|
|
|
JUNE: WEEK 4
June, 21st
Planning of the activity of the week.
Freezer cleaning: for each ligation, we choosed the correct clone and stored it in the iGEM 2010 ligations box. All these cloned were gel screened and sequenced and re correct!
The colonies we choosed are:
| colony choosed |
ligation name |
| I0-2 | I0 |
| I1-2 | I1 |
| I2-1 | I2 |
| I3-1 | I3 |
| I4-2 | I4 |
| I5-1 | I5 |
| I6-2 | I6 |
Other colonies resulted psitives at the screening but were NOT sequenced and are stored in the iGEM 2010 cemetery box. These clones are: I1-1, I2-2, I4-1 and I6-3.
June, 22nd
Inoculum of I6, <partinfo>BBa_J23118</partinfo>, <partinfo>BBa_J23110</partinfo>, <partinfo>BBa_J23114</partinfo>, <partinfo>BBa_J23116</partinfo> from glycerol stock in 5ml LB+Amp. Cultures were grown ON at 37°C 220rpm.
June, 23rd
Cultures incubated ON were all grown. Plasmids were extracted with MiniPrep.
After MiniPrep, purified DNA was quantified with NanoDrop.
| I6 | 193 ng/ul
|
| <partinfo>BBa_J23118</partinfo> | 107,9 ng/ul
|
| <partinfo>BBa_J23110</partinfo> | 83 ng/ul
|
| <partinfo>BBa_J23114</partinfo> | 89,8 ng/ul
|
| <partinfo>BBa_J23116</partinfo> | 82,7 ng/ul
|
|
Digestion of:
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 | Enzyme 2 | Buffer H
|
| I6 | Insert | 25 | 9,3 | 11,2 | 1 XbaI | 1 PstI | 2,5
|
| I6-bis | Insert | 25 | 9,3 | 11,2 | 1 XbaI | 1 PstI | 2,5
|
| <partinfo>BBa_J23118</partinfo> | Vector | 25 | 9,3 | 11,2 | 1 SpeI | 1 PstI | 2,5
|
| <partinfo>BBa_J23110</partinfo> | Vector | 25 | 12 | 8,5 | 1 SpeI | 1 PstI | 2,5
|
| <partinfo>BBa_J23114</partinfo> | Vector | 25 | 11,2 | 9,3 | 1 SpeI | 1 PstI | 2,5
|
| <partinfo>BBa_J23116</partinfo> | Vector | 25 | 12,1 | 8,4 | 1 SpeI | 1 PstI | 2,5
|
Digestions were incubated at 37°C for 3 hours, gel run and gel-extracted.
Ligations were performed ON at 16°C:
- I7: <partinfo>BBa_J23118</partinfo> (S-P)+ I6 (X-P)
- I8: <partinfo>BBa_J23110</partinfo> (S-P)+ I6 (X-P)
- I9: <partinfo>BBa_J23114</partinfo> (S-P)+ I6 (X-P)
- I10: <partinfo>BBa_J23116</partinfo> (S-P)+ I6 (X-P)
NanoDrop quantifications were not reliable, so every ligation was performed with 3ul of vector, 2ul of insert, 3ul of ddH20, 1ul of T4 ligase and 1ul of T4 ligase buffer.
These four promoters from Andersone Promoters Collection were choosed for their strength, measured in Arbitrary Units:
| Part | RFP (a.u.)
|
| <partinfo>BBa_J23118</partinfo> | 1429
|
| <partinfo>BBa_J23110</partinfo> | 844
|
| <partinfo>BBa_J23114</partinfo> | 256
|
| <partinfo>BBa_J23116</partinfo> | 396
|
Soon quantitative tests will be performed to quantify each promoter's strength in terms of RPU.
Today we received 2 stabs from registry HQ for <partinfo>BBa_K208001</partinfo> (one for each registry loation of this part),the Silver-fusion compatible BioBrick part that codes for phasin. This part is in a pSB3K3 plasmid is in E. coli ??? . Cultures were straked on LB+Kan (50 ug/ml) agar plates. Cultures were named PhaP-1 and PhaP-2. Plates were incubated ON at 37°C.
June, 24th
PhaP-1 and PhaP-2 plates showed colonies!!
A colony from each plate was peaked and used to infect 1ml LB+Kan. Liquid cultures of PhaP-1 and PhaP-2 were grown for 6 hours at 37°C 220 rpm and then used to prepare glycerol stocks. Remaining liquid cultures were re-filled with 5 ml LB+Kan an grown ON at 37°C 220 rpm for tomorrow MiniPrep.
Glycerol stocks of PhaP-1 and PhaP-2 are stored at -80°C in the iGEM 2010 Registry box.
Ligations were heated at 65°C for 5 minutes to inactivate ligase ad then tranformed in E. coli TOP10 home made competent cells. Plates were incubated ON at 37°C (we decided to incubate plates 5 hours longer than usual, because cell grotwh was already slower for I6).
June, 25th
All plates showed colonies after 19 hours at 37°C!!
- I7: small green colonies with satellite colonies were observed. Negative colonies are present and can be easily distinguished, becaue they are red.
- I8:green colonies were observed, few negative colonies are pink.
- I9: big colonies were observed, some of them are surrounded by satellite colonies.
- I10: big colonies were observed, some light pink colonies are present, surrounded by satellite colonies.
These results are encouraging, because they are consistent with our expectations: ligations provide a high metabolic burden to the cell, so growth is slower. Positive green colonies are smaller and not surrounded by satellite colonies. Negative colonies are bigger and surrounded by satellite colonies, confirmingthat they had a faster growth than our ligations!
2 colonies from each plate were peaked and incubated in 1ml LB+Amp.
| culture |
| I7-1 |
| I7-2 |
| I8-1 |
| I8-2 |
| I9-1 |
| I9-2 |
| I10-1 |
| I10-2 |
|
Cultures were grown for 6 hour at 37°C 220 rpm, then glycerol stockes were prepared for each culture. Glycerol stocks are stoed at -80°C in iGEM 2010 ligations box.
|
PhaP-1 and PhaP-2 cultures were grown. MiniPre was performed, with the following NanoDrop quantifications:
| PhaP-1 | 9,6 ng/ul
|
| PhaP-2 | 4,3 ng/ul
|
DNA quantification was poor, so we decided to perform digestion screening and to repeat Miniprep next week for sequencing.
Digestion of:
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 | Enzyme 2 | Buffer H
|
| PhaP-1 | Screening | 25 | 20,5 | 0 | 1 EcoRI | 1 PstI | 2,5
|
| PhaP-2 | Screening | 25 | 20,5 | 0 | 1 EcoRI | 1 PstI | 2,5
|
Digestion eas performed for 2 hours at 37°C. DNA was then gel run for screening. Both cultures were positives! Sequencing will be performed next week!
 Screening of PhaP-1 and PhaP-2
June, 28th
Inoculum from glycerol stock for I7-1, I7-2, I8-1, I8-2, I9-1, I9-2, I10-1 and I10-2 in 5ml LB+Amp. Cultures were gown ON at 37°C 220rpm.
Inoculum of PhaP-1 and PhaP-2 in 5ml LB+Kan and ON growth at 37°C 220rpm.
We received strains and plasmid from ???Yale University???
| name | description | growth conditions
|
| BT340 | pCP20 plasmid | LB+Amp LC 30°C,LB+Amp HC 30°C
|
| BW23473 | E. coli pir+ strain | LB, 37°C
|
| BW23474 | E. coli pir116 strain | LB, 37°C
|
| BW25141 | E. coli pir+ strain | LB, 37°C
|
| BW25142 | E. coli pir116 strain | LB, 37°C
|
| BW5328 /pAH123 | helper plasmid | LB+Amp LC 30°C,LB+Amp HC 30°C
|
| MC1061 | E. coli strain for integration | LB, 37°C
|
| MG165 | E. coli wil type strain | LB, 37°C
|
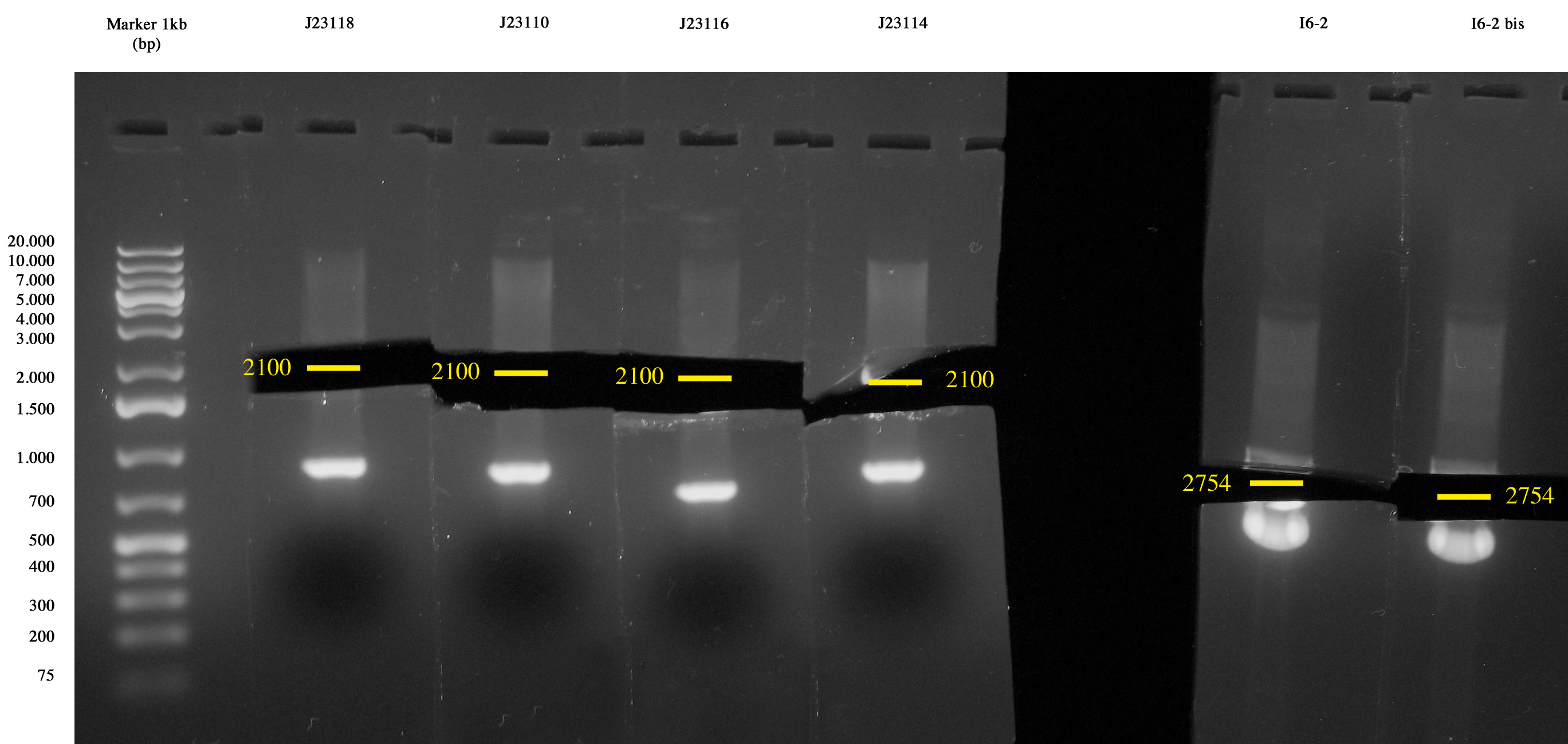
 Screening of I7, I8, I9, I10 June, 29th
June, 30th
July, 1st
July, 2nd
July, 5th
|
|
 "
"