|
JULY: WEEK 5
July, 26th
July, 27th
July, 28th
July, 29th
We used our new synthesized primers to modify through PCR <partinfo>BBa_K208001</partinfo> phasin in order to create two new parts without stop codon but with Standard prefix and Silver suffix the first one, Silver prefix and suffix the second one.
 Gel run for PCR-modified phasins Gel extraction for phasins showed the following quantifications:
- Pha-10S-1: 15 ng/ul
- Pha-10S-2: 18,5 ng/ul
- Pha-SS-1: 22,8 ng/ul
- Pha-SS-2: 22,4 ng/ul
Phasins were digested X-S for 3 hours and were quantified:
- Pha-10S-1 (X-S): 14 ng/ul
- Pha-10S-2 (X-S): 13,2 ng/ul
- Pha-SS-1 (X-S): 15 ng/ul
- Pha-SS-2 (X-S): 14,9 ng/ul
Competentization of MC1061 (again) because we suspect the presence of a contaminant (it grew without reason on Cm plates).
Transformation of new MC1061 competent cells with:
- 1 ul (4ng) of miniprepped ENTERO-pSB4C5 (positive control);
- 1 ul of RING ligation (RING shouldn't be propagated in MC1061);
- 1 ul of MilliQ (negative control).
Transformed cells have been plated on Cm 12,5 ug/ml agar plates and incubated overnight at 37°C.
Tecan Test
July, 30th
We checked the presence of colonies in plates.
 MC1061 transformed with ENTERO-pSB4C5 (positive control) |  MC1061 transformed with RING |  MC1061 transformed with MilliQ (negative control) |
We calculated (thanks Federica) efficiency as #colonies/ug DNA plated
| Strain | Vector | #colonies | Efficiency
|
| MC1061 | ENTERO-pSB4C5 | 3700 | ~10^6
|
| MC1061 | RING | 0 | -
|
| MC1061 | NOTHING | 0 | -
|
As espected RING and NOTHING didn't grow.
MC1061 competent cells don't replicate RING :) !!!
Miniprep of <partinfo>J04450</partinfo> to take the vector <partinfo>pSB1A3</partinfo>.
3-hour digestion (X-S) and gel run to extract the backbone (~2157 bp).
| Culture | Kind | Final reaction volume (ul) | DNA (ul) | H20 (ul) | Enzyme 1 (ul) | Enzyme 2 (ul) | Buffer H
|
| <partinfo>BBa_J04450</partinfo> | Vector | 25 | 17,2 | 3,3 | 1 X | 1 S | 2,5
|
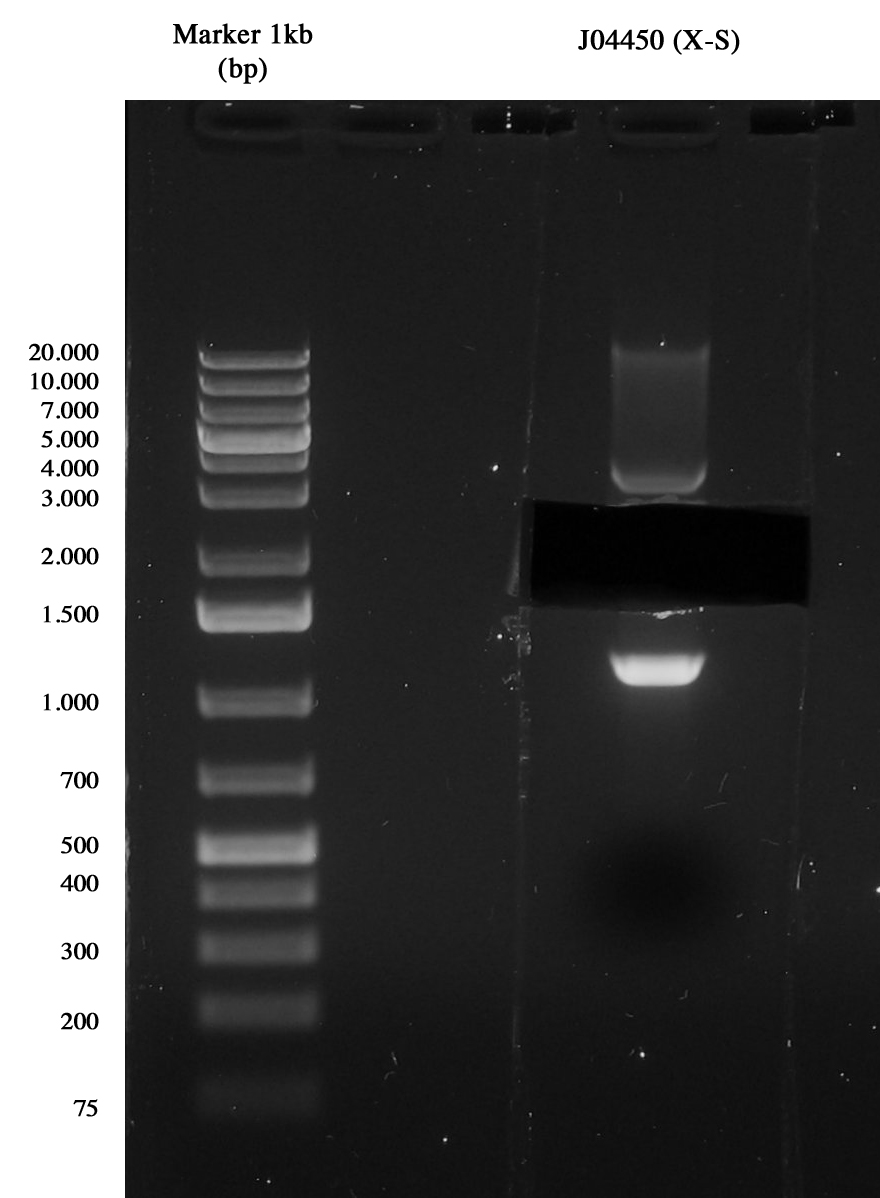 Gel run of J04450 to take pSB1A3 vector Gel extraction was quantified 15,5 ng/ul.
Ligation of:
- I20: Pha-10S-1 (X-S) + pSB1A3 (X-S)
- I21: Pha-SS-1 (X-S) + pSB1A3 (X-S)
July, 31st
Transformation of I20 and I21 into E. coli DH5-alpha. Cells were plated on LB+Amp agar plates and grown overnight at 37°C.
August, 1st
All plates showed colonies (there were also red colonies that contained a wrong ligated plasmid). So we picked three colonies each plate (the right ones) and inoculated them into 5 ml LB+Amp. They were let grow ON at 37°C, 220 rpm.
|  "
"







