Team:MIT mammalian Mechanosensation
From 2010.igem.org
The Search for Mechanosensitive Promoters |
|
Literature Search To begin, we searched the literature for well-characterized mechanosensitive promoters; we restricted our pool of candidates to promoters that had been shown, mostly by upregulation of a fluorescent marker, to be directly responsive to mechanical stress. The nature of the promoters varied. We included several short oligonucleotide sequences SRE/CRE2 and NR1/2, that had been shown to confer sensitivitiy to shear stress when placed in front of a ‘dummy’ SV40 promoter. Promoters for the genes PAI-2 and cfos were identified as mechanosensitive in the literature; we used portions of these promoters which were shown to be responsible for the mechanosensitivity. Click here and here for more in-depth notes on our literature review. Promoter Cloning We used PCR to amplify the relevant regions of the PAI-2 and cfos promoters from human genomic DNA; the short oligonucleotide sequences SRE/CRE2 and NR1/2 were added in front of an SV40 ‘dummy’ promoter, with appropriate primers. We then used three-way Gateway recombination (see our new standardization for mammalian cloning) to construct a lentiviral plasmid that contained the promoter of interest controlling EGFP expression. We used these constructs to transiently transfect cells with in the presence of helper viral plasmids; this yielded a virus containing our construct of interest. We then infected HEK cells with these constructs and selected with the appropriate antibiotic for two weeks to create stable cell lines. Several of the experiments done below were carried out before creation of the stable cell lines; to quickly obtain cell lines containing the relevant construct, we transiently transfected cells with the construct and no viral helper plasmids. Using that protocol, the construct did not get integrated into the genome, but it allowed us to carry out short term characterization assays. Experimental Outlines We planed to tested the sensitivity of our candidate promoters to an array of mechanical stresses. To accomplish this, we created and tested cell lines containing fluorescent reporter constructs for each promoter, and observed changes in fluorescence after the cells were exposed to mechanical stimulation. We applied shear stress with two different methods; the first involved periodic vibration of the cell plate on automated shaker. For the second method, we worked with a students in Roger Kamm's lab to build microfluidic devices; we seeded cells inside the devices and flowed media through the channels to approximate fluid shear stress. Shaker Experiments Experimental Setup ****SHAKER EXPERIMENT DESCRIPTION****** Results 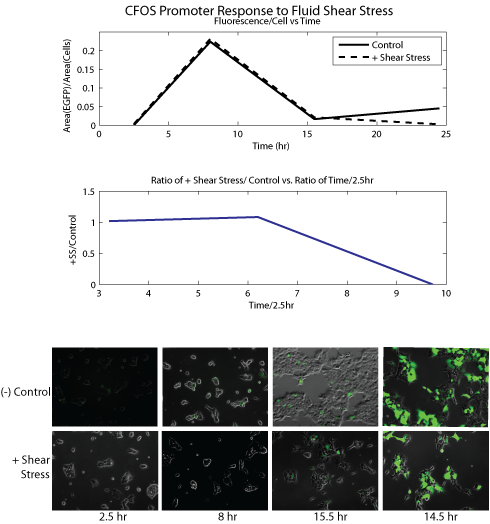


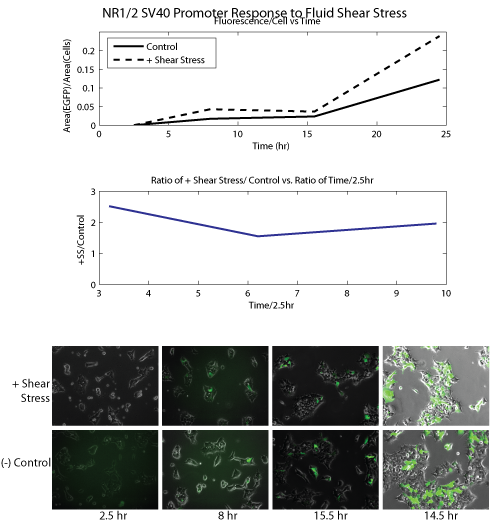 Conclusion
Conclusion We will use two different types of microfluidic devices, to assay the cellular response to fluid shear stress and cell layer ‘stretching’, respectively. However the two are not mutually exclusive; stretching of the cell layer could create interstitial flow, cross-activating the fluid shear stress response. |
 "
"
 iGEM 2010
iGEM 2010