Team:TU Delft/22 July 2010 content
From 2010.igem.org
(Difference between revisions)
Ravandervalk (Talk | contribs) (→Salt tolerance) |
Ravandervalk (Talk | contribs) (→Salt tolerance) |
||
| Line 116: | Line 116: | ||
A number of colonies look promising! To check if they really are the BioBricks we want, tomorrow we will do a plasmid isolation with the cultures of lane 2, 4, 6, 7, 9 and 14. We will cut the isolated plasmids with various restriction enzymes and analyze the digestion products on gel. | A number of colonies look promising! To check if they really are the BioBricks we want, tomorrow we will do a plasmid isolation with the cultures of lane 2, 4, 6, 7, 9 and 14. We will cut the isolated plasmids with various restriction enzymes and analyze the digestion products on gel. | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
Revision as of 09:23, 10 August 2010
Lab work
Alkane degradation
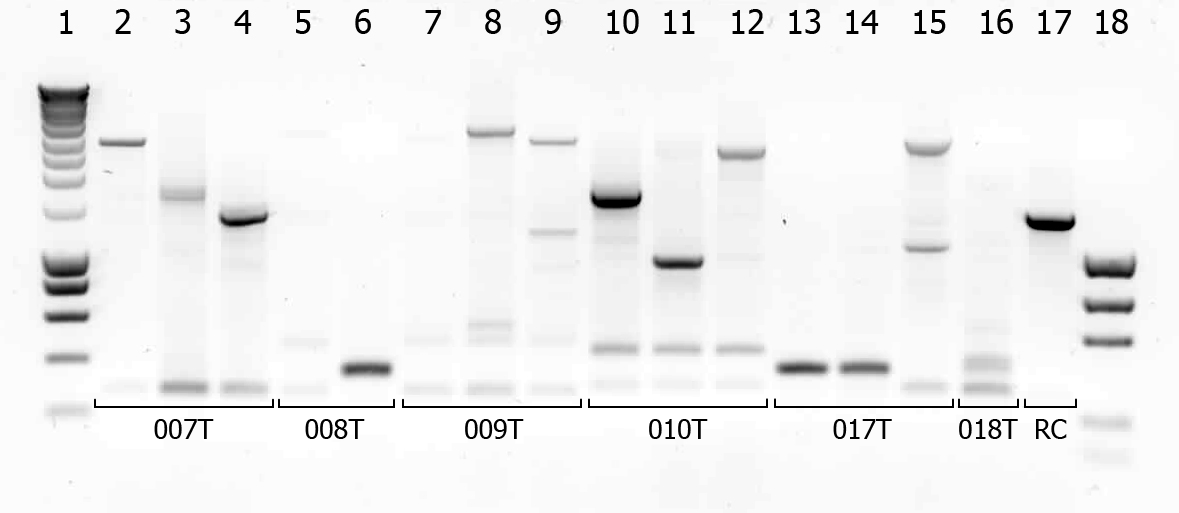
There were some colonies on Tuesday's plates! We had left the plates @ 37°C yesterday after having seen that there were no colonies. When checking this morning on all plates (except the negative control) there were a few colonies! (2-50 colonies). Chances are it's not what we're looking for, but maybe they are good transformants... to check we will do a colony PCR.
| # | Description | Expected length (bp) | Primers | Status |
| 1 | SmartLadder | n/a | n/a | n/a |
| 2 | Transformant #1 of ligation mix 007T | 1616 | G00100 + G00101 | |
| 3 | Transformant #2 of ligation mix 007T | 1616 | G00100 + G00101 | |
| 4 | Transformant #3 of ligation mix 007T | 1616 | G00100 + G00101 | |
| 5 | Transformant #1 of ligation mix 008T | 551 | G00100 + G00101 | |
| 6 | Transformant #2 of ligation mix 008T | 551 | G00100 + G00101 | |
| 7 | Transformant #1 of ligation mix 009T | 551 | G00100 + G00101 | |
| 8 | Transformant #1 of ligation mix 009T | 560 | G00100 + G00101 | |
| 9 | Transformant #1 of ligation mix 009T | 560 | G00100 + G00101 | |
| 10 | Transformant #1 of ligation mix 010T | 1657 | G00100 + G00101 | |
| 11 | Transformant #1 of ligation mix 010T | 1657 | G00100 + G00101 | |
| 12 | Transformant #1 of ligation mix 010T | 1657 | G00100 + G00101 | |
| 13 | Transformant #1 of ligation mix 017T | 1130 | G00100 + G00101 | |
| 14 | Transformant #1 of ligation mix 017T | 1130 | G00100 + G00101 | |
| 15 | Transformant #1 of ligation mix 017T | 1130 | G00100 + G00101 | |
| 16 | Transformant #1 of ligation mix 018T | 1874 | G00100 + G00101 | |
| 17 | Transformant #1 of Red colony | 1360 | G00100 + G00101 |
A number of colonies look promising! To check if they really are the BioBricks we want, tomorrow we will do a plasmid isolation with the cultures of lane 2, 4, 6, 7, 9 and 14. We will cut the isolated plasmids with various restriction enzymes and analyze the digestion products on gel.
 "
"