Tuesday, July 20 By: Wataru, Tomo, Yuki, Kazuya, Ken, Makoto
Solubilization of Antibiotics
| Ampicillin(Amp) | Kanamycin(Kan) |
|
| Mix 1.0g Amp and 20ml MilliQ | Mix 0.5g Kan and 10ml MilliQ | Final concentration is 50mg/ml
|
| Dispense 1.1ml of the solution into 1.5ml tubes |
|
| Store in the freezer (-20℃) |
|
Making plates for LB (Amp+) and LB (Kan+)
Wednesday, July 21 By: Wataru, Ken, Makoto, Takuya Y.
Culture of plates in which colonies was observed at 37℃ from 07/21 20:50 to 07/22 17:00
Making a master plate of the above plates
PCR for S-R-Rz/Rz1 and S
| No. | Water | 25mM MgSO4 | 2mM dNTPs | 10xBuffer for KOD Plus ver.2 | TemplateDNA (5ng/µl) | Primer Forward (10µM) | Primer S-R-Rz/Rz1 Reverse (10µM) | Primer S Reverse (10µM) | KOD Plus ver.2 | Total
|
| 1 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | 1.5µl | - | 1µl | 50µl
|
| 2 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | 1.5µl | - | 1µl | 50µl
|
| 3 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | - | 1.5µl | 1µl | 50µl
|
| 4 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | - | 1.5µl | 1µl | 50µl
|
| 5 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | 1.5µl | - | 1µl | 50µl
|
| 6 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | 1.5µl | - | 1µl | 50µl
|
| 7 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | - | 1.5µl | 1µl | 50µl
|
| 8 | 28µl | 3µl | 5µl | 5µl | 5µl | 1.5µl | - | 1.5µl | 1µl | 50µl
|
PCR condition
| 94℃ | 2min |
|
| 98℃ | 10sec | 30 cycles
|
| 55℃ | 30sec
|
| 68℃ | 4min
|
| 4℃ | forever |
|
Thursday, July 22 By: Wataru
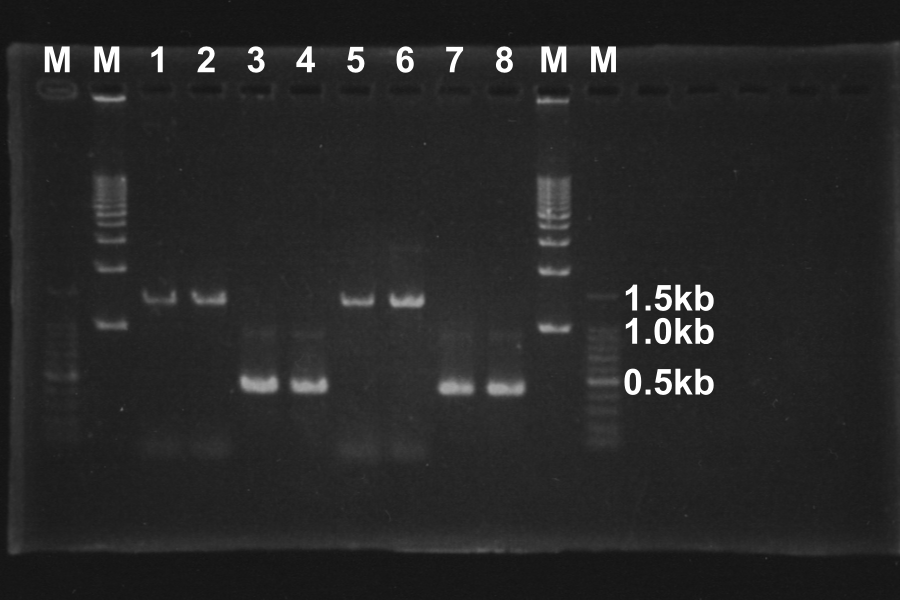
Length of S and S-R-Rz/Rz1 is 370bp and 1300bp, so PCR succeeded.
| Name | Concentration(ng/µl)
|
| <partinfo>J23100</partinfo> | 18.5
|
| <partinfo>J23105</partinfo> | 12.5
|
| <partinfo>J23116</partinfo> | 14.6
|
| <partinfo>R0011</partinfo> | 8.6
|
| <partinfo>E0840</partinfo> | 12.1
|
| <partinfo>J06702</partinfo> | 14.7
|
The concentration of all samples was very week. Probably our shaking incubation was week.
Culture of plates and making master plates of <partinfo>pSB4K5</partinfo> and <partinfo>B0015</partinfo> from 07/22 17:00 to 07/23 10:00
Friday, July 23 By: Wataru, Tomo, Makoto
| Name | Concentration(ng/µl)
|
| <partinfo>pSB4K5</partinfo> | 79.2
|
| <partinfo>B0015</partinfo> | -
|
We lost <partinfo>B0015</partinfo> by our mistake. The concentration of <partinfo>pSB4K5</partinfo> is high, so this condition of shaking incubation is moderate.
Picking up number 1, 3, 5, and 7 of the products of PCR, and PCR-purification
| No. | Name | Concentration (ng/µl) | New Name
|
| 1 | S-R-Rz/Rz1 | 18.6 | -
|
| 3 | S | 77.6 | S1
|
| 5 | S-R-Rz/Rz1 | 33.6 | -
|
| 7 | S | 65.4 | S2
|
The concentration of sample number 1 and 5, the PCR products of S-R-Rz/Rz1, is week, so we desided to retry PCR.
| No. | Water | 25mmol/l MgSO4 | 2mmol/l dNTPs | 10×Buffer for KOD plus ver.2 | Template DNA (5ng/µl) | Primer S-R-Rz/Rz1 Forward (10µmol/l) | Primer S-R-Rz/Rz1 Reverse (10µmol/l) | KOD plus ver.2 | Total
|
| 1 | 28µl | 3 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
| 2 | 28 | 3 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
| 3 | 26.5 | 4.5 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
| 4 | 26.5 | 4.5 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
| 5 | 25 | 6 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
| 6 | 25 | 6 | 5 | 5 | 5 | 1.5 | 1.5 | 1 | 50
|
PCR condition
| 94℃ | 2min |
|
| 98℃ | 10sec | 30 cycles
|
| 55℃ | 30sec
|
| 68℃ | 4min
|
| 4℃ | forever |
|
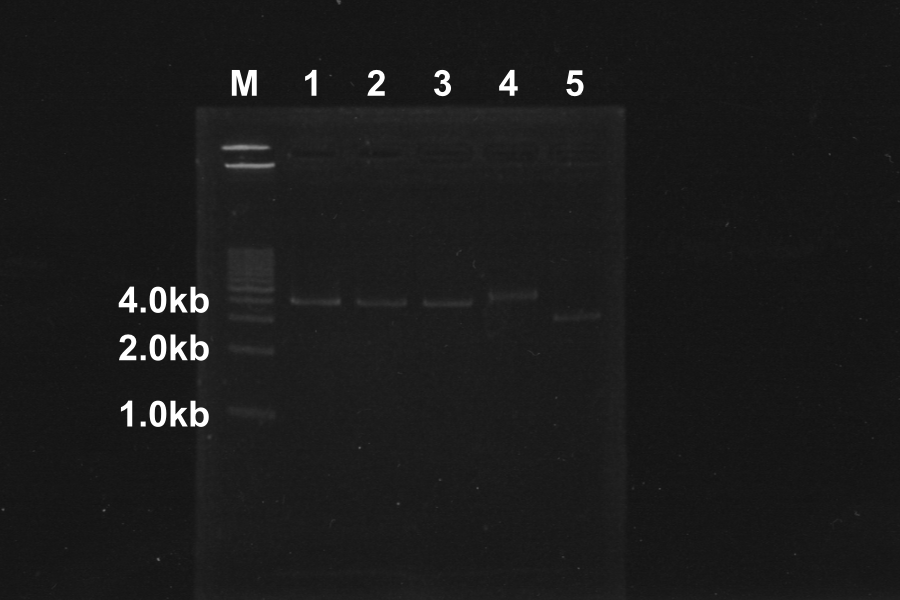
Restriction Digestion of <partinfo>J06702</partinfo> by EcoRI, XbaI, SpeI, and PstI to check function of our Restriction Enzyme
| No. | 10xBuffer | BSA | Enzyme | MilliQ | Total | Incubation
|
| 1 | 5µl | 1 | EcoRI 0.1 | 3.6 | 10 | At 37℃ 7/23 18:00 - 7/23 18:30
|
| 2 | 5 | 1 | XbaI 0.1 | 3.6 | 10
|
| 3 | 5 | 1 | SpeI 0.1 | 3.6 | 10
|
| 4 | 5 | 1 | PstI 0.1 | 3.6 | 10
|
| 5 | 5 | 1 | - | 3.7 | 10
|
Comparison to sample 5(control, circular DNA), the bands of sample 1, 2, 3, 4 was shifted. The DNA of sample 1, 2, 3, 4 was linearized by Restriction enzymes. So, our restriction enzymes work correctly.
Digestion of the PCR products of S gene by EcoRI and SpeI and <partinfo>E0840</partinfo> by EcoRI and XbaI to insert S gene to <partinfo>E0840</partinfo>
| Name | Sample Volume (µl) | 10×Buffer | Enzyme 1 | Enzyme 2 | MilliQ | Total | Incubation
|
| S1 | 11 | 5 | EcoRI 0.2 | SpeI 0.2 | 33.6 | 50 | At 37℃ for 2h
|
| S2 | 11 | 5 | EcoRI 0.2 | SpeI 0.2 | 33.6 | 50
|
| <partinfo>E0840</partinfo> | 45 | 5 | EcoRI 0.2 | XbaI 0.2 | 0 | 50
|
After PCR purification, evaporated them and diluted 3ul.
| Name | Vector | Insert | Ligation High | Total
|
| S-E08401 | <partinfo>E0840</partinfo> 0.5µl | S1 0.5 | 1 | 2
|
| S-E08402 | <partinfo>E0840</partinfo> 0.5 | S2 0.5 | 1 | 2
|
 "
"