Safety
From 2010.igem.org
Resources
People

- Markus Schmidt
- Biosafety Working Group, IDC, Austria
IDC`s Biosafety Working Group is active in the field of management and use of plant genetic resources, risk assessment of new biotechnologies, and safety and security issues of synthetic biology.
Reports
 Schmidt M. 2009. Do I understand what I can create? Biosafety issues in synthetic biology. Chapter 6 in: Schmidt M. Kelle A. Ganguli A, de Vriend H. (Eds.) 2009. Synthetic Biology. The Technoscience and its Societal Consequences. Springer Academic Publishing
Schmidt M. 2009. Do I understand what I can create? Biosafety issues in synthetic biology. Chapter 6 in: Schmidt M. Kelle A. Ganguli A, de Vriend H. (Eds.) 2009. Synthetic Biology. The Technoscience and its Societal Consequences. Springer Academic Publishing
 HSE 2009. The SACGM Compendium of guidance Part 2: Risk assessment of genetically modified microorganisms
HSE 2009. The SACGM Compendium of guidance Part 2: Risk assessment of genetically modified microorganisms
 Tucker JB and Zilinska,s RA. 2006. The Promise and Perils of Synthetic Biology . The new Atlantis. Spring 2006, p.25-45
Tucker JB and Zilinska,s RA. 2006. The Promise and Perils of Synthetic Biology . The new Atlantis. Spring 2006, p.25-45
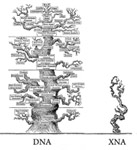 Schmidt M. 2010. Xenobiology: a new form of life as the ultimate biosafety tool . Bioessays 32:322-331
Schmidt M. 2010. Xenobiology: a new form of life as the ultimate biosafety tool . Bioessays 32:322-331
 Schmidt M, 2008. Diffusion of synthetic biology: a challenge to biosafety. Systems and Synthetic Biology. Vol.2(1-2):1-6
Schmidt M, 2008. Diffusion of synthetic biology: a challenge to biosafety. Systems and Synthetic Biology. Vol.2(1-2):1-6
 |
 |
 |
 |
 |
Intro
According to the WHO biosafety is the prevention of unintentional exposure to pathogens and toxins, or their accidental release, whereas biosecurity is the prevention of loss, theft, misuse, diversion or intentional release of pathogens and toxins.
Key questions
For iGEM 2010 teams are asked to detail how they approached any issues of biological safety associated with their projects. Specifically, teams should consider the following questions:
|
|
Teams, please document any answers to these safety questions on your wiki safety page. Judges will be asked to evaluate your project, in part, on the basis of if and how you considered and addressed issues of biological safety. If any questions arise regarding iGEM and biological safety please send an email to safety AT igem.org.
1) Identifying safety issues in your project:
The factors of interest in a risk assessment dealing with biological material include: pathogenicity, route of transmission, agent stability, infectious dose, concentration, origin of the potentially infectious material, availability of information, availability of an effective prophylaxis, availability of medical surveillance, experience and skill level of at-risk personnel. Your iGEM project is usually working with a non-infectious host organism (Biosafety level 1 or 2) so you may concentrate more on the engineered parts, devices and systems.
From an engineering and scientific point of view, risk assessment deals with the probability that a certain hazard is going to happen. In risk assessment: Risk = probability x hazard
Probability:
- Could there be an unplanned event or series of events involving your project, resulting in either death, injury, occupational illness, damage to equipment or property, or damage to the environment? How likely is that going to happen?
- Does your project require the exposure or release of the engineered organism to people or the environment (e.g. as medicine, for bioremediation)?
Hazard:
- Could your device, when working properly, represent a hazard to people or the environment?
- Is your engineered organism infectious? Does it produce a toxic product? Does it interfere with human physiology or the environment?
- What would happen if one or several bioparts change their function or stop working as intended (e.g. through mutation)? How would the whole device or system change its properties and w hat unintended effects would result thereof?
- What unintended effects could you foresee after your engineered organism is released to the environment?
- Try to think outside the box, what is the absolut worst case scenario for human health or the environment, that you could imagine?
Risks need to be seen in conjunction with the benefits. Although we would like to decrease the risk to absolute zero, this is hardly possible. So the question is not so much if something is safe or not, but rather if it is safe enough! Deciding whether a risk is acceptable or safe enough is no easy task and people may have different opinions. A whole professional field, so called risk management deals with that issue.
2) Documentation and management of safety issues
Datasheets on registered biobricks already contain some but few information on safety. For example, reliability of parts is be included, distinguishing genetic reliability and performance reliability that describe the number of generations it takes to cripple 50% of the circuits in the cells. This is a first step towards a more comprehensive safety characterization of biological circuits, but more detailed safety characterizations will be necessary to do a proper risk assessment to decide whether or not a device is safe enough for your particular application. Your contributions to documenting safety issues in parts, devices and systems are therefore greatly appreciated!
Here are some examples how you could document your work:
- Parts: Most bioparts will not pose any safety problems, but some can. The simplest example would be a part that encodes for a toxic protein (e.g. Botox, botulinum toxin, or ricin WIKI link). Other parts may produce milder toxins or anaphylatoxins (causing allergic reactions in some people). The fact that a protein can be toxic doesn't automatically mean that you cannot use it, some proteins are helpful pharmaceuticals in lower doses but become toxic in higher doses. In general the safety categorization of parts would best be based on the conventional BSL 1 to 4 levels and Select Agents and Toxins list (see e.g. the HHS AND USDA Select Agents AND TOXINS list http://www.selectagents.gov/Select%20Agents%20and%20Toxins%20List.html).
- Devices and systems: a genetic circuit could exhibit different safety characteristics than the parts it is based upon. Thus different safety categories should also be used for devices and systems.
- Cell chassis enhancement: Parts that extend the environmental range of a cell chassis, by increasing for example the tolerance of relevant biotic and abiotic conditions, should be documented as well.
Other questions are: How can a safety issue be reported that was discovered in a certain bio-circuit and that was not foreseen (emergent) so other people can learn from that experience? How can safety and security aspects be integrated into the design process so the design software automatically informs the designer in case the newly designed circuit exhibits certain safety problems?
3) Playing by the rules:
There is already a number of international and national guidelines, laws and professional associations that you have to consider. Here is an overview of some of them:
International
- World Health Organisation (WHO)
- Convention on Biological Diversity
USA
- National Institute of Health (NIH)
- American Biological Safety Association
- Centre for Disease Control (CDC)
Europe
- European Commission
- Directive 2009/41/EC on the contained use of genetically modified micro-organisms
- Directive 2001/18/EC on the deliberate release into the environment of genetically modified organisms
- Directive 2001/18/EC concerning the traceability and labelling of genetically modified organisms and the update Regulation 1830/2003
- Regulation 1829/2003 on genetically modified food and feed
- Regulation (EC) No 1946/2003 on transboundary movements of genetically modified organisms
- European Biosafety Association EBSA
- Switzerland
- Verordnung über den Umgang mit Organismen in der Umwelt: Freisetzungsverordnung
- Verordnung über den Umgang mit Organismen in geschlossenen Systemen: Einschliessungsverordnung
- Germany
- Bundesamt für Verbraucherschutz und Lebensmittelsicherheit:
- Liste risikobewerteter Spender- und Empfängerorganismen für gentechnische Arbeiten (list of risk assessed donor and host organisms for genetic engineering)
- Zentrale Kommission für die Biologische Sicherheit
- Bundesamt für Verbraucherschutz und Lebensmittelsicherheit:
- United Kingdom
- Belgium
- Netherlands
- Commissie Genetische Modificatie COGEM
Asia
- PR China
- National Biosafety Office, Ministry of Environmental Protection
- Biosafety of GMOs
- Biosafety regulation on pathogenic microbes
- Japan
- Asia-Pacific Biosafety Association: A-PBA
- Singapure:
Africa
- Republic of South Africa
Latin America
- Brasil
- Associação Nacional de Biossegurança: ANBio
If you have additonal relevant info, send them to safety AT igem.org
4) Other ideas
4.1. Biosafety engineering
Synthetic biology holds the potential to make biology not only easier to engineer but also safer to engineer. In many established engineering disciplines (e.g. mechanical engineering, aviation, space flight, electronics, software) safety engineering is already an established subset of systems engineering. (System) safety engineering is an engineering discipline that employs specialized professional knowledge and skills in applying scientific and engineering principles, criteria, and techniques to identify and eliminate hazards, in order to reduce the associated risks. Safety engineering assures that a system doesn't pose a risk even when parts of it fail. This is more than needed in synthetic biology due to the evolutionary forces of biological systems. If synthetic biology is going to become the new systems engineering of biology, then it needs to establish an equivalent subset in safety engineering: biosafety engineering (Schmidt 2009).
Biosafety engineering could be practiced by designing robust genetic circuits that account for possible failure of single parts or subsystems, but still keep working or at least don't cause any harm to human health or the environment. Safety engineering has many techniques to design safer circuits (systems), for example
- Event Tree Analysis and
- Fault Tree Analysis
Both methods are normally used in assessing the safety of engineering systems (e.g. aircraft, space travel, mechanical engineering, nuclear energy) based on standardized parts and true engineering designs.
In a device or system, for example, a mutation in one of the bioparts could cause the part to become dysfunctional. The Event Tree Analysis (ETA) would look at the way the whole system is going to be affected by the failed part. It will answer the questions: Will the device or system still be able to fulfill its tasks? Will it behave in a different way, and if yes in which way? Or will it shut down completely? Based on this analysis additional safety systems could be installed, such as redundant sub-circuits.
The Fault Tree Analysis (FTA), on the other hand, looks at defined unwanted failures of the systems and then traces backward to the necessary and sufficient causes. For example, a genetic circuit should not fail in a way that leads to the overproduction of a particular protein that is regulated by the network. The FTA can show which basic events could cause such an overproduction, and thus help to improve the circuit to avoid these unwanted failure, for example in designing the circuit in a way that all basic events would cause the expression of the protein to diminish but never to increase.
The full range of possibilities to include safety considerations in designing biological circuits has not yet been explored in great detail but will be extremely helpful. How could you contribute to make it happen?
4.2. Designing and using a safer host organims/chassis
4.3. Public perception of risks and safety issues
As we work with a technology that is in the public eye, we need to understand that besides scientific risk ''assessment'', as described before, there is also a public risk ''perception'' of what we do. It is useful to understand the "soft facts" of risk perception that, especially in case of lay people, outdo the "hard facts" such as technical or medical expertise. Experts typically define risk strictly in terms of the probability of a certain damage (e.g. mortalities, life years lost, financial loss). Lay people, however, almost always include other factors in their definition of risk, such as catastrophic potential, equity, effects on future generations, controllability, involuntariness and trust in the people responsible. These differing conceptions often result in lay people assigning relatively little weight to risk assessments conducted by technical experts or government officials, instead they use these other factors to form an opinion. See table for some of the most relevant factors affecting risk perception:attenuate risk perception |
amplify risk perception | |
familiar |
↔ |
exotic |
individual control |
↔ |
controlled by others |
natural |
↔ |
manmade |
statistical |
↔ |
catastrophic |
clear benefits |
↔ |
little or no benefit |
fairly distributed |
↔ |
unfairly distributed |
voluntary |
↔ |
imposed |
information by trusted sources |
↔ |
information by untrusted sources |
trust in responsible persons/organisation |
↔ |
lack of trust |
not in the media |
↔ |
in the media |
How do you think synthetic biology, and especially the iGEM competition, is perceived by the public? What could you do to influence that?
There is already heaps of articles on risk perception, for an introduction see:
Schmidt M. 2004. Investigating risk perception: a short introduction. Chapter 3 PhD Thesis, Vienna, Austria
Sjöberg L. 2000. Factors in Risk Perception. Risk Analysis, Vol. 20, No. 1, pp.1-11
Slovic P. 1987. Perception of Risk. Science Vol. 236, pp. 280-285
If there is anything here that has caught your interest, bothered you or sparked an idea you would like to tell us, then get in touch with us. You can leave comments, thoughts and suggestions below but also feel free to contact us directly if you want something a little more interactive.
Responsible for pictures, links and content: Markus Schmidt
Photo Credits (from left to right)
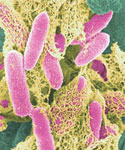
E. coli http://en.wikipedia.org/wiki/File:E-coli-in-color.jpg
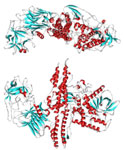
Botulinum toxin: http://en.wikipedia.org/wiki/File:Botulinum_toxin_3BTA.png
Working in a suit under biolevel 4 conditions. http://www.cihr-irsc.gc.ca/e/17766.html
 "
"