|
AUGUST: WEEK 4
August, 23rd
Colony PCR as screening for ligations I31..I41. For each plate three colonies were picked and PCR amplification was performed. At the same time colonies were let grow in 750 ml LB+Amp in order to be ready to make glycerol stocks for those with right amplicons.
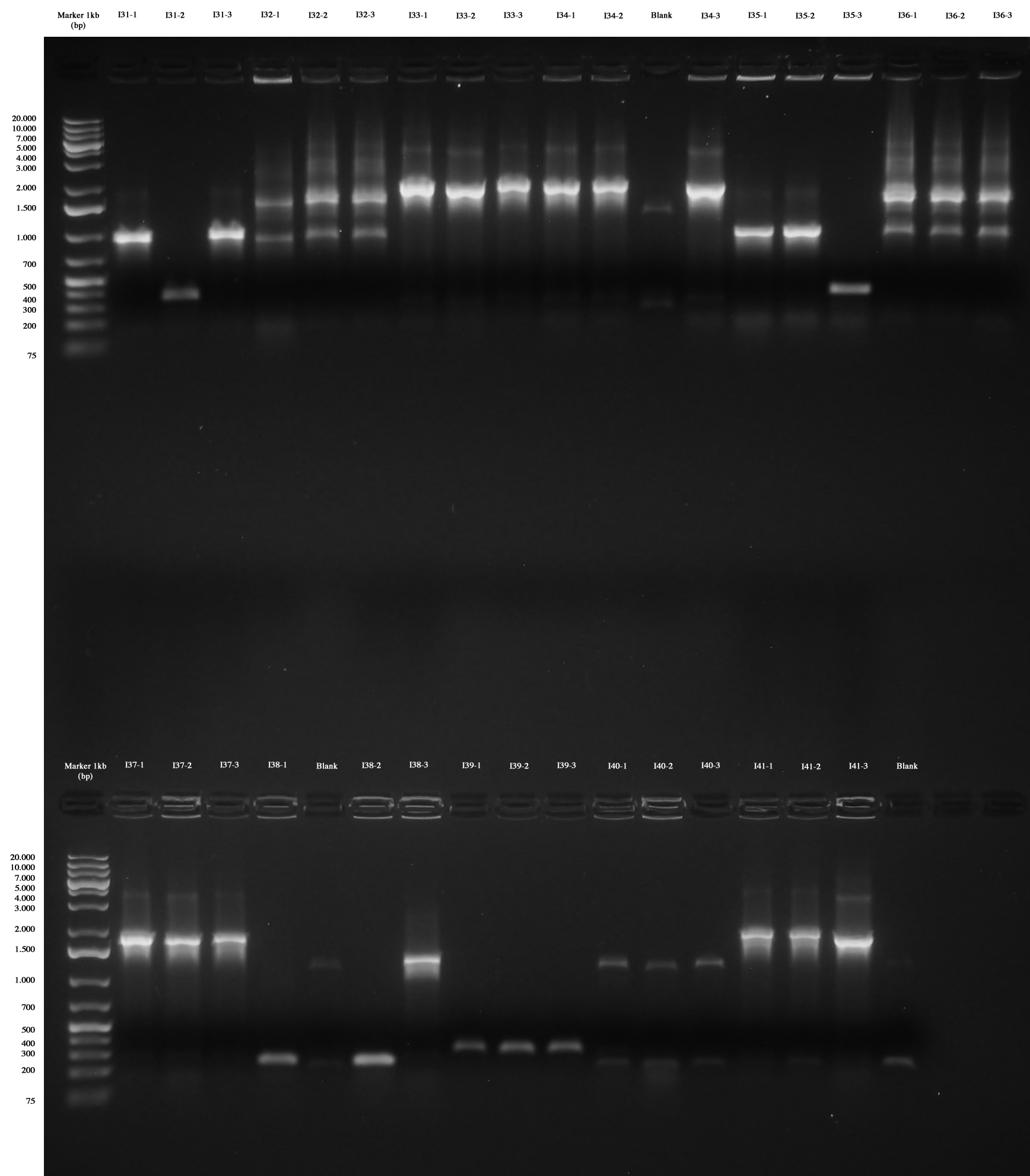 Colony PCR and gel run as screening for ligations I31..I41 Only I31, I33, I34, I35, I37, I38, I41 are positive without any doubt (we took I31-1, I33-1. I34-1, I35-1, I37-1, I38-3, I41-1), while I39 were all wrong and we weren't sure about I32, I36 and I40 (we made however glycerol stocks for I32-2, I36-2, I40-1).
We decided to repeat colony PCR for I32, I36, I39 and I40 next day.
MyCrim-9, <partinfo>BBa_K173001</partinfo>, <partinfo>BBa_J23101</partinfo>, <partinfo>pSB4C5</partinfo> were digested E-P and MyCrim-9, Ring were digested HindIII for three hours.
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 (ul) | Enzyme 2 (ul) | Buffer (ul)
|
| MyCrim | Vector | 25 | 6,5 | 14 | 1 EcoRI | 1 PstI | 2,5 H
|
| MyCrim | Vector/Screening | 25 | 4 | 16,5 | 1 HindIII | 1 HindIII | 2,5 B
|
| BBa_K173001 | Insert | 25 | 8 | 12,5 | 1 EcoRI | 1 PstI | 2,5 H
|
| BBa_J23101 | Insert | 25 | 5 | 15,5 | 1 EcoRI | 1 PstI | 2,5 H
|
| pSB4C5 | Insert | 25 | 5 | 15,5 | 1 EcoRI | 1 PstI | 2,5 H
|
| Ring | Vector/Screening | 25 | 4 | 16,5 | 1 HindIII | 1 HindIII | 2,5 B
|
Gel extraction of <partinfo>BBa_I52002</partinfo>(E-P) (insert of pSB4C5), MyCrim (E-P), BBa_K173001 (E-P), MyCrim (HindIII), vector of pSB4C5.
Quantification:
| Sample | Quantifications"
|
| MyCrim (HindIII) | 13 ng/ul
|
| MyCrim (E-P) | 23,4 ng/ul
|
| BBa_K173001 (E-P) | 11 ng/ul
|
| BBa_J23101 (E-P) | 9 ng/ul
|
| pSB4C5 vector (E-P) | 23 ng/ul
|
| BBa_I52002 (insert of pSB4C5) (E-P) | 6 ng/ul
|
These parts were used to perform following ligations:
| Name | Vector | Insert
|
| I42 | MyCrim (E-P) | BBa_K173001 (E-P)
|
| I43 | MyCrim (E-P) | BBa_J23101 (E-P)
|
| I44 | MyCrim (E-P) | BBa_I52002 (insert of pSB4C5) (E-P)
|
| I45 | MyCrim (HindIII) | MyCrim (HindIII)
|
August, 24th
Only this morning we remembered that I40 is in a commercial vector (pMA), we will screen it next day through an E-P digest (today we made glycerol stocks for other four colonies: I40-4/5/6/7) since it's impossible through colony PCR.
With this method we checked four new colonies of I32 (I32-4/5/6/7), I36 (I36-4/5/6/7) and I39 (I39/4/5/6/7).
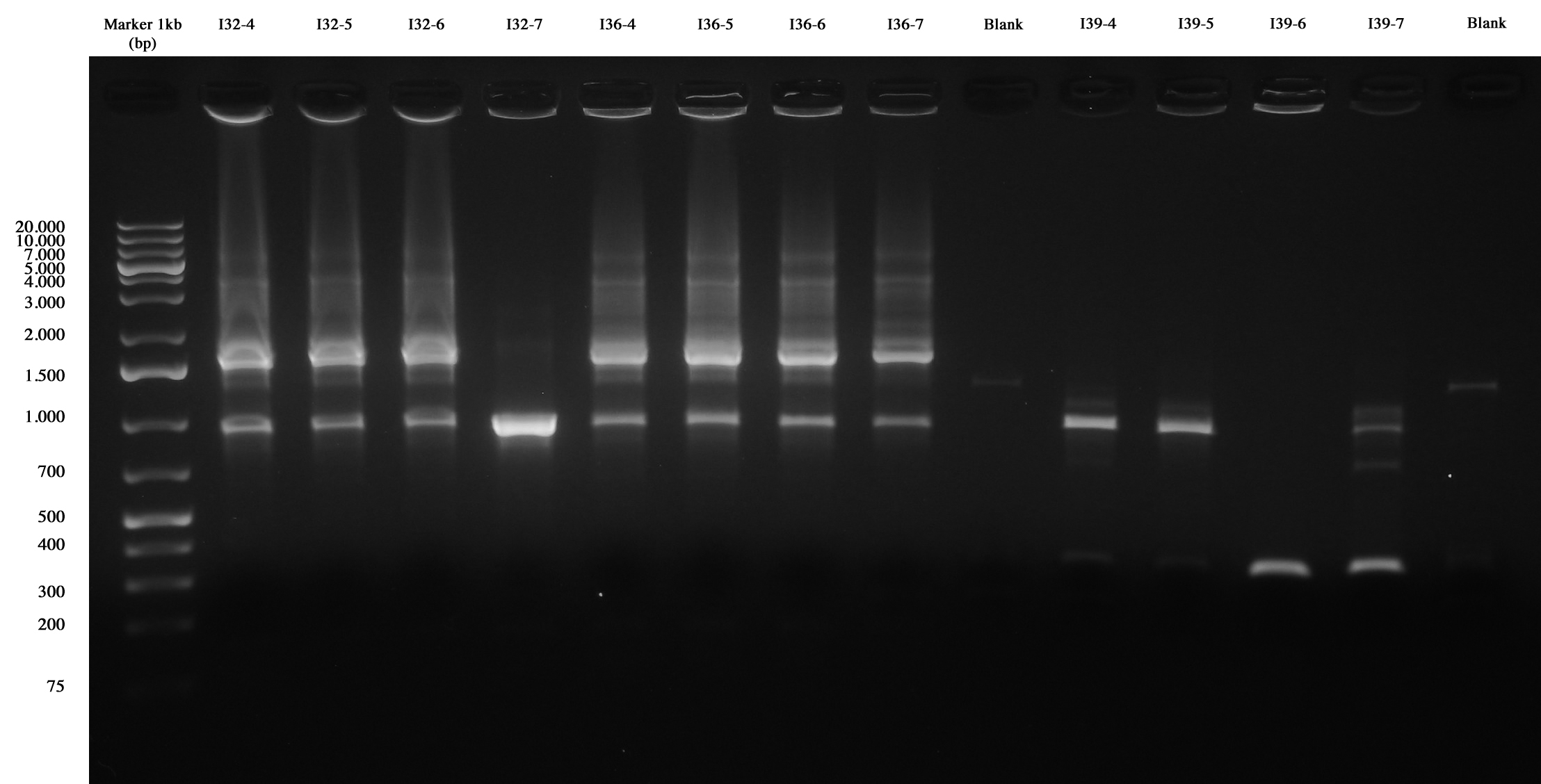 Colony PCR and gel run as screening for ligations I32, I36, I39 I39-4/5 are right (we made glycerol stock for I39-5); I32 and I36 still show the presence of cotrasformation of our ligation and a simple phasin (we took I36-7 instead of I36-2: cleaner gel run). We inoculated 5ml LB+Amp with I40-1/4/5/6/7 and we let them grow ON at 37°C, 220 rpm for E-P digestion/screening.
Inoculum of:
- <partinfo>BBa_J23105</partinfo>
- <partinfo>BBa_J23106</partinfo>
- <partinfo>BBa_J23114</partinfo>
- <partinfo>BBa_J23116</partinfo>
for tomorrow MiniPrep. They will be processed with other promoters, for wich we retrieved purified DNA from our freezer. These further parts are:
- <partinfo>BBa_J23100</partinfo>
- <partinfo>BBa_J23101</partinfo> already digested E-P and purified
- <partinfo>BBa_J23110</partinfo>
- <partinfo>BBa_J23118</partinfo>
- <partinfo>pSB4C5</partinfo> already digested E-P and purified
These promoters, expressing RFP, will be moved from high copy plasmid <partinfo>pSB1A2</partinfo> to the low copy plasmid <partinfo>pSB4C5</partinfo> and will be tested in both condition in order to establish a strength ranking.
Inoculum of PBHR68 BioPlastic producing device and <partinfo>BBa_B0032</partinfo> from glycerol stock in 5ml LB+Amp to check the production of BioPlasic with Sudan Black staining protocol.
Cultures were grown ON at 37°C, 220 rpm.
I42, I43 ligations were transformed in BW23474 strain and plated on selective LB agar plate with chloramphenicol concentration of 34 ug/ml. Instead I44 was transformed in DB3.1 strain and plated on the same type of plate of the other two transformations.
August, 25th
Minipreps of I40-1/4/5/6/7 (to make the screening of ligations) were quantified as follows:
- I40-1: 415,7 ng/ul
- I40-4: 382,8 ng/ul
- I40-5: 420 ng/ul
- I40-6: 487,8 ng/ul
- I40-7: 479,9 ng/ul
Samples were digested E-P for three hours
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 (ul) | Enzyme 2 (ul) | Buffer H (ul)
|
| I40-1 | Insert/Screening | 25 | 2 | 19,5 | 0,5 EcoRI | 0,5 PstI | 2,5
|
| I40-4 | Insert/Screening | 25 | 2 | 19,5 | 0,5 EcoRI | 0,5 PstI | 2,5
|
| I40-5 | Insert/Screening | 25 | 2 | 19,5 | 0,5 EcoRI | 0,5 PstI | 2,5
|
| I40-6 | Insert/Screening | 25 | 2 | 19,5 | 0,5 EcoRI | 0,5 PstI | 2,5
|
| I40-7 | Insert/Screening | 25 | 2 | 19,5 | 0,5 EcoRI | 0,5 PstI | 2,5
|
and than gel run to check the length of ligations.
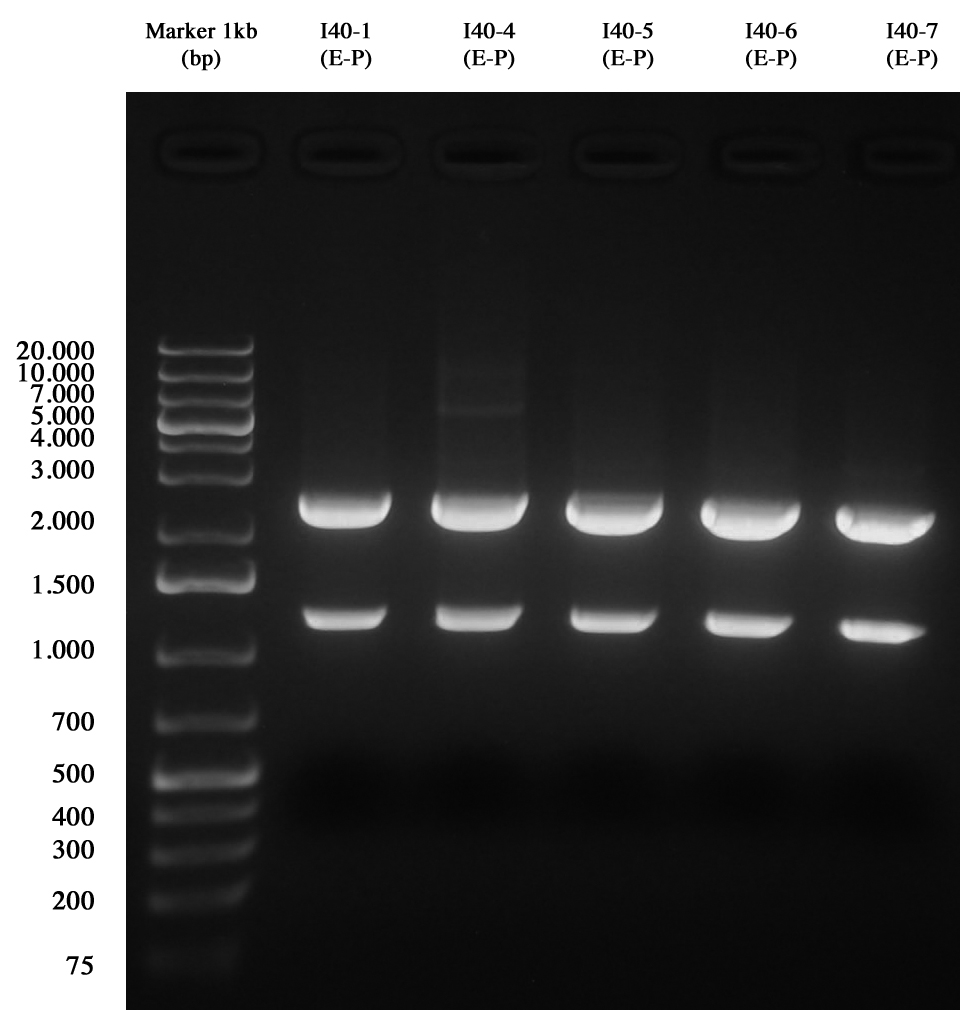 Gel run for I40-1/4/5/6/7 digested E-P As you can see all samples are positive, so we decided to keep glycerol stock of I40-1.
MiniPrep was performed for following cultures, and DNA was quantified as follows:
| Culture name | Quantifiaction (ng/ul)
|
| <partinfo>BBa_J23105</partinfo> | X ng/ul
|
| <partinfo>BBa_J23106</partinfo> | X ng/ul
|
| <partinfo>BBa_J23114</partinfo> | X ng/ul
|
| <partinfo>BBa_J23116</partinfo> | X ng/ul
|
Purified DNA was digested as follows:
Digestion of:
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 | Enzyme 2 | Buffer H
|
| <partinfo>BBa_J23100</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23105</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23106</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23110</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23114</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23116</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
| <partinfo>BBa_J23118</partinfo> | Insert | 25 | 10 | 10,5 | 1 EcoRI | 1 PstI | 2,5
|
Ligation of:
- <partinfo>BBa_J23100</partinfo>_4C5=<partinfo>BBa_J23100</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23101</partinfo>_4C5=<partinfo>BBa_J23101</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23105</partinfo>_4C5=<partinfo>BBa_J23105</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23106</partinfo>_4C5=<partinfo>BBa_J23106</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23110</partinfo>_4C5=<partinfo>BBa_J23110</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23114</partinfo>_4C5=<partinfo>BBa_J23114</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23116</partinfo>_4C5=<partinfo>BBa_J23116</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
- <partinfo>BBa_J23118</partinfo>_4C5=<partinfo>BBa_J23118</partinfo>(E-P)+<partinfo>pSB4C5</partinfo>(E-P)
Preparation of samples for BioPlastic screening:
- Cultures of PBHR68 and <partinfo>BBa_B0032</partinfo> were diluted 1:100 in fresh LB+Amp and werew prepared as follows:
- <partinfo>BBa_B0032</partinfo> with NOTHING added (negative control)
- PBHR68 with NOTHING added
- PBHR68 + 2% glycerol (carbon source for BioPlastic production)
- PBHR68 + 1mM IPTG (inducer for Plac promoter, expressing BioPlastic enzymes)
- PBHR68 + 2% glycerol + 1mM IPTG
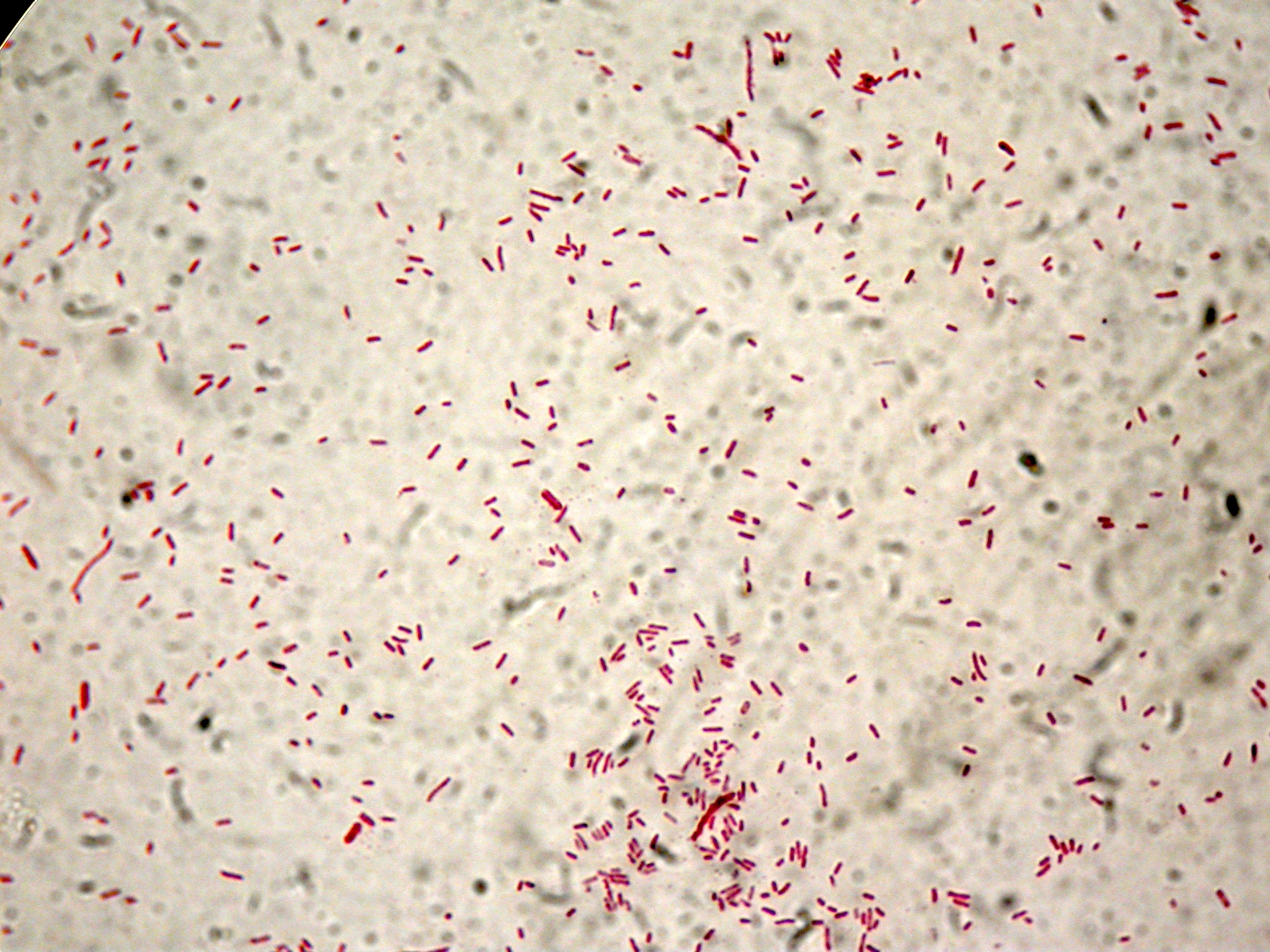
After 8 hours, Sudan Black staining protocol was performed on 70ul cultures and 5 microscope slides were prepared. The resulting images are shown here:
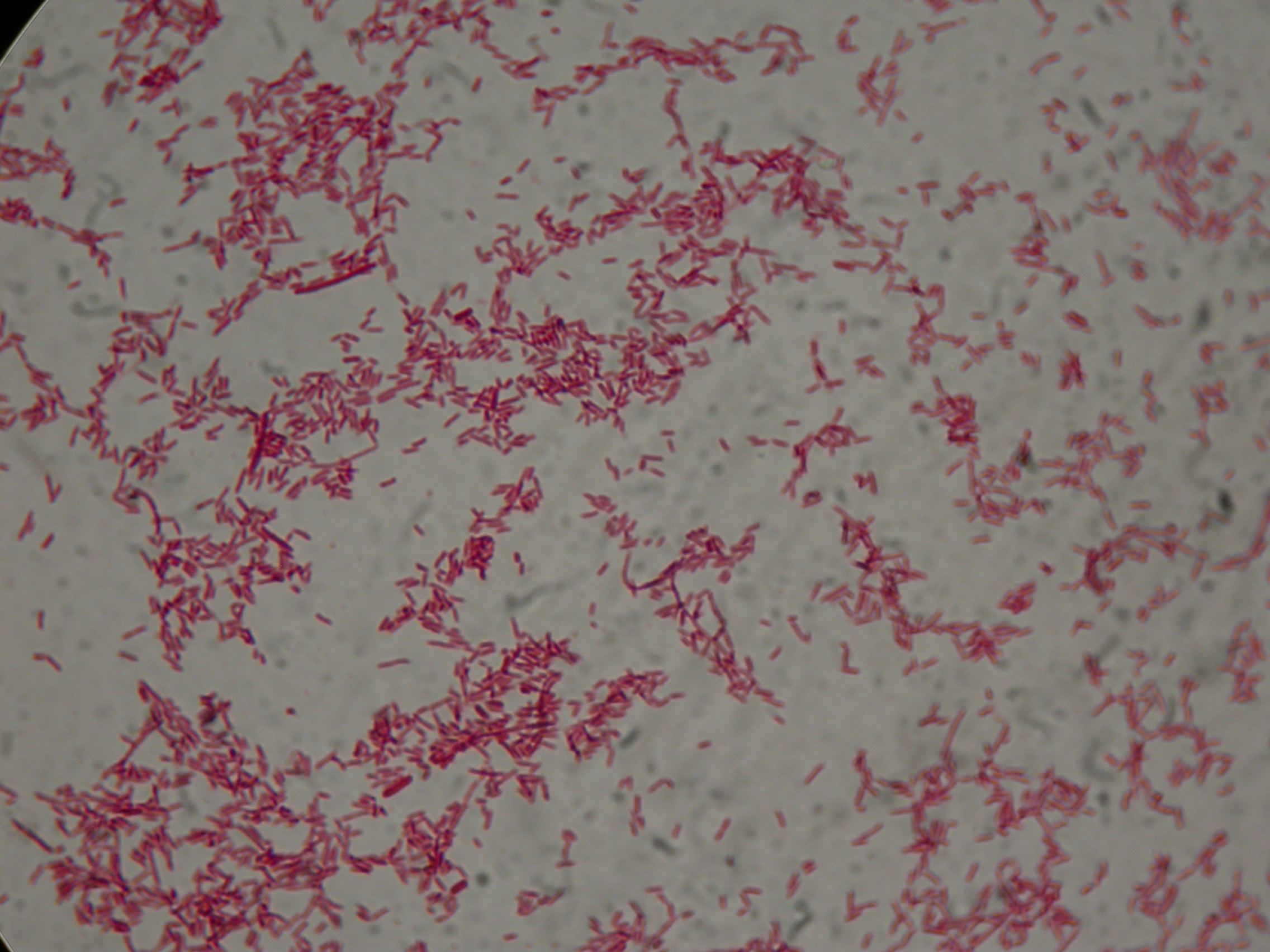 PBHR68 with nothing added in the culture, after 8 hours | 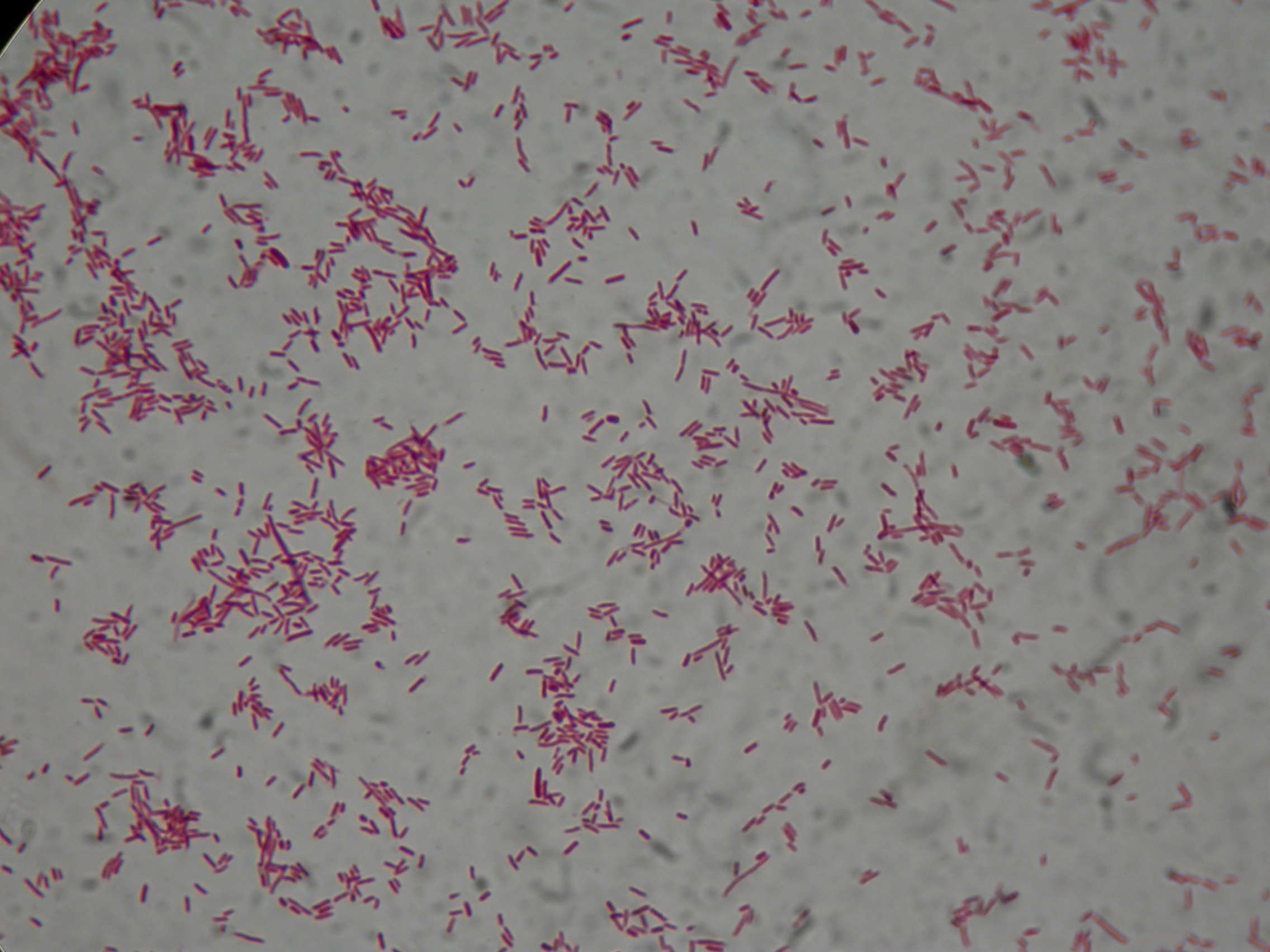 PBHR68 with nothing added in the culture, after 8 hours |
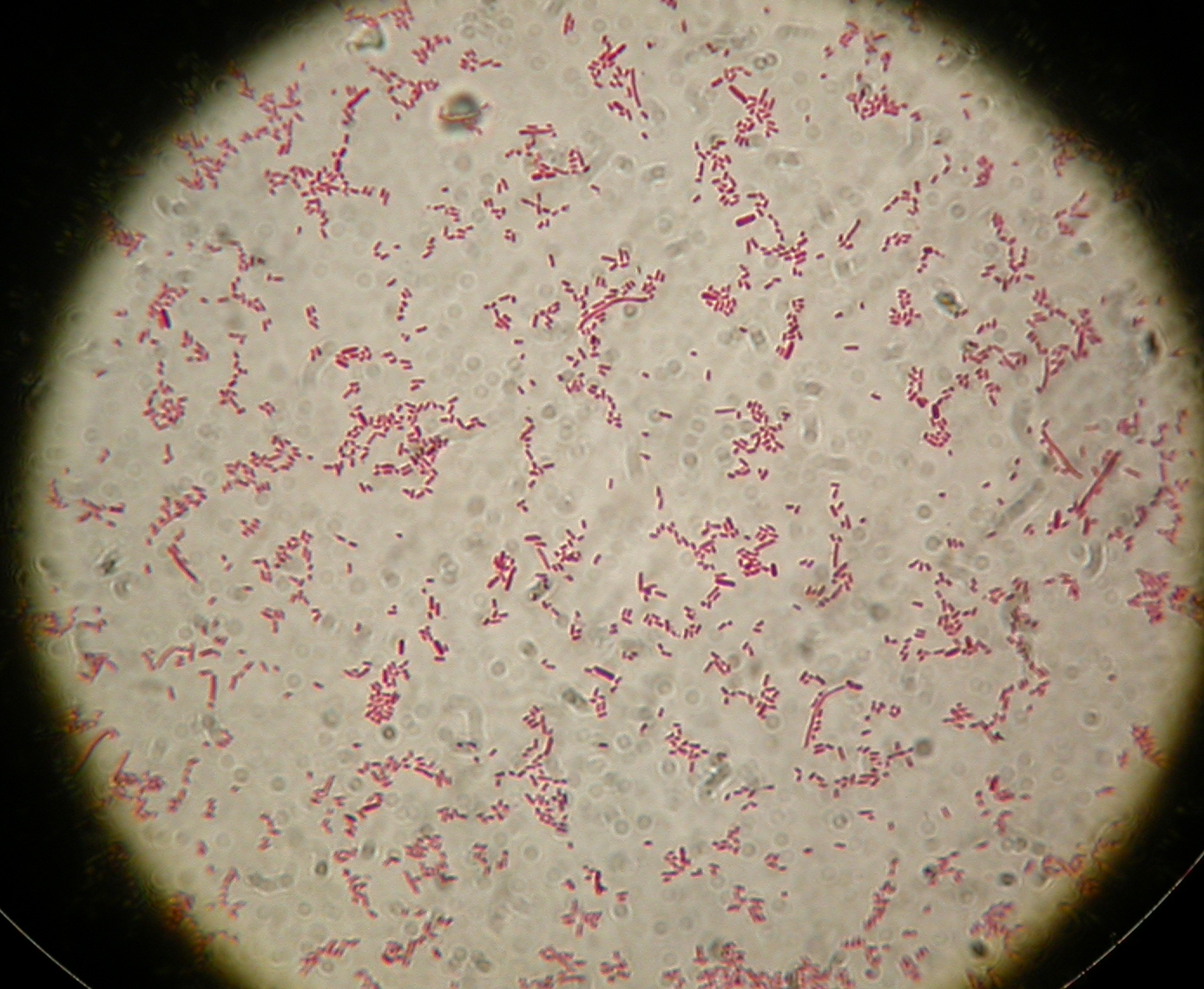 <partinfo>BBa_B0032</partinfo> with nothing added in the culture, after 8 hours (negative control) | 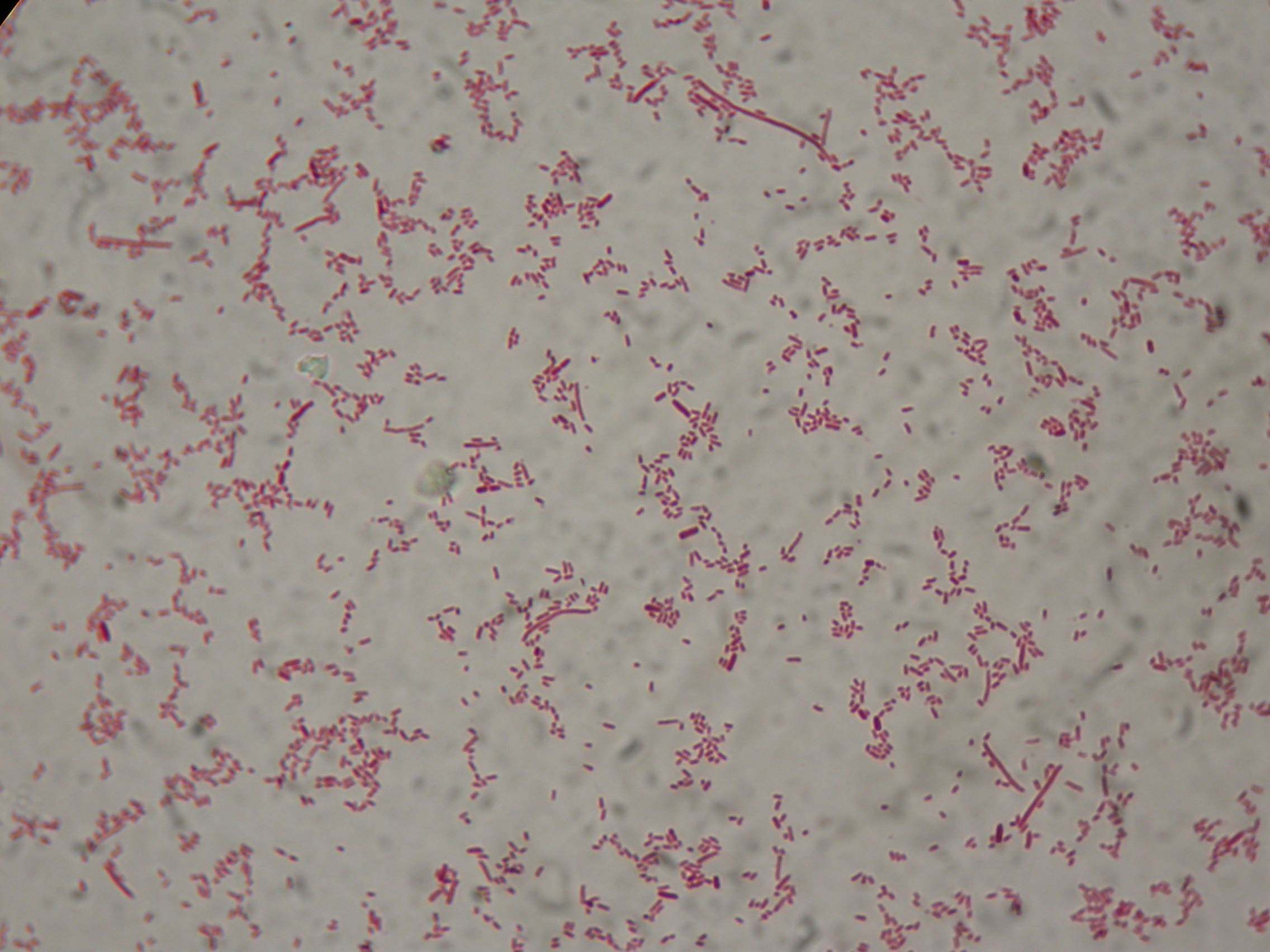 <partinfo>BBa_B0032</partinfo> with nothing added in the culture, after 8 hours (negative control) |
 PBHR68 with 2% glycerol added in the culture, after 8 hours | 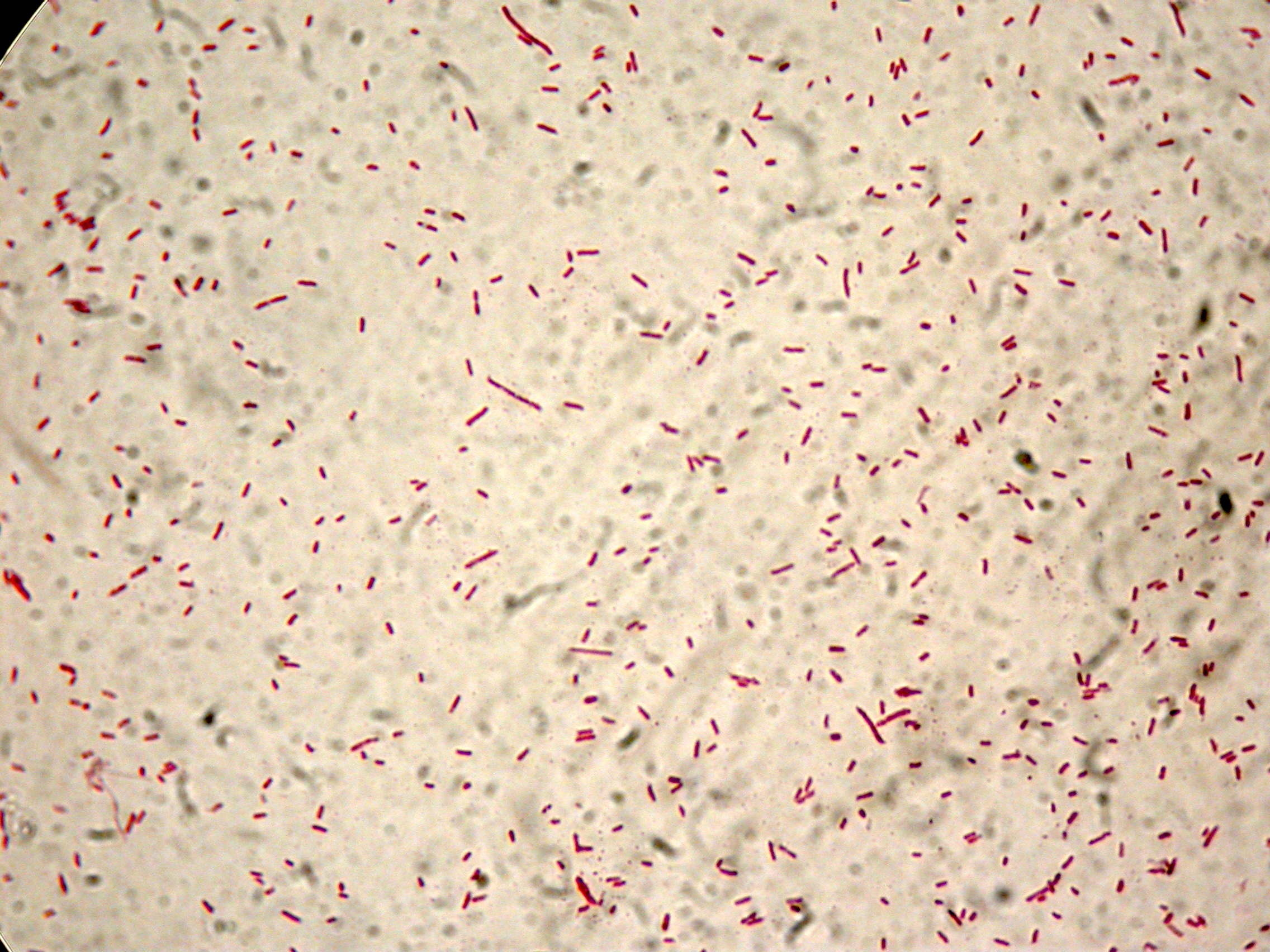 PBHR68 with 2% glycerol added in the culture, after 8 hours |
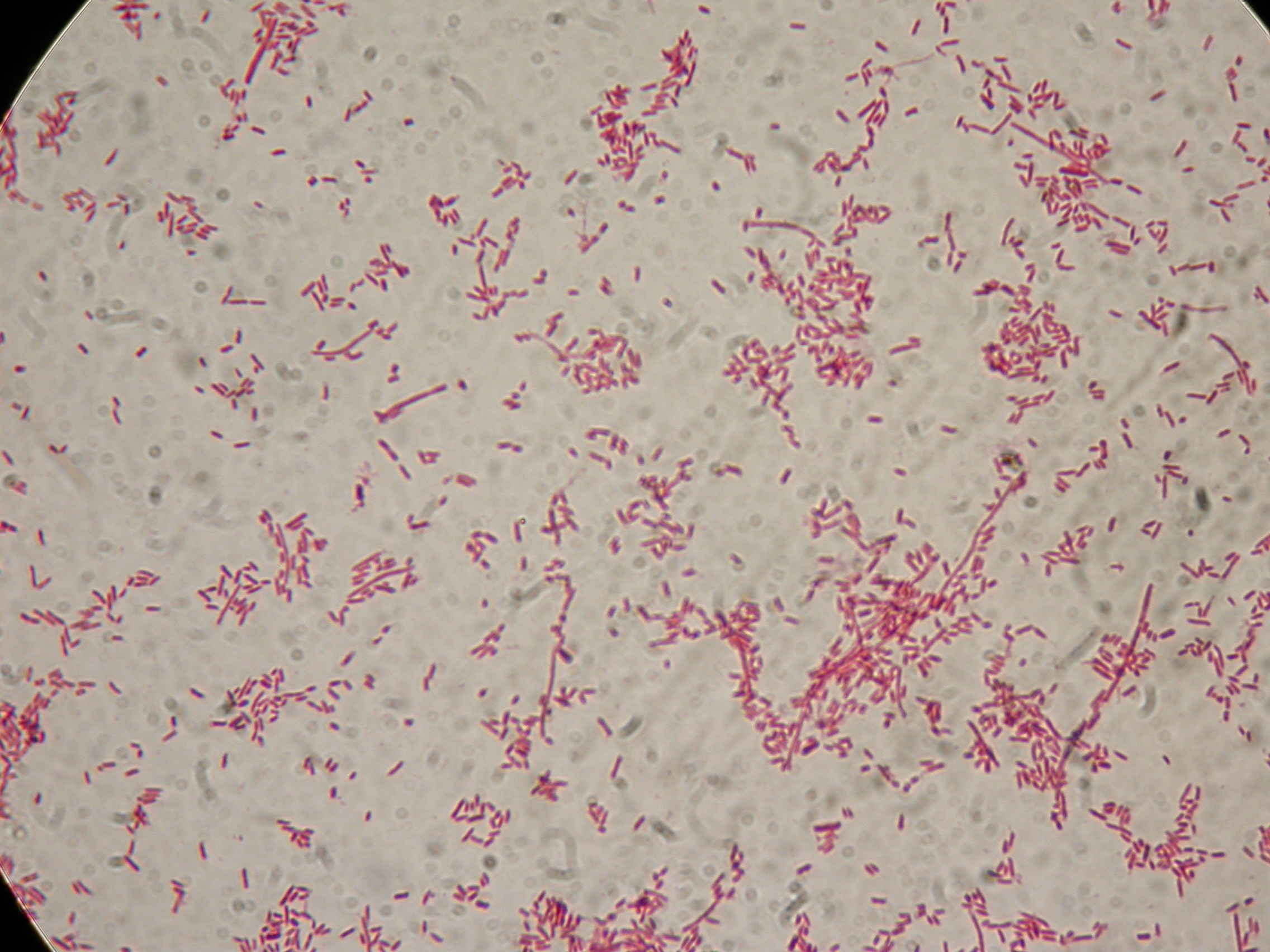 PBHR68 with 1mM IPTG added in the culture, after 8 hours | 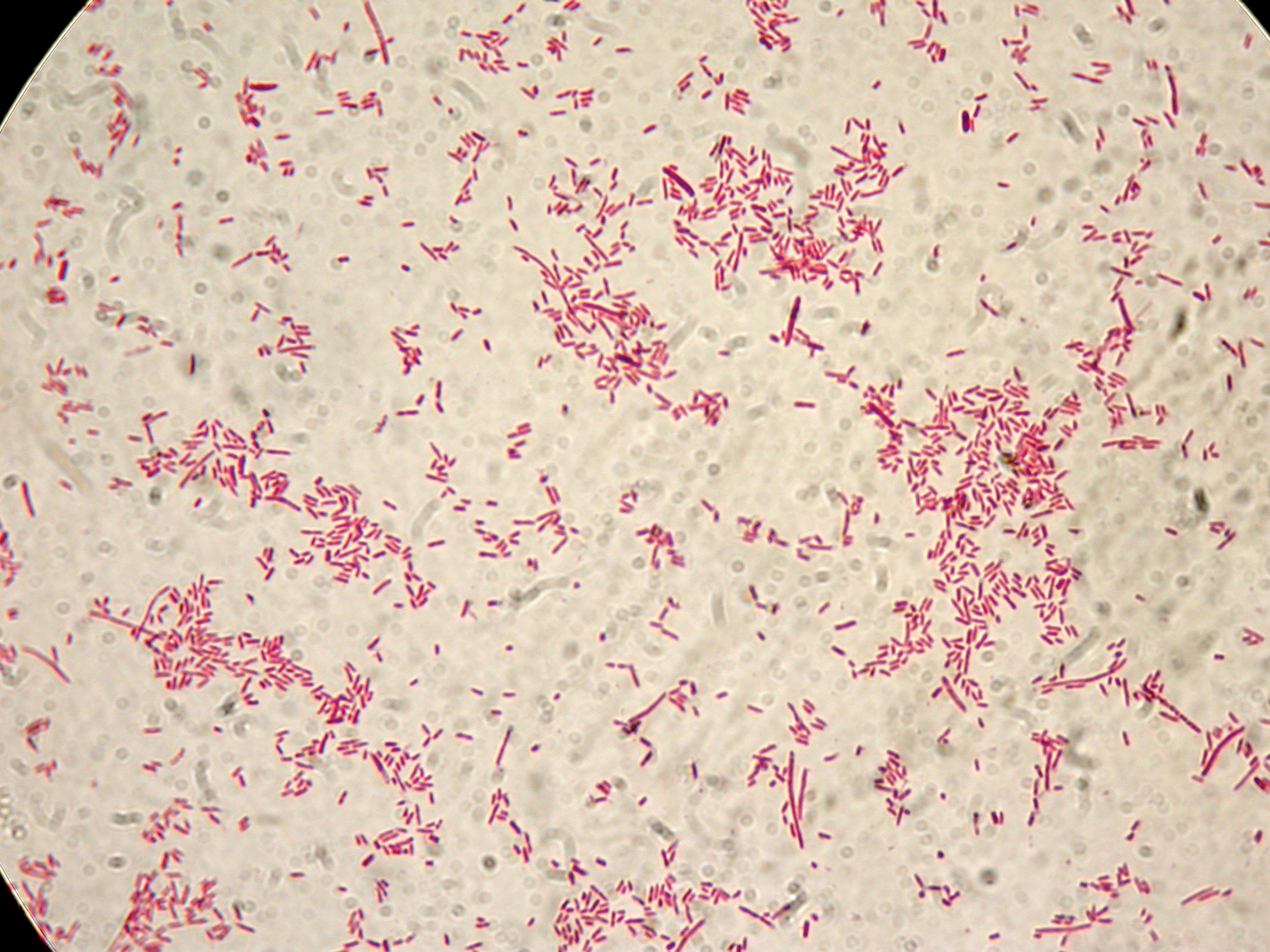 PBHR68 with 1mM IPTG in the culture, after 8 hours |
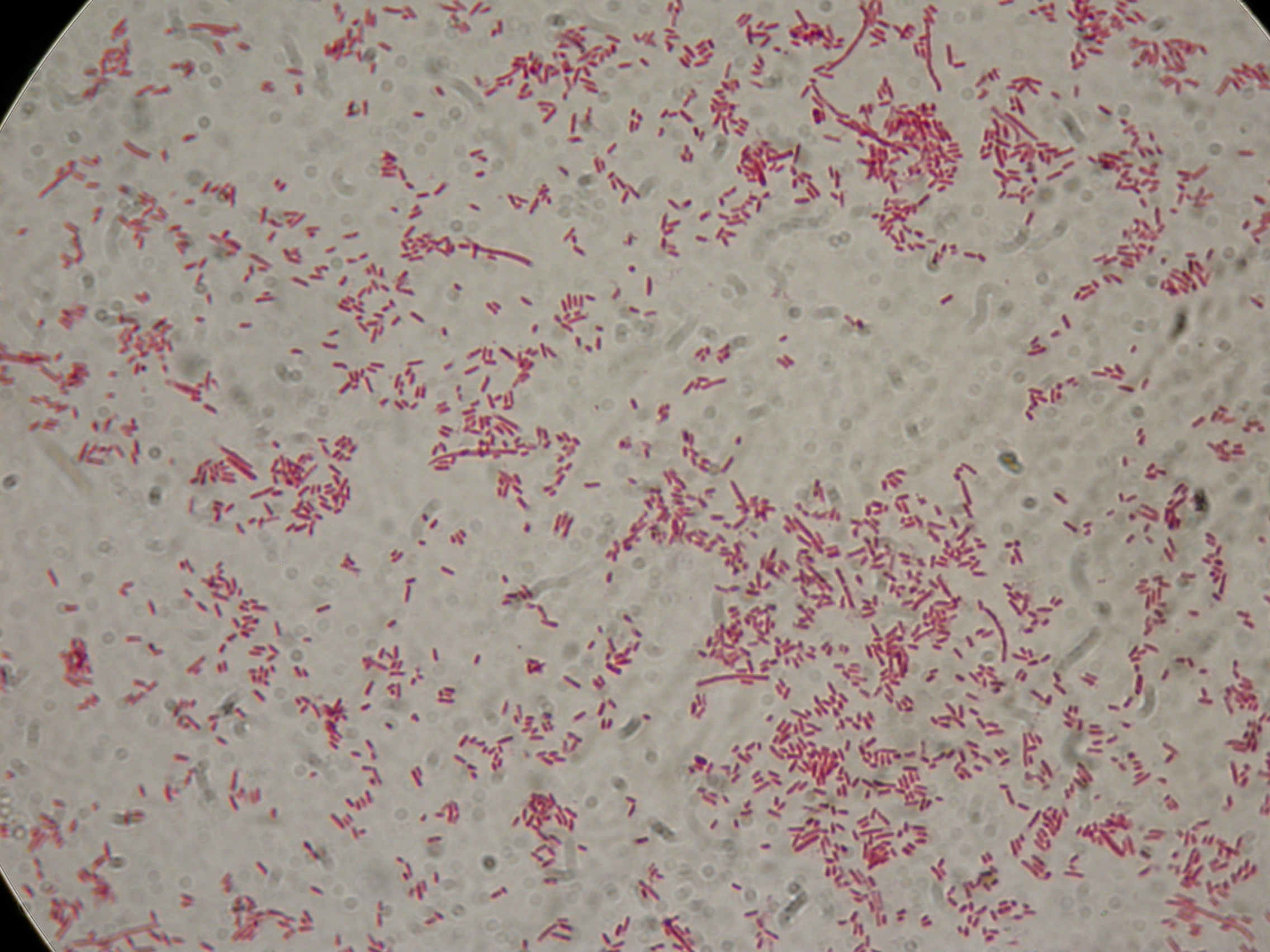 PBHR68 with 1mM IPTG and 2% glycerol added in the culture, after 8 hours | 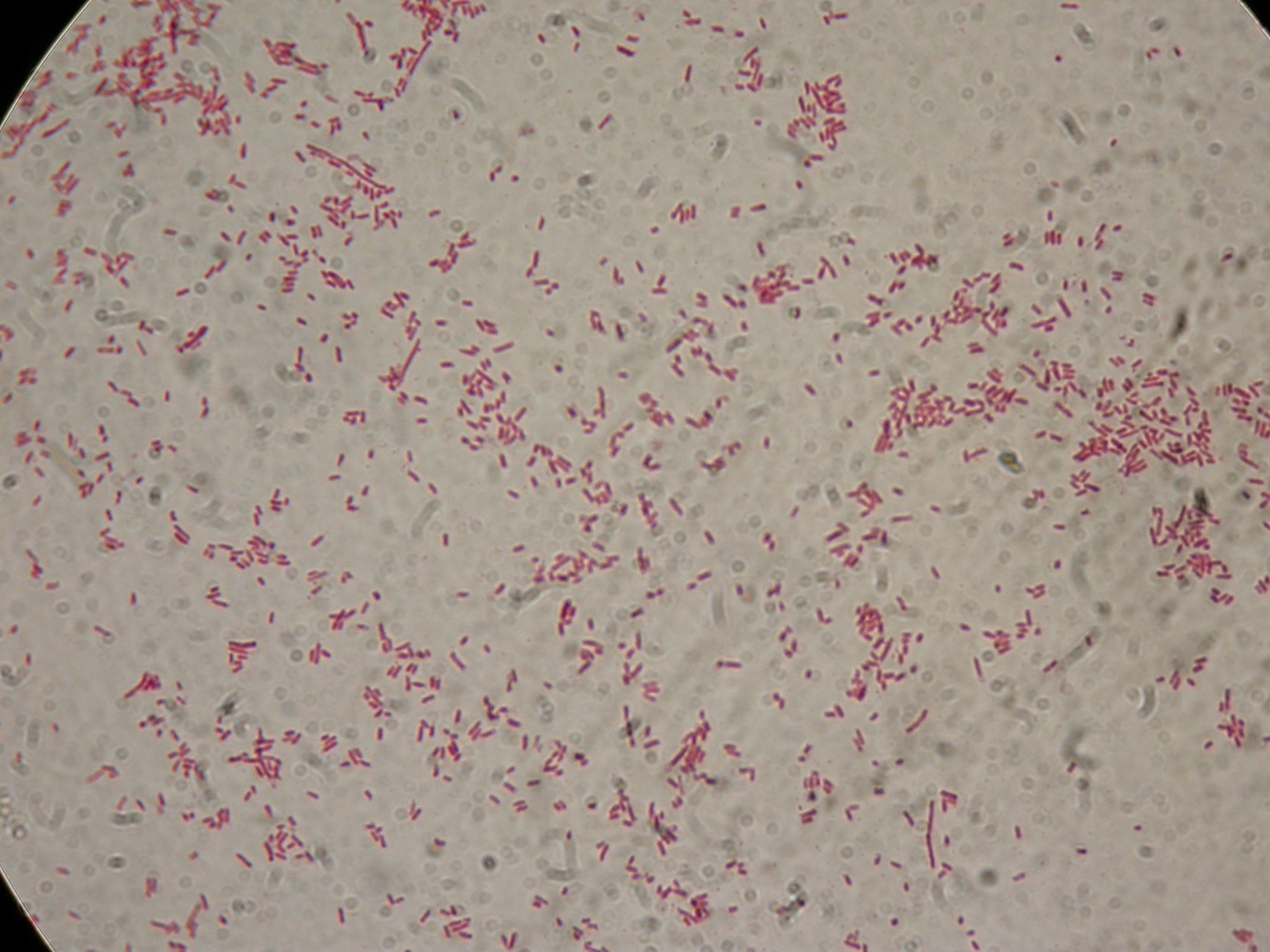 PBHR68 with 1mM IPTG and 2% glycerol added in the culture, after 8 hours |
Cultures were further incubated for 22 hours at 37°C, 220 rpm. Tomorrow, we will repeat the staining protocol after 30 hours from inoculum to check the time E. coli takes to produce BioPlastic granules.
August, 26th
Transformation of ligations:
| Ligation name | Strain | Resistance
|
| <partinfo>BBa_J23100</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23101</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23105</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23106</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23110</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23114</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23116</partinfo>_4C5 | TOP10 | Cm 12,5
|
| <partinfo>BBa_J23118</partinfo>_4C5 | TOP10 | Cm 12,5
|
Plates were incubated at 37°C overnight.
Inoculum of self inducible promoters for TECAN test:
| I7 | I8 | I9 | I10
|
| I12 | I8_4C5 | I15 | I16
|
| I17 | I18 | I19 | I27
|
| I28 | I30 | <partinfo>BBa_B0032</partinfo> | A2 (<partinfo>BBa_J23101</partinfo>+GFP)
|
After 30 hours, Sudan Black staining protocol was performed on 70ul cultures and 5 microscope slides were prepared. The resulting images are shown here:
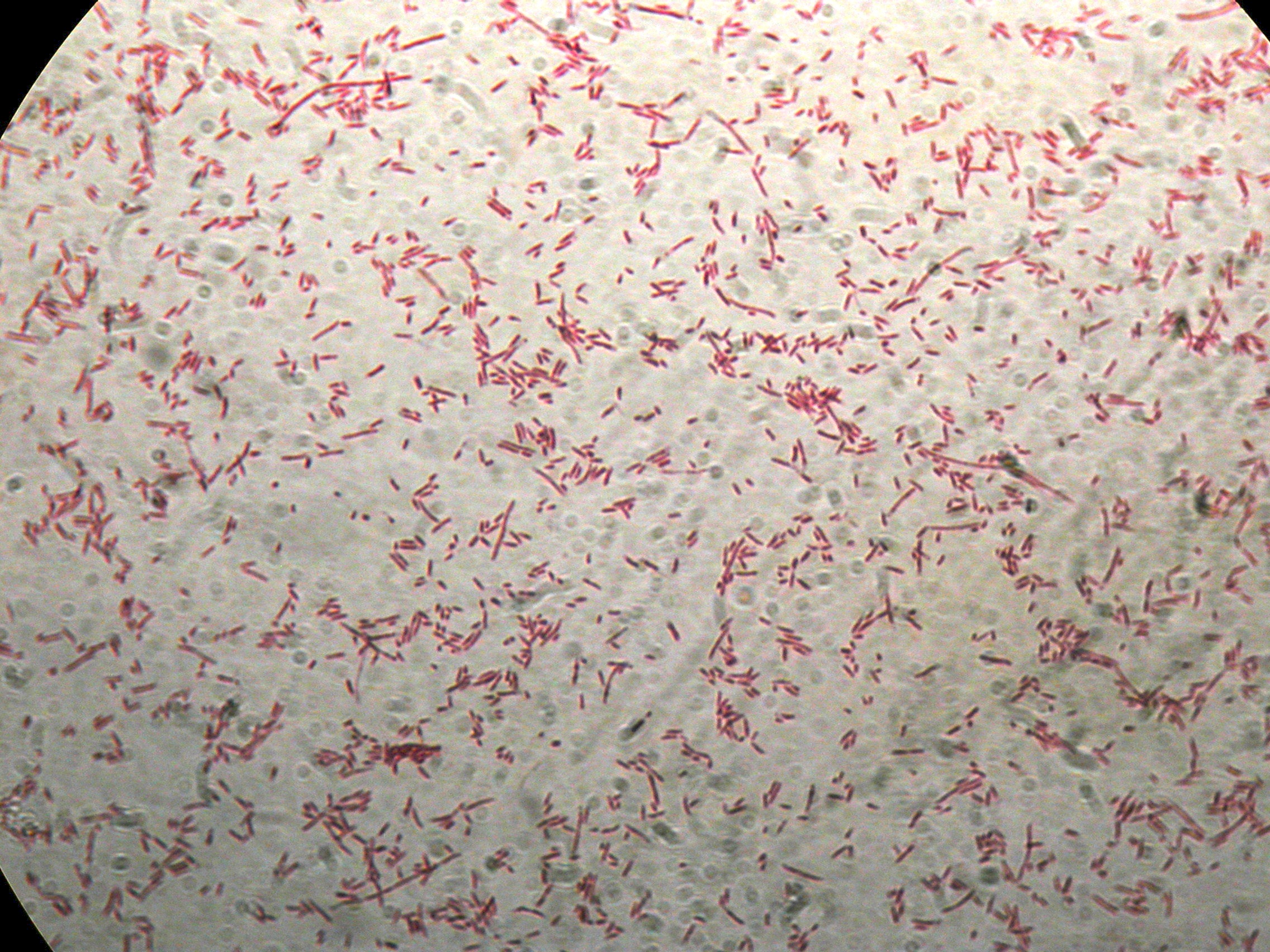 PBHR68 with nothing added in the culture, after 30 hours | 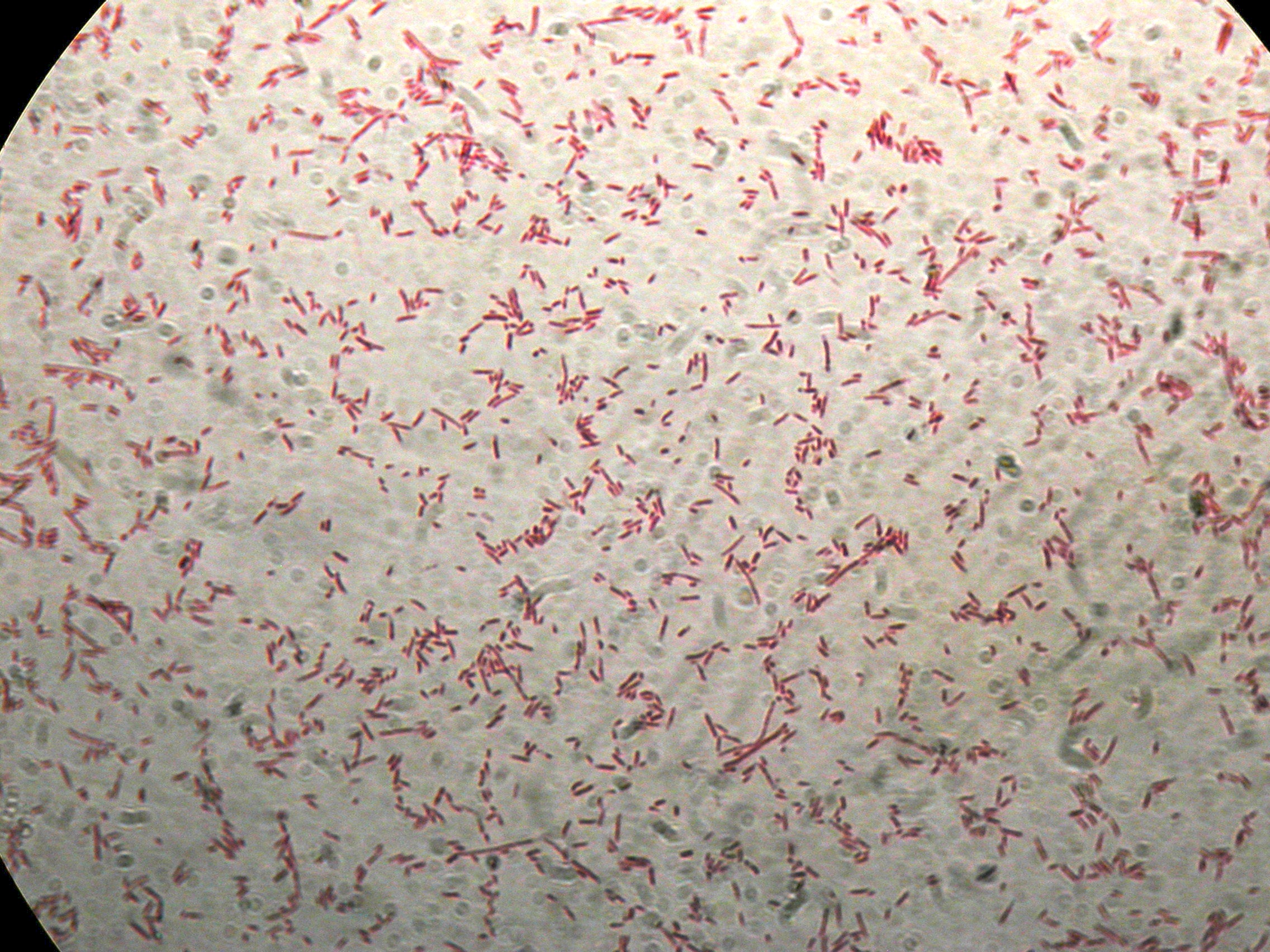 PBHR68 with nothing added in the culture, after 30 hours |
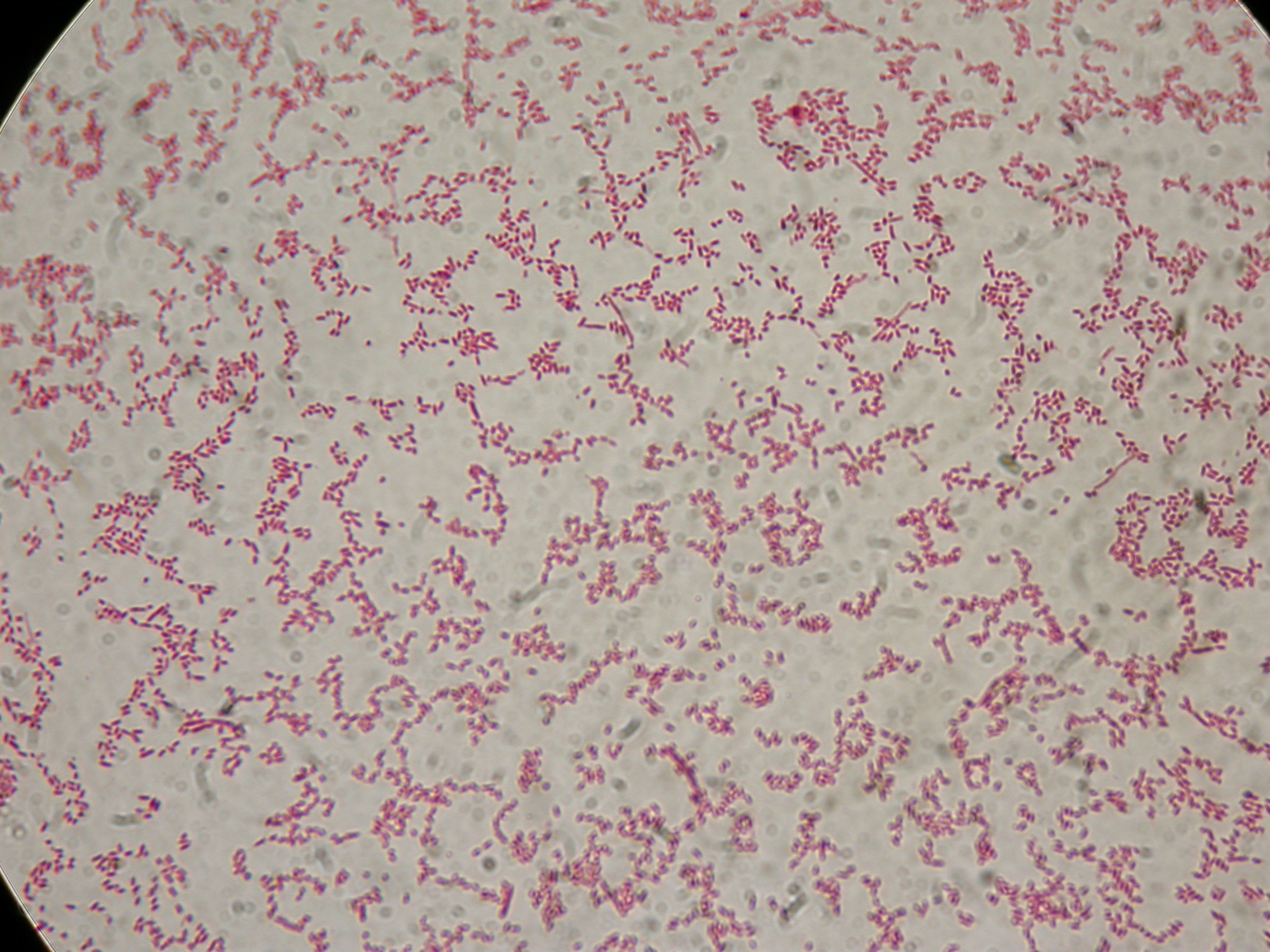 <partinfo>BBa_B0032</partinfo> with nothing added in the culture, after 30 hours (negative control) | 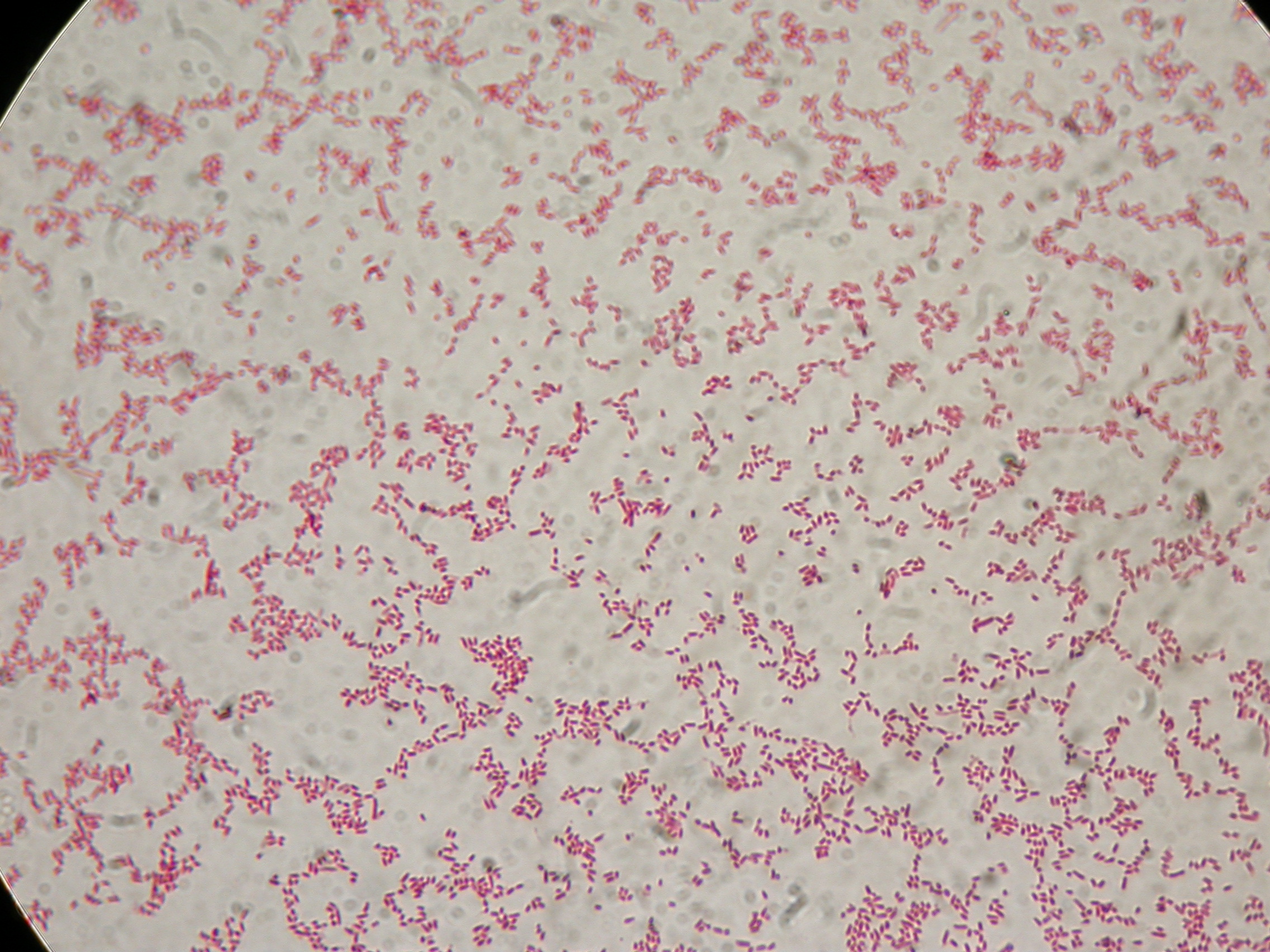 <partinfo>BBa_B0032</partinfo> with nothing added in the culture, after 30 hours (negative control) |
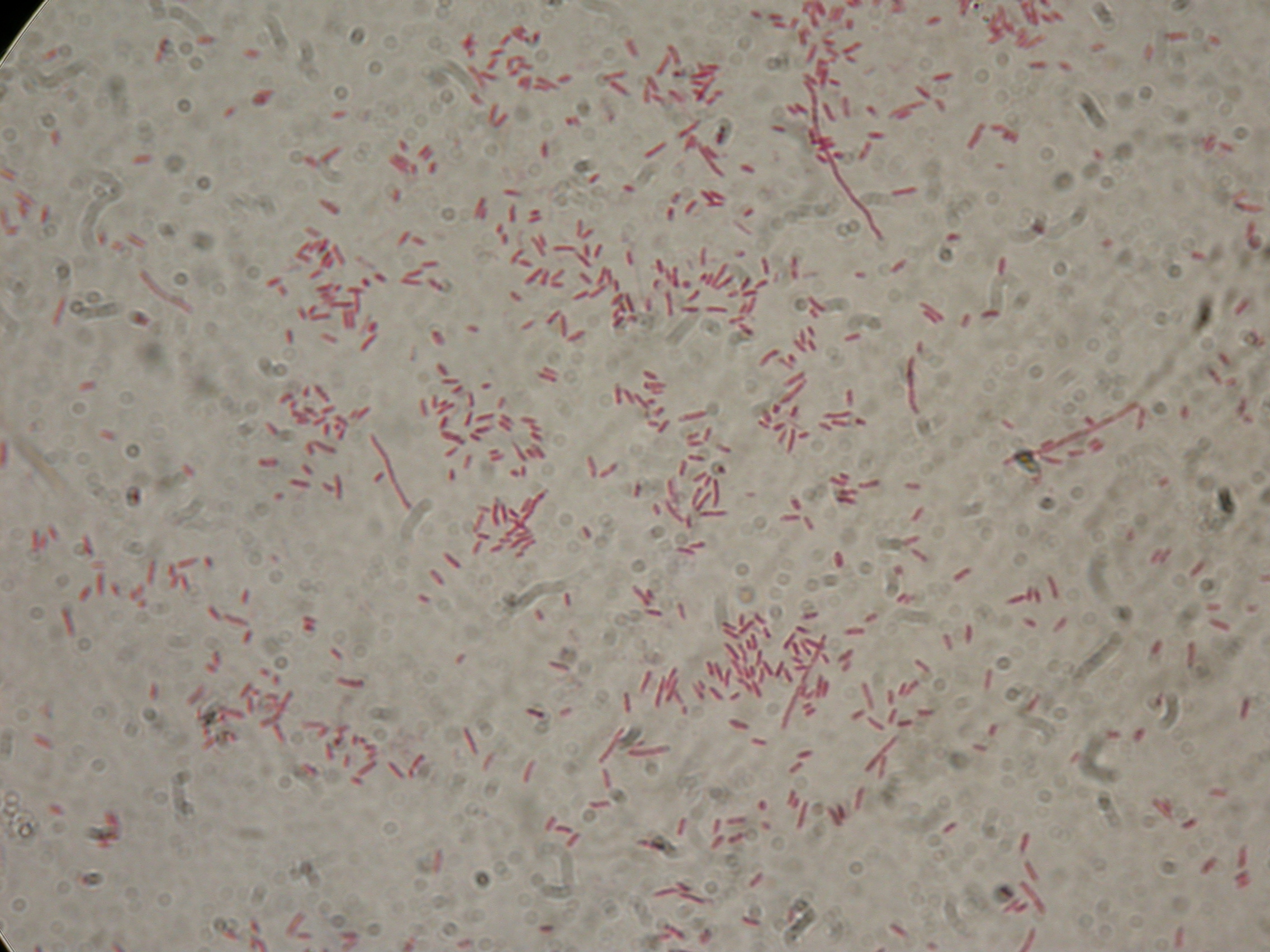 PBHR68 with 2% glycerol added in the culture, after 30 hours | 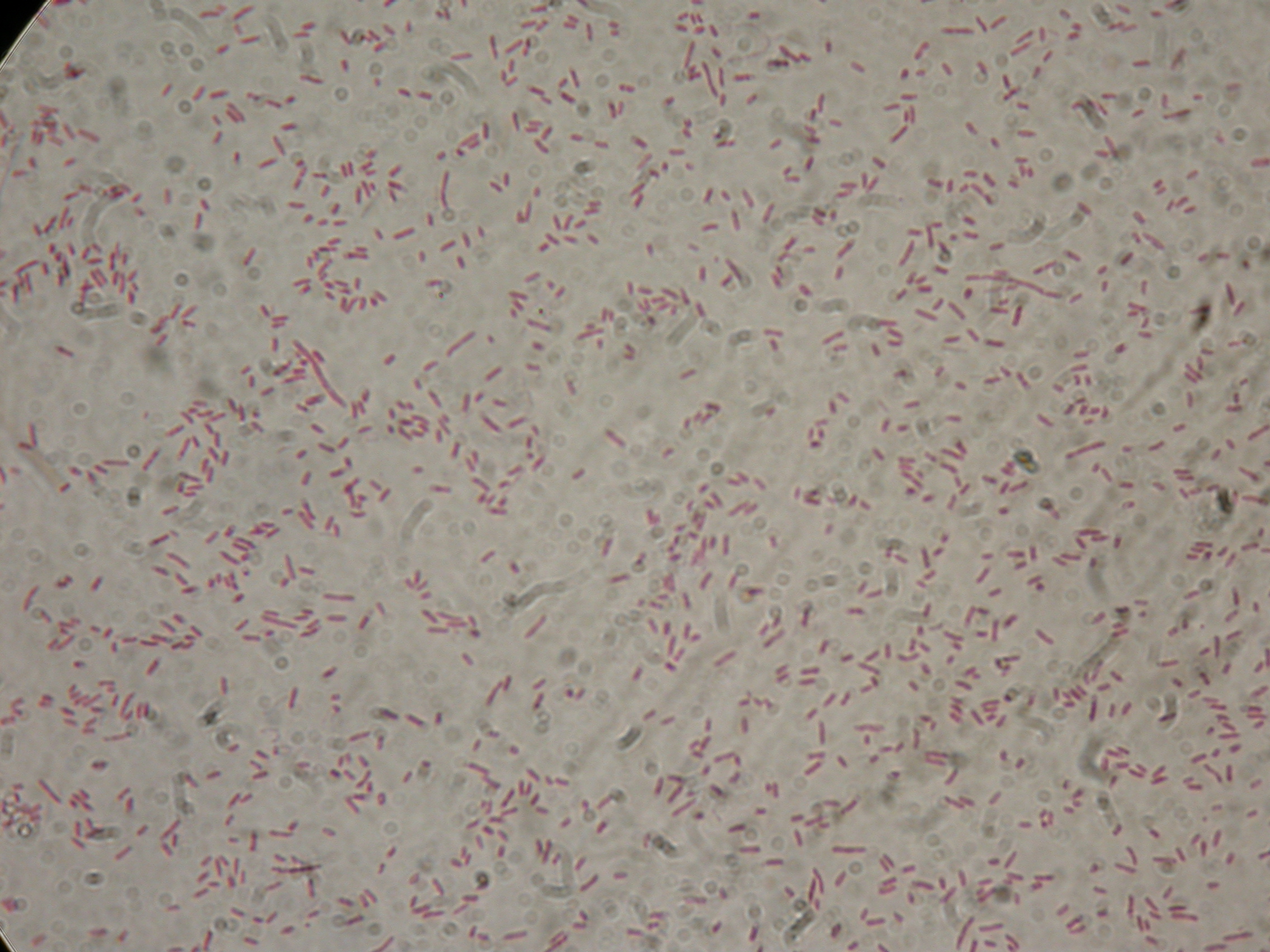 PBHR68 with 2% glycerol added in the culture, after 30 hours |
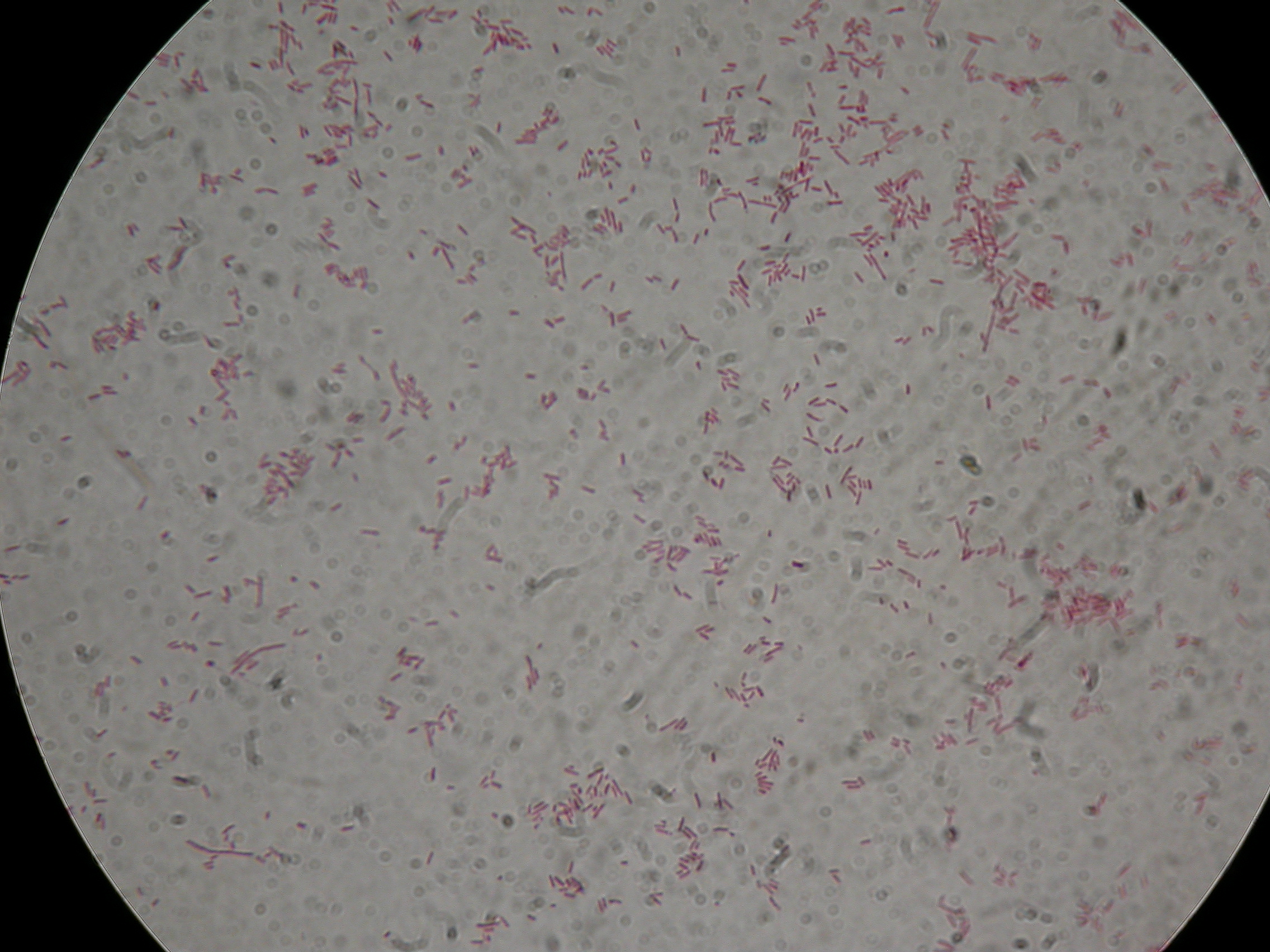 PBHR68 with 1mM IPTG added in the culture, after 30 hours | 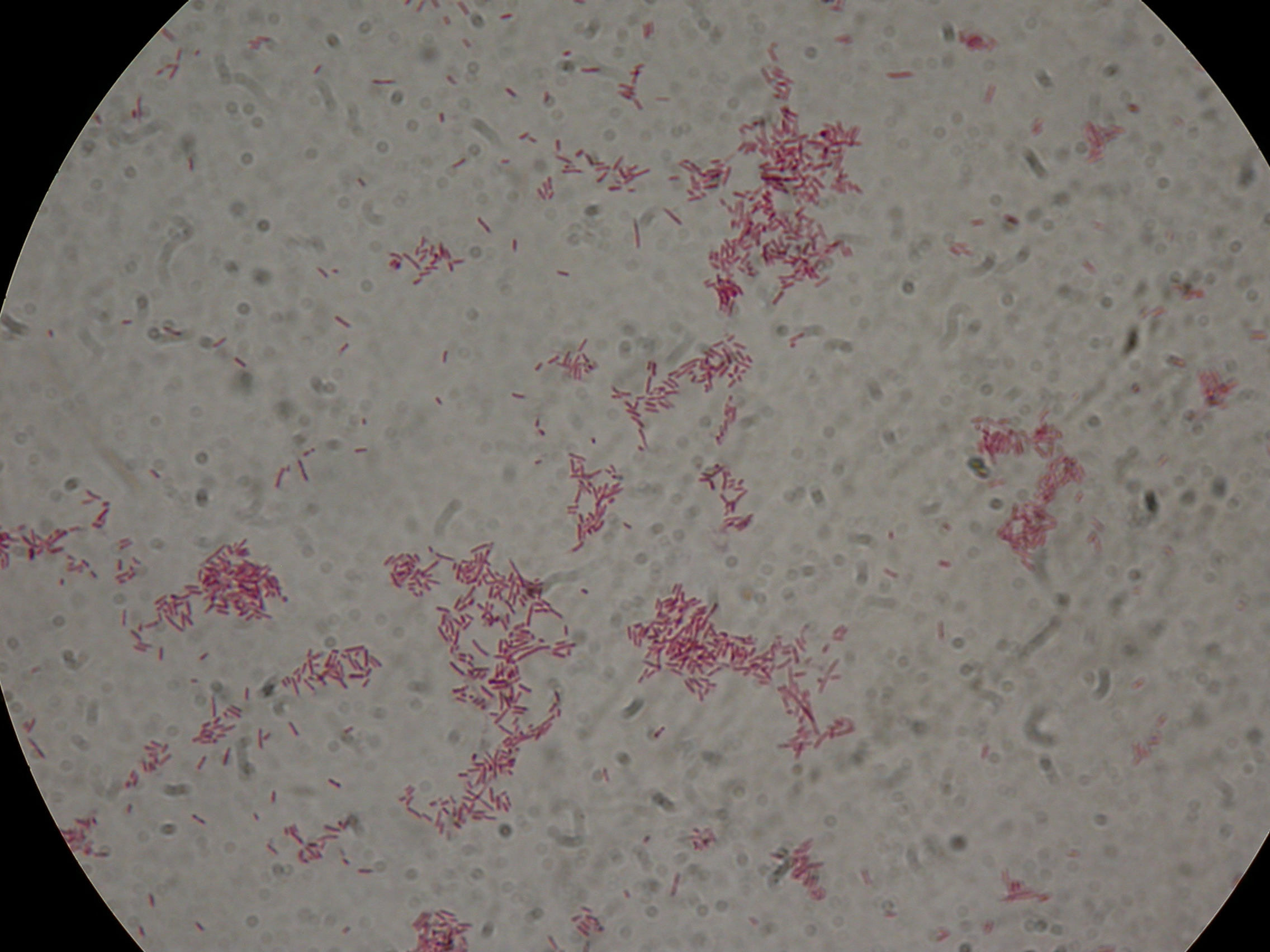 PBHR68 with 1mM IPTG in the culture, after 30 hours |
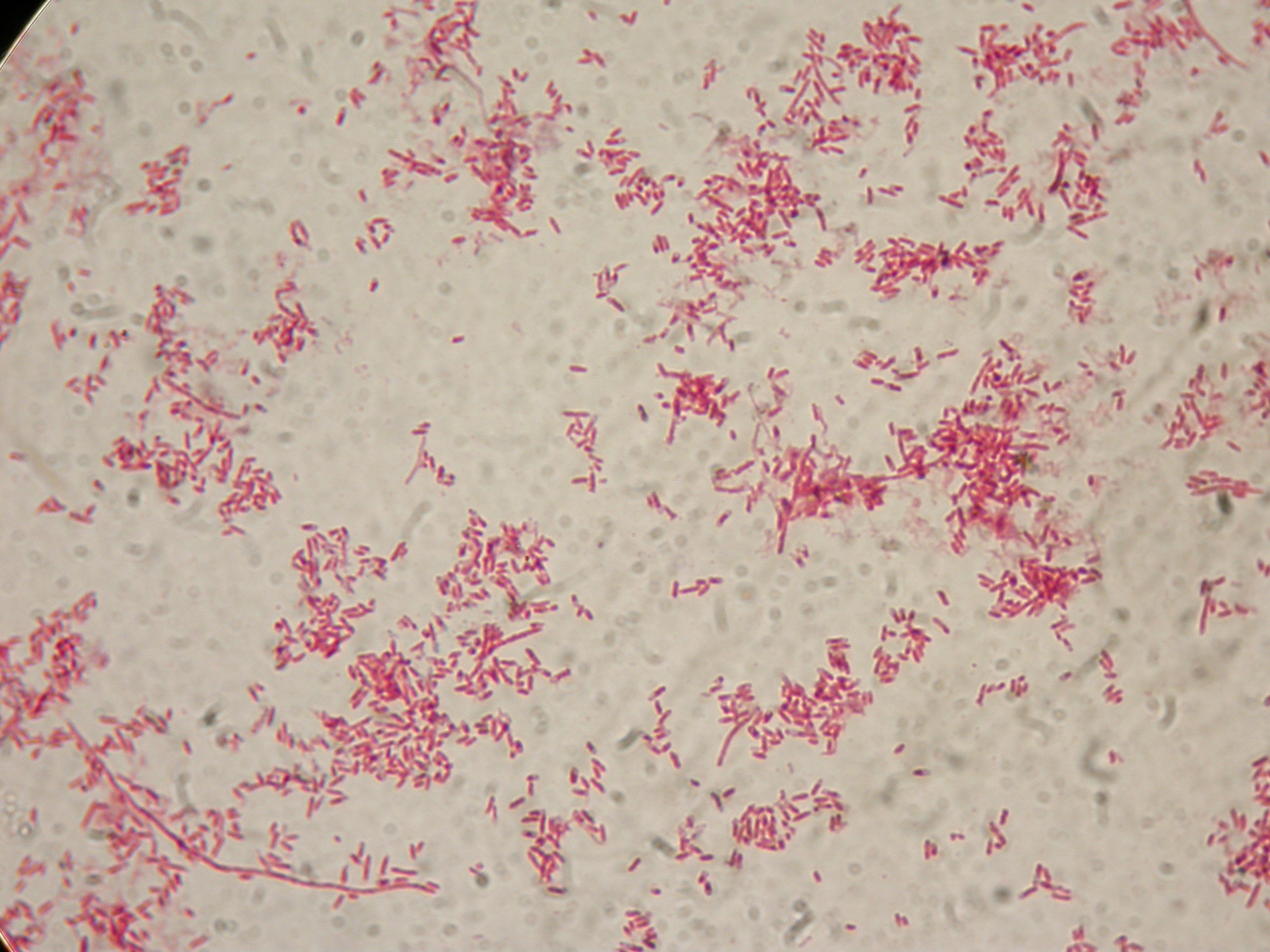 PBHR68 with 1mM IPTG and 2% glycerol added in the culture, after 30 hours | 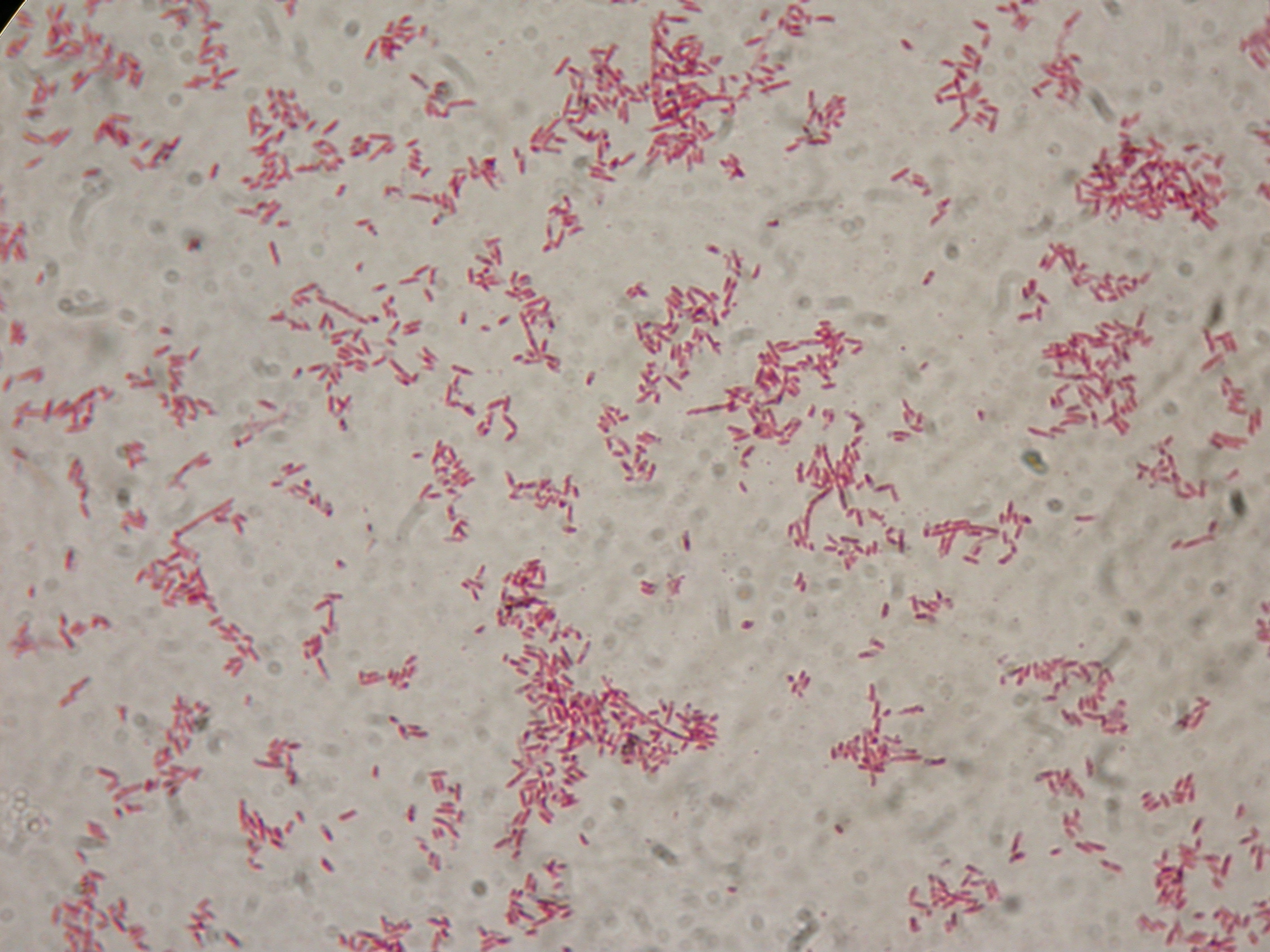 PBHR68 with 1mM IPTG and 2% glycerol added in the culture, after 30 hours |
Inoculum of
- <partinfo>BBa_J13002</partinfo>
- I33
- I34
- I39
- I41
in 5 ml LB+Amp
August, 27th
Previously inoculated strains were miniprepped and quantified as follows:
- <partinfo>BBa_J13002</partinfo>: 29,9 ng/ul
- I33: 171,6 ng/ul
- I34: 219,4 ng/ul
- I39: 21,2 ng/ul
- I41: 94,7 ng/ul
Digestions (3,5 hours) for ligations:
- I47: <partinfo>BBa_J13002</partinfo> (S-P) + I33 (X-P)
- I48: I39 (S-P) + I34 (X-P)
- I49: I39 (S-P) + I41 (X-P)
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 (ul) | Enzyme 2 (ul) | Buffer H (ul)
|
| <partinfo>BBa_J13002</partinfo> | Vector | 25 | 21,5 | 0 | 0,5 SpeI | 0,5 PstI | 2,5
|
| I39 | Vector | 25 | 21,5 | 0 | 0,5 S | 0,5 P | 2,5
|
| I33 | Insert | 25 | 10,5 | 11 | 0,5 XbaI | 0,5 P | 2,5
|
| I34 | Insert | 25 | 13,5 | 8 | 0,5 X | 0,5 P | 2,5
|
| I41 | Insert | 25 | 2 | 19,5 | 0,5 X | 0,5 P | 2,5
|
Samples were loaded into a medium gel, run and cut
Gel extraction was quantified as follows:
- <partinfo>BBa_J13002</partinfo> (S-P): 19,9 ng/ul
- I33 (X-P): 17,7 ng/ul
- I34 (X-P): 11,1 ng/ul
- I39 (S-P): 4,7 ng/ul
- I41 (X-P): 6,2 ng/ul
Ligation of
- I47: <partinfo>BBa_J13002</partinfo> (S-P) + I33 (X-P)
- I48: I39 (S-P) + I34 (X-P)
- I49: I39 (S-P) + I41 (X-P)
Plates containing the transformed ligations were all grown. Colony PCR was performed to screen if the length of insert was correct.
| ligation name | number of colonies screened
|
| <partinfo>BBa_J23100</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23101</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23105</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23106</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23110</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23114</partinfo>_4C5 | 1 colony
|
| <partinfo>BBa_J23116</partinfo>_4C5 | 2 colonies
|
| <partinfo>BBa_J23118</partinfo>_4C5 | 2 colonies
|
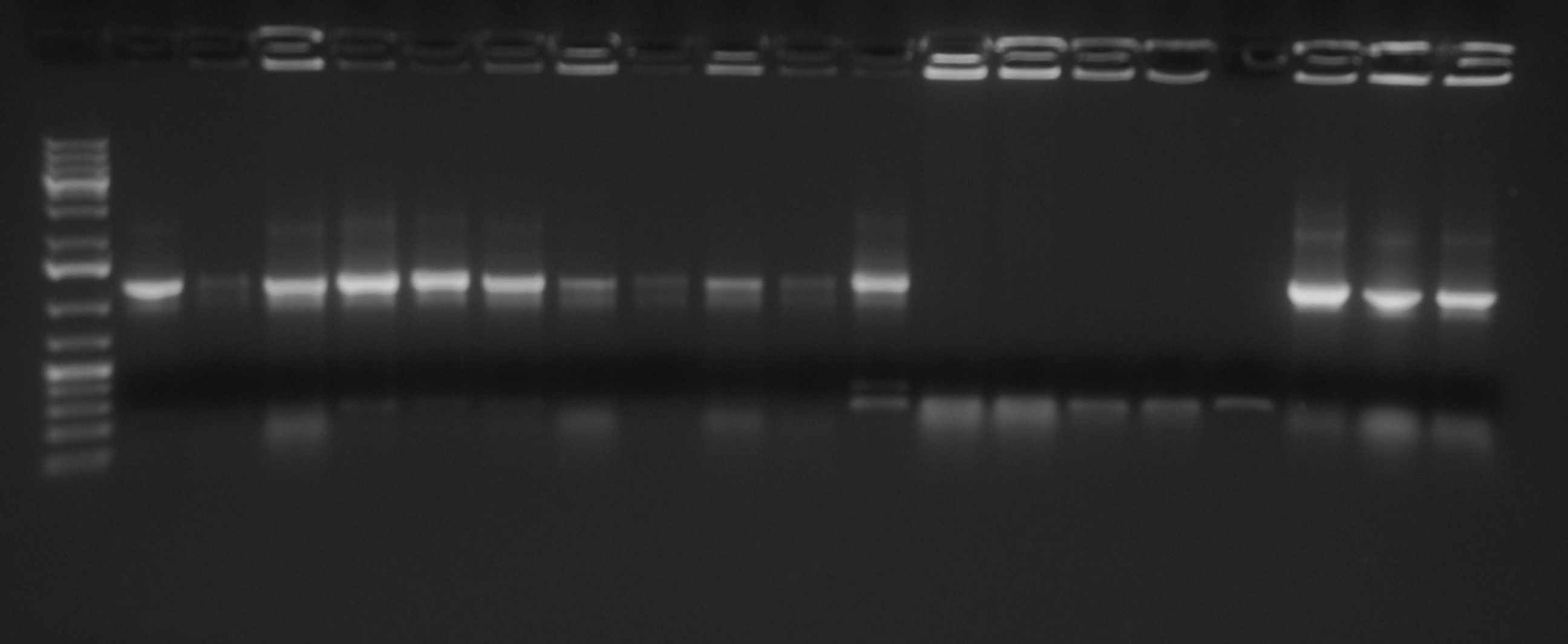 PCR screening for Anderson Promoters transferred in low copy plasmid <partinfo>pSB4C5</partinfo> Gel results show that we have the correct clones for <partinfo>BBa_J23100</partinfo>_4C5, <partinfo>BBa_J23101</partinfo>_4C5, <partinfo>BBa_J23105</partinfo>_4C5, <partinfo>BBa_J23106</partinfo>_4C5, <partinfo>BBa_J23110</partinfo>_4C5, <partinfo>BBa_J23114</partinfo>_4C5. Glycerol stocks were prepared for these parts and are stored at -80°C.
<partinfo>BBa_J23116</partinfo>_4C5 and <partinfo>BBa_J23118</partinfo>_4C5 need to be further screened, so we will pick other colonies next week and we will perform a new colony PCR.
Tecan Test was performed on prepared samples, after the usual protocol (dilution, medium change and dilution 1:1000).
Transformation of I29 in E. coli TOP10
August, 28th
Ligations I47, I48, I49 were stored at +4°C.
I29 plate was stored at +4°C.
|
|