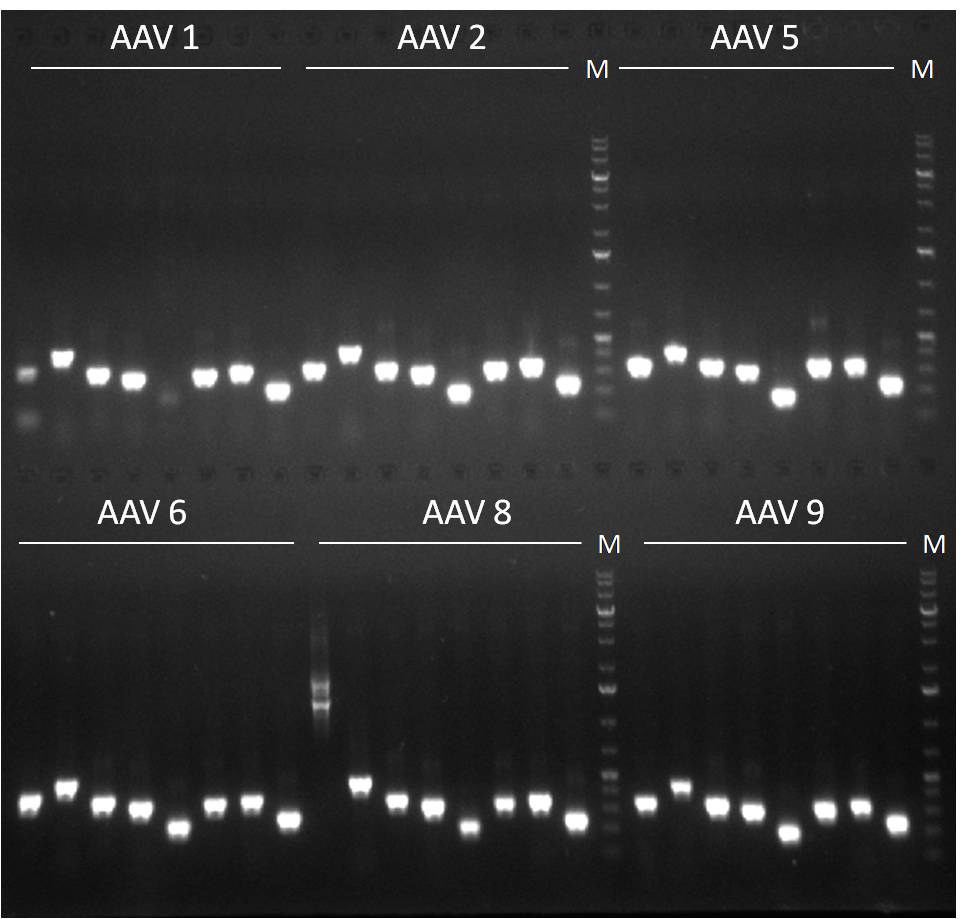
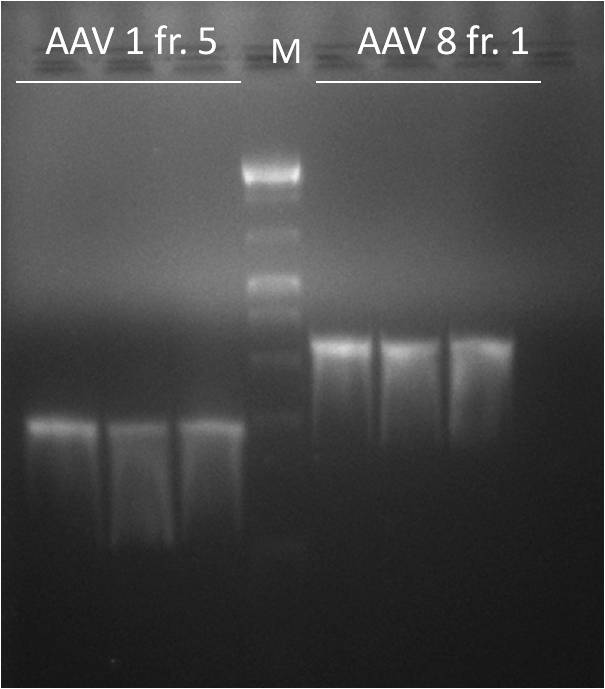
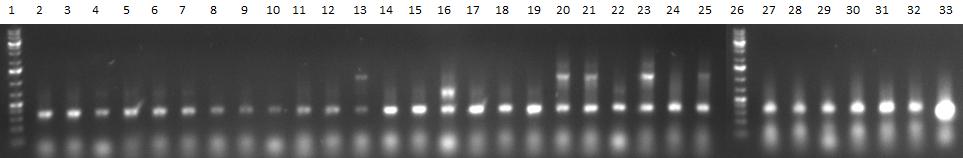
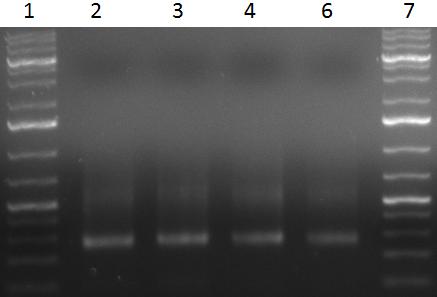
Team:Heidelberg/Notebook/ViroBytes/September
From 2010.igem.org
(Difference between revisions)
Laura Nadine (Talk | contribs) |
Laura Nadine (Talk | contribs) |
||
| Line 460: | Line 460: | ||
|- style="background:#f2f2f2; color:#78b41e" | |- style="background:#f2f2f2; color:#78b41e" | ||
|'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#26.2F10.2F2010 26]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#27.2F10.2F2010 27]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#28.2F10.2F2010 28]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#29.2F10.2F2010 29]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#22.2F30.2F2010 30]'''||colspan="3"| | |'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#26.2F10.2F2010 26]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#27.2F10.2F2010 27]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#28.2F10.2F2010 28]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#29.2F10.2F2010 29]'''||'''[https://2010.igem.org/Team:Heidelberg/Notebook/ViroBytes/October#22.2F30.2F2010 30]'''||colspan="3"| | ||
| + | |- style="background:#f2f2f2; color:#78b41e" | ||
| + | |colspan="7"| | ||
<span style="color:#ffffff">-</span> | <span style="color:#ffffff">-</span> | ||
|} | |} | ||
Revision as of 00:07, 27 October 2010

|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"