', DH5alpha, as controls. As expected these cells did not show anything like the behavior described above, which indicates that the effect stems from the modification to our photosensor bacteria. These results were useable, but not fully conclusive, since there were some non-optimal conditions present in this experiment. We used ambient light instead of pure blue light, and the exposure to light for the multiple samples was not exactly even. Therefore we had to improve our experiment setup and see if we could reproduce these results with a more reliable setup.<br> | ||
<br> | <br> | ||
'''2. Growth of bacterial culture on semi-solid agar plates (Experiment 2):'''<br> | '''2. Growth of bacterial culture on semi-solid agar plates (Experiment 2):'''<br> | ||
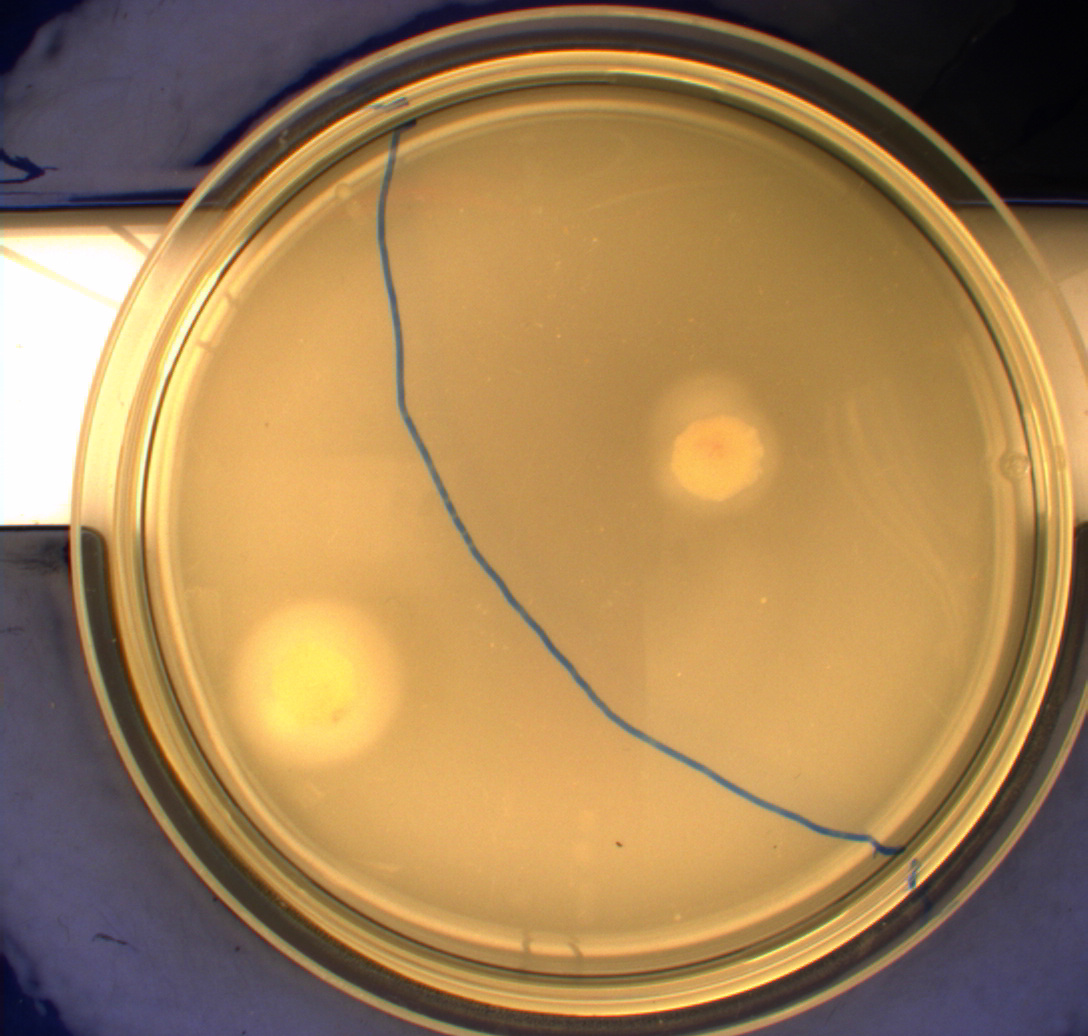
| - | The experiment described above was repeated in a more controlled environment. This means that there were no changes as to how the plates or bacterial cultures were prepared, so we refer again to [https://2010.igem.org/Team:SDU-Denmark/protocols#Photosensor_characterisation] in regards to how this was done. The difference lies in the setup of the light-controlled environment. What we did this time was that the plates was illuminated from above by a single light source in an otherwise completely dark environment, we prepared a cut-out so that the light would only hit one half of our plates and the other half would remain in the dark. [[Image:Lightbox5k.gif|thumb|Schematic over plate positioning and areas exposed to light.]] Since the problem with the last experiment was that the light source was just normal white light (which contains a lot of different wavelengths), this time around we used an optical filter so that only light with a wavelength of around 470nm could pass through, which resulted in a blue light shining down on the plates. The light-source itself was a run-of-the-mill flashlight, with a blue light filter installed in front of the lens. To eliminate the effect of temperature gradients inside the incubator, the three samples were placed in a triangle formation in the center of the incubator. <br> | + | The experiment described above was repeated in a more controlled environment. This means that there were no changes as to how the plates or bacterial cultures were prepared, so we refer again to [https://2010.igem.org/Team:SDU-Denmark/protocols#Photosensor_characterisation PS1.1] in regards to how this was done. The difference lies in the setup of the light-controlled environment. What we did this time was that the plates was illuminated from above by a single light source in an otherwise completely dark environment, we prepared a cut-out so that the light would only hit one half of our plates and the other half would remain in the dark. [[Image:Lightbox5k.gif|thumb|Schematic over plate positioning and areas exposed to light.]] Since the problem with the last experiment was that the light source was just normal white light (which contains a lot of different wavelengths), this time around we used an optical filter so that only light with a wavelength of around 470nm could pass through, which resulted in a blue light shining down on the plates. The light-source itself was a run-of-the-mill flashlight, with a blue light filter installed in front of the lens. To eliminate the effect of temperature gradients inside the incubator, the three samples were placed in a triangle formation in the center of the incubator. <br> |
Another deviation from the protocol PS1.1 is that the culture was inoculated at the center of the plate, instead one colony each was inoculated in the center of the light exposed and dark half respectively. This would give us more information on how the bacteria would behave when directly exposed to either the dark or light surroundings, instead of the gradient that was present in the first experiment.<br> | Another deviation from the protocol PS1.1 is that the culture was inoculated at the center of the plate, instead one colony each was inoculated in the center of the light exposed and dark half respectively. This would give us more information on how the bacteria would behave when directly exposed to either the dark or light surroundings, instead of the gradient that was present in the first experiment.<br> | ||
From the results of the first experiment we expected the culture containing K343007 to spread out in the dark and not to spread when exposed to light. The wildtype bacteria should spread out evenly no matter if exposed to light or not and the non-motile strain (DH5alpha) should not move regardless of the light conditions. Our expectations from the first experiment were fulfilled, as the bacteria behaved exactly as expected. This made it possible to conclude that the photosensor has an effect on the bacteria's tumbling frequency, but if it does in fact reduce the tumbling is not possible to say, since both an increased and reduced tumbling frequency will look alike.<br><br> | From the results of the first experiment we expected the culture containing K343007 to spread out in the dark and not to spread when exposed to light. The wildtype bacteria should spread out evenly no matter if exposed to light or not and the non-motile strain (DH5alpha) should not move regardless of the light conditions. Our expectations from the first experiment were fulfilled, as the bacteria behaved exactly as expected. This made it possible to conclude that the photosensor has an effect on the bacteria's tumbling frequency, but if it does in fact reduce the tumbling is not possible to say, since both an increased and reduced tumbling frequency will look alike.<br><br> | ||
| Line 95: | Line 95: | ||
''' 5. Stability assay: '''<br> | ''' 5. Stability assay: '''<br> | ||
''' 6. Growth measurement: ''' <br><br> | ''' 6. Growth measurement: ''' <br><br> | ||
| + | |||
== K343005 == | == K343005 == | ||
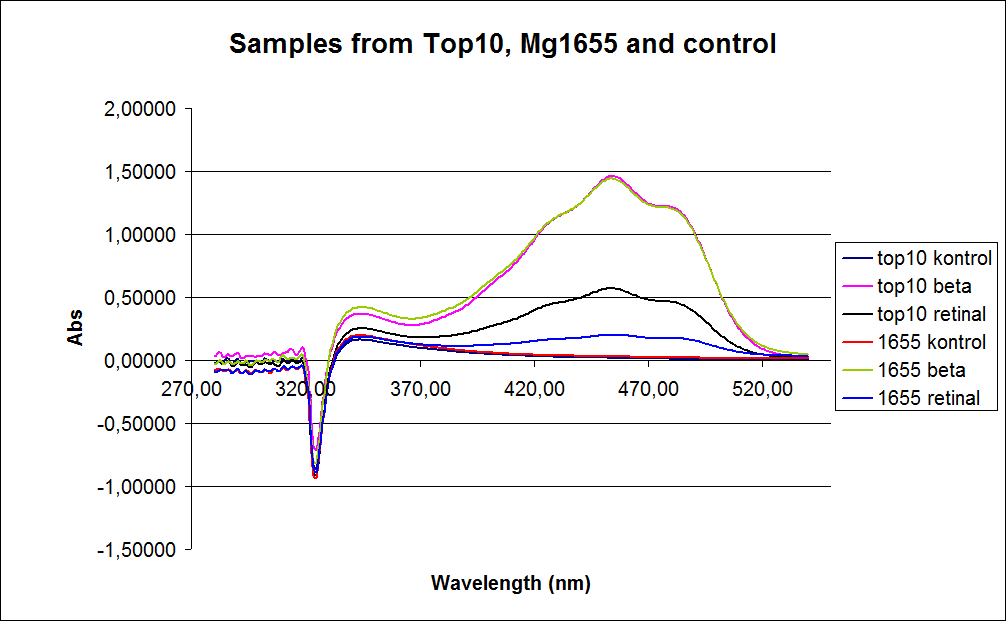
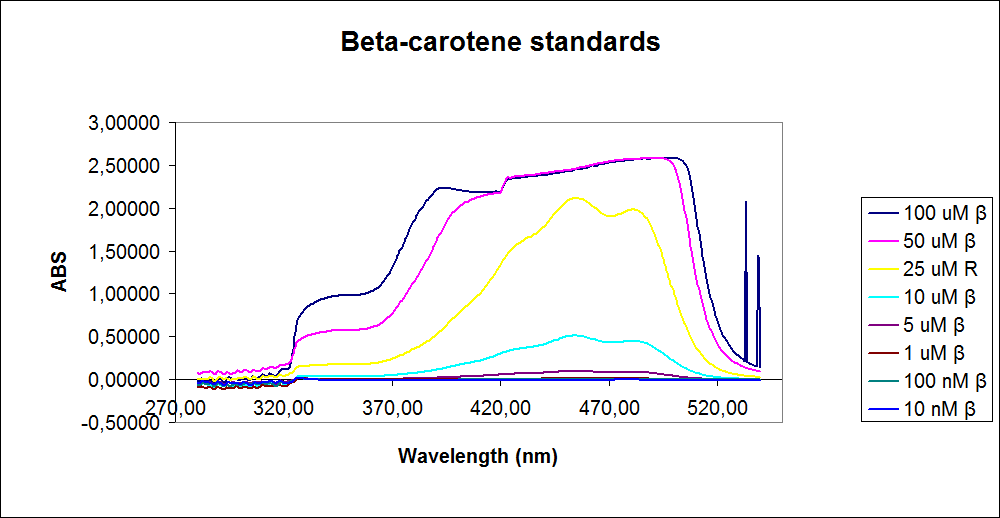
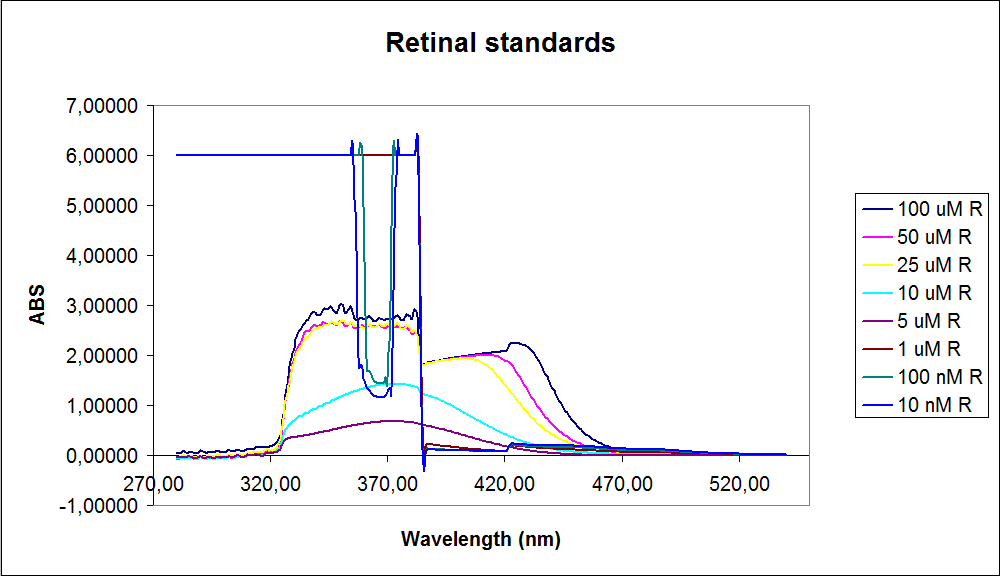
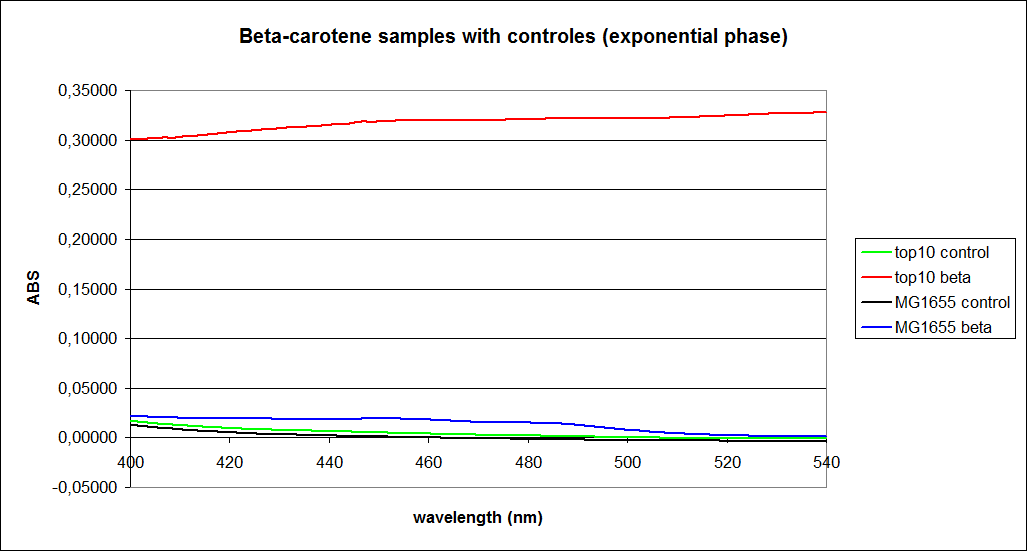
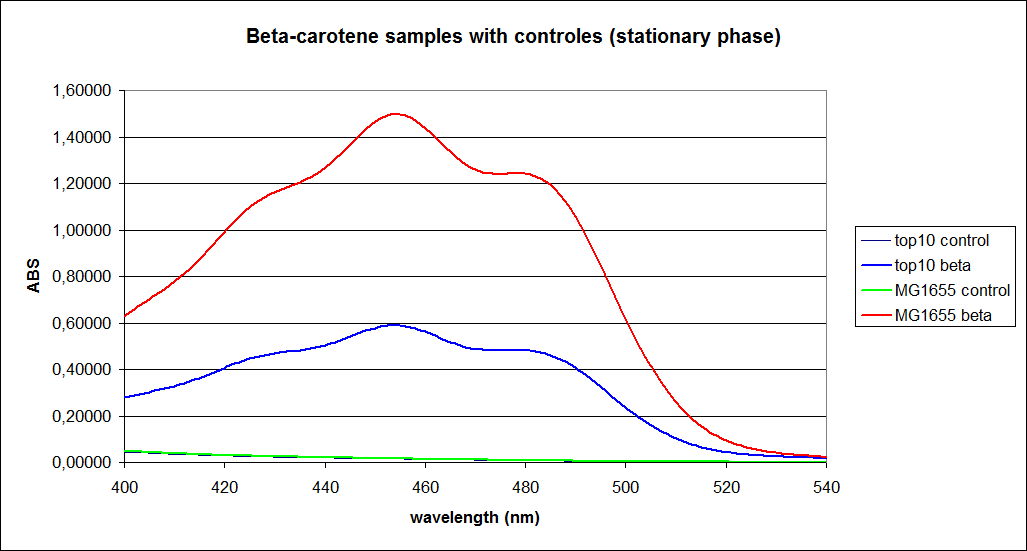
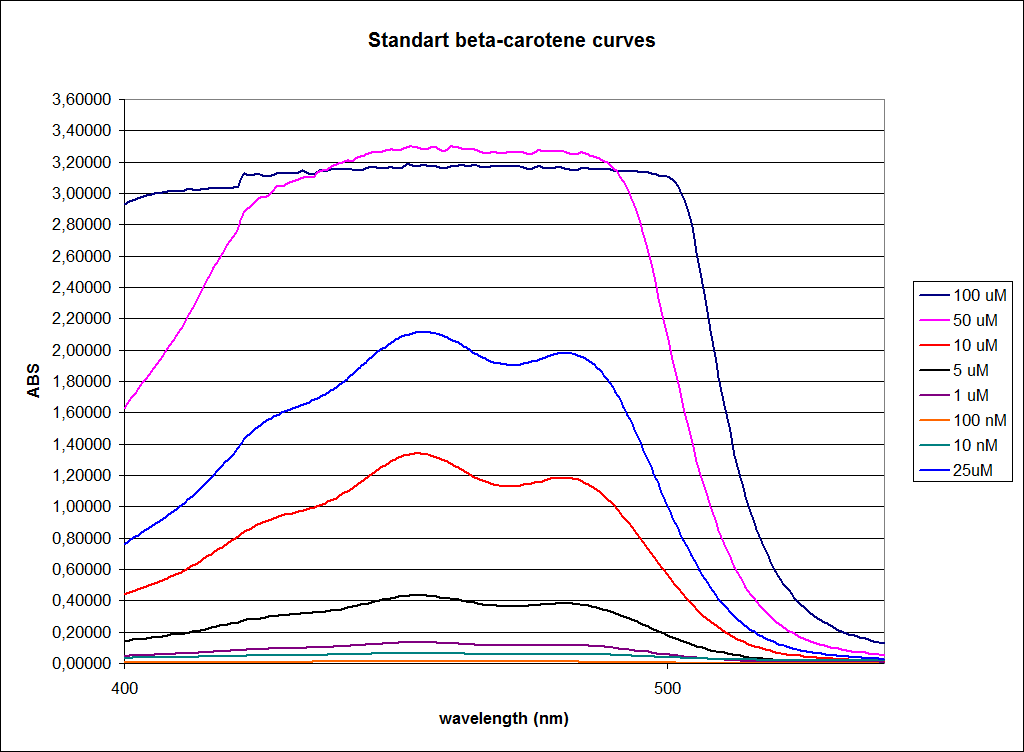
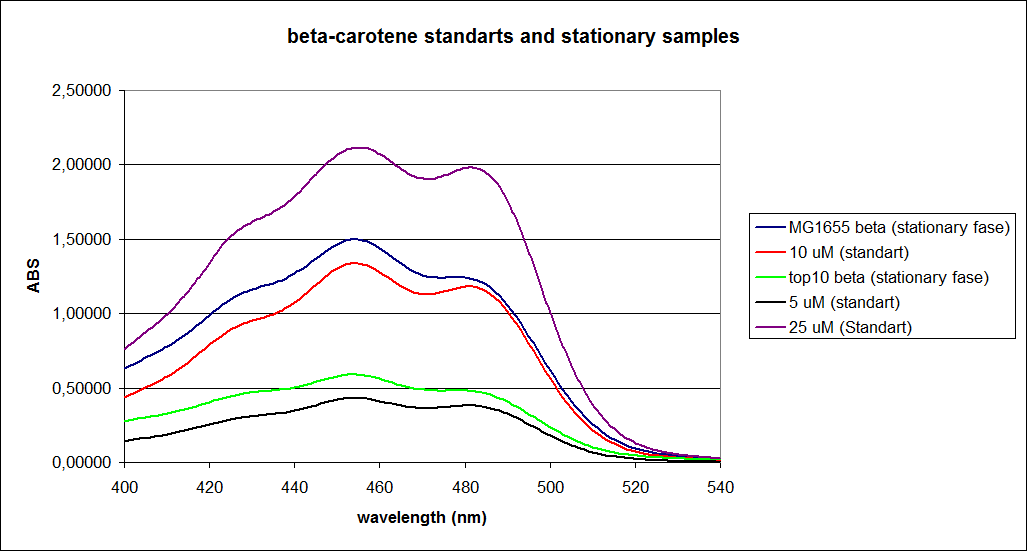
''' 1.UV-Vis determination of beta-carotene and retinal production: '''<br> | ''' 1.UV-Vis determination of beta-carotene and retinal production: '''<br> | ||
Revision as of 23:59, 24 October 2010
 "
"