Team:Heidelberg/Project/miRNA Kit
From 2010.igem.org
(→general procedure overview) |
(→general procedure overview) |
||
| Line 91: | Line 91: | ||
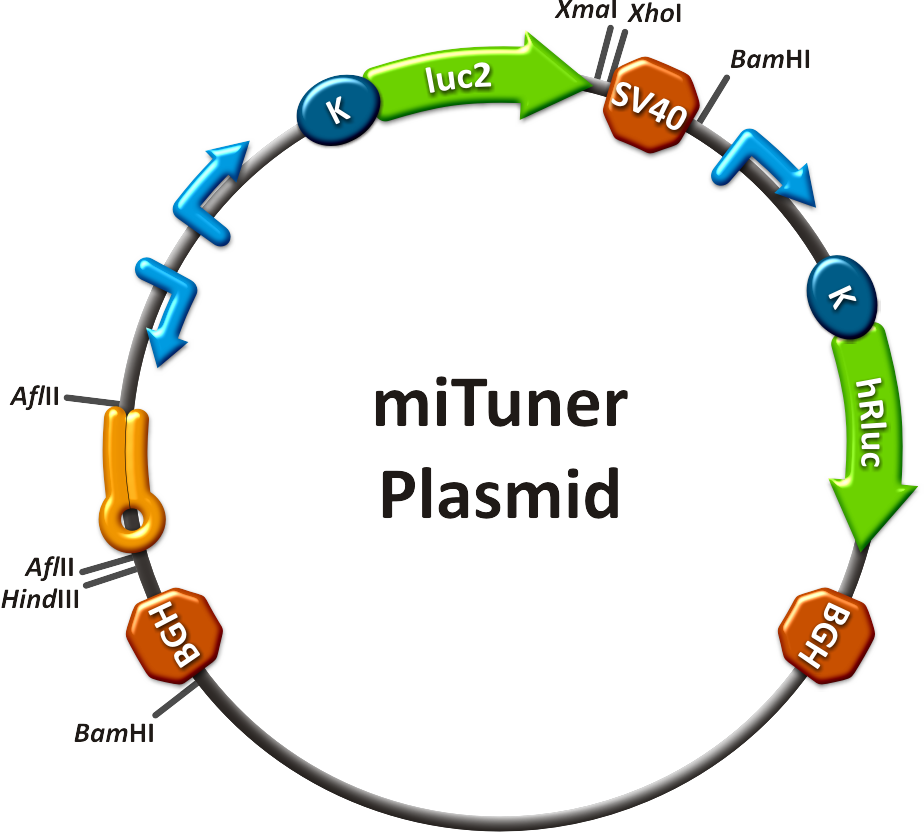
=== general procedure overview === | === general procedure overview === | ||
* choose an [[Team:Heidelberg/Project/Introduction | interesting microRNA]] | * choose an [[Team:Heidelberg/Project/Introduction | interesting microRNA]] | ||
| - | * | + | * create referring binding sites with the [https://2010.igem.org/Team:Heidelberg/Modeling/miBSdesigner miBS designer] |
* order your binding site oligos | * order your binding site oligos | ||
* [https://2010.igem.org/Team:Heidelberg/Notebook/Methods#Cloning clone] them into your [https://2010.igem.org/Team:Heidelberg/Parts#final_constructs miTuner construct] (one step) | * [https://2010.igem.org/Team:Heidelberg/Notebook/Methods#Cloning clone] them into your [https://2010.igem.org/Team:Heidelberg/Parts#final_constructs miTuner construct] (one step) | ||
Revision as of 22:19, 26 October 2010

|
|
||
 "
"