Team:Stockholm/10 September 2010
From 2010.igem.org
Revision as of 09:59, 13 September 2010 by AndreasConstantinou (Talk | contribs)
Contents |
Andreas
Cloning of N-CPPs into pSB1C3 & Extraction of RBS BioBrick (BBa_B0030)
Transformations from 9/9 resulted in good colony yields on all plates. Chose "pSB1C3.N-CPP* 9/9" for colony PCR.
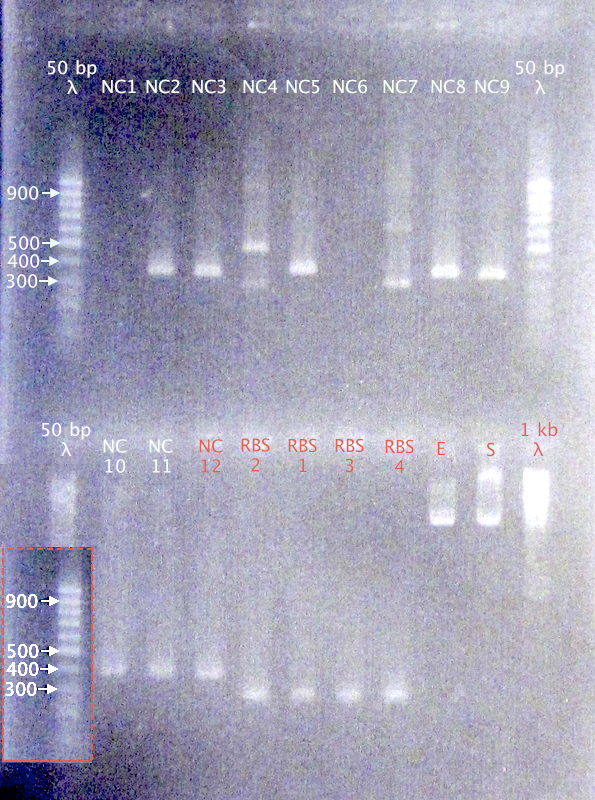
Colony PCR
Picked 12 N-CPP* clones (NC 1-12) and 4 RBS clones (RBS 1-4)
| PCR tubes | |
|---|---|
| dH2O | 16.22 |
| DreamTaq buffer | 2 |
| dNTPs, 10 mM | 0.4 |
| VF2 | 0.4 |
| VR | 0.4 |
| DreamTaq pol. | 0.08 |
| Template DNA | 0.5 |
| 20 μl | |
PCR settings
Standard colony PCR protocol.
- 1:00 elongation
Gel verification
Also ran two samples for Mimmi (E & S)
1.5 % agarose, 90 V
Expected bands
- N-Tra10: 389 bp
- N-TAT: 359 bp
- N-LMWP: 368 bp
- RBS B0030: 253 bp
Results
- N-CPPs: Potentially correct bands for clones 2, 3, 5, 8, 9, 10, 11 & 12
- RBS B0030: Correct-sized bands for all four clones.
ON cultures
Set ON cultures for all relevant N-CPPs, for plasmid prep.
- N-CPP 2, 3, 5, 8, 9, 10, 11, 12
- 5 ml LB + 25 Cm
- 37 °C, 220 rpm
Selected clone 4 of RBS 30. Both plasmid prep and glycerol stock.
- RBS 30 4
- 5 ml LB + 100 Amp
- 37 °C, 220 rpm.
- 3 ml LB + 100 Amp
- 30 °C
- 5 ml LB + 100 Amp
Preparation of chemically competent Top10
Since I've previously experienced slight AmpR contamination in our latest batch of competent Top10, I streaked an LB agar plate with competent Top10 cells to isolate single clones.
Plate grown ON in 37 °C.
 "
"