Team:Mexico-UNAM-CINVESTAV/Notebook/Week Four
From 2010.igem.org
| Line 10: | Line 10: | ||
==''Monday''== | ==''Monday''== | ||
| - | ==='''Well...we are stuck with the vector | + | ==='''Well...we are stuck with the vector. This week we have to get it.'''=== |
| - | *'''We have achived a | + | *'''We have achived a nano spectrophotometer and readings are below:''' |
| Line 73: | Line 73: | ||
| - | *'''Yep our trouble is the | + | *'''Yep, our trouble is the miniprep method''' |
| - | '''we have a low concentration of | + | '''we have a low concentration of vector plasmid.''' |
| - | '''We have to work an try to get more plasmid and | + | '''We have to work an try to get more plasmid and used diluted PCR products''' |
| - | '''is not | + | '''is not possible achieve ligations with this vector an insert ratio.''' |
| - | ''' | + | '''Our advisor proposed use low Melting agarose''' |
| - | '''to | + | '''to extract the plasmid from the gel.''' |
==''Tuesday''== | ==''Tuesday''== | ||
| - | ==='''We ran with a low meelting | + | ==='''We ran with a low meelting agarose and cut off the band '''=== |
| - | ==='''we | + | ==='''we precipitated using alchol and salts in order '''=== |
| - | ==='''to get vector | + | ==='''to get enough vector to do the ligations.'''=== |
| Line 97: | Line 97: | ||
| - | =='' | + | ==''Wenesday''== |
==='''Plan'''=== | ==='''Plan'''=== | ||
Revision as of 21:33, 26 October 2010
Week #4
27th September - 01 October 2010
Monday
Well...we are stuck with the vector. This week we have to get it.
- We have achived a nano spectrophotometer and readings are below:
| ng/μl | 260/280 | 260/230 | |
| PsB1C3 | 0.60 | -0.73 | -0.0 |
| PCR 1 red | 427.70 | 1.71 | 1.71 |
| PCR 2 red | 483.20 | 1.74 | 1.71 |
| PCR 3 red | 410.70 | 1.76 | 2.02 |
| PCR 4 red | 431.05 | 1.61 | 1.88 |
| PCR 1 blue | 533.35 | 1.71 | 1.75 |
| PCR 2 blue | 536.00 | 1.72 | 1.82 |
| PCR 3 blue | 577.50 | 1.69 | 1.59 |
| PCR 4 blue | 627.55 | 1.70 | 1.56 |
| PsB1C3 | 4.10 | 2.62 | 1.01 |
- Yep, our trouble is the miniprep method
we have a low concentration of vector plasmid.
We have to work an try to get more plasmid and used diluted PCR products
is not possible achieve ligations with this vector an insert ratio.
Our advisor proposed use low Melting agarose
to extract the plasmid from the gel.
Tuesday
We ran with a low meelting agarose and cut off the band
we precipitated using alchol and salts in order
to get enough vector to do the ligations.
Wenesday
Plan
- We prepared all to do ligations PsB1C3 with our Pcr's.
- For this we made a digest with EcoRI and PstI both vector and insert.
- We have run a low melting gel slow 1 hour at 80 volts, then purified from the band.
At Claudia's lab we cut tree bands from 2% agarose's gel of which two of
them where purified by Axigen kit the third one by a diferent kit.
The digestion was done with following amounts.
| DNA | 20μl |
| Buffer NB2 | 3μl |
| EcoRI | 2μl |
| PstI | 2μl |
| H2O | 2.4μl |
| BSA | 0.6μl |
| Total | 30μl |
Thursday
Plan
We going to proceed to do dilutions then ligations
For this we did the following accounts
Poner las fórmulas usadas en un formato bonito
We did the ligations using the following amounts.
| Insert [20ng/μl] | 5μl |
| Vector | 1μl |
| Reaction buffer | 1μl |
| T4 DNA Ligase | 1μl |
| H2O | 2μl |
| Total | 10μl |
We used the follow notation.
- For vector purified from band: Pu
- For iGEM's vector (Vial with green cover): P
- Vector only digest: S
After one hour of ligation (time recomended by the protocol) we transformed by thermic shock
and left overnight plates to 37º
Friday
Today we discused what we going to do with the AFP (anti freeze protein),
We going to transform by thermic shock plating on kanamicina medium, then prepare
miniprep and cut with Xba and PstI we have to ligate at J13002 this as blackbone
the blackbone will cut with SpeI and PstI.
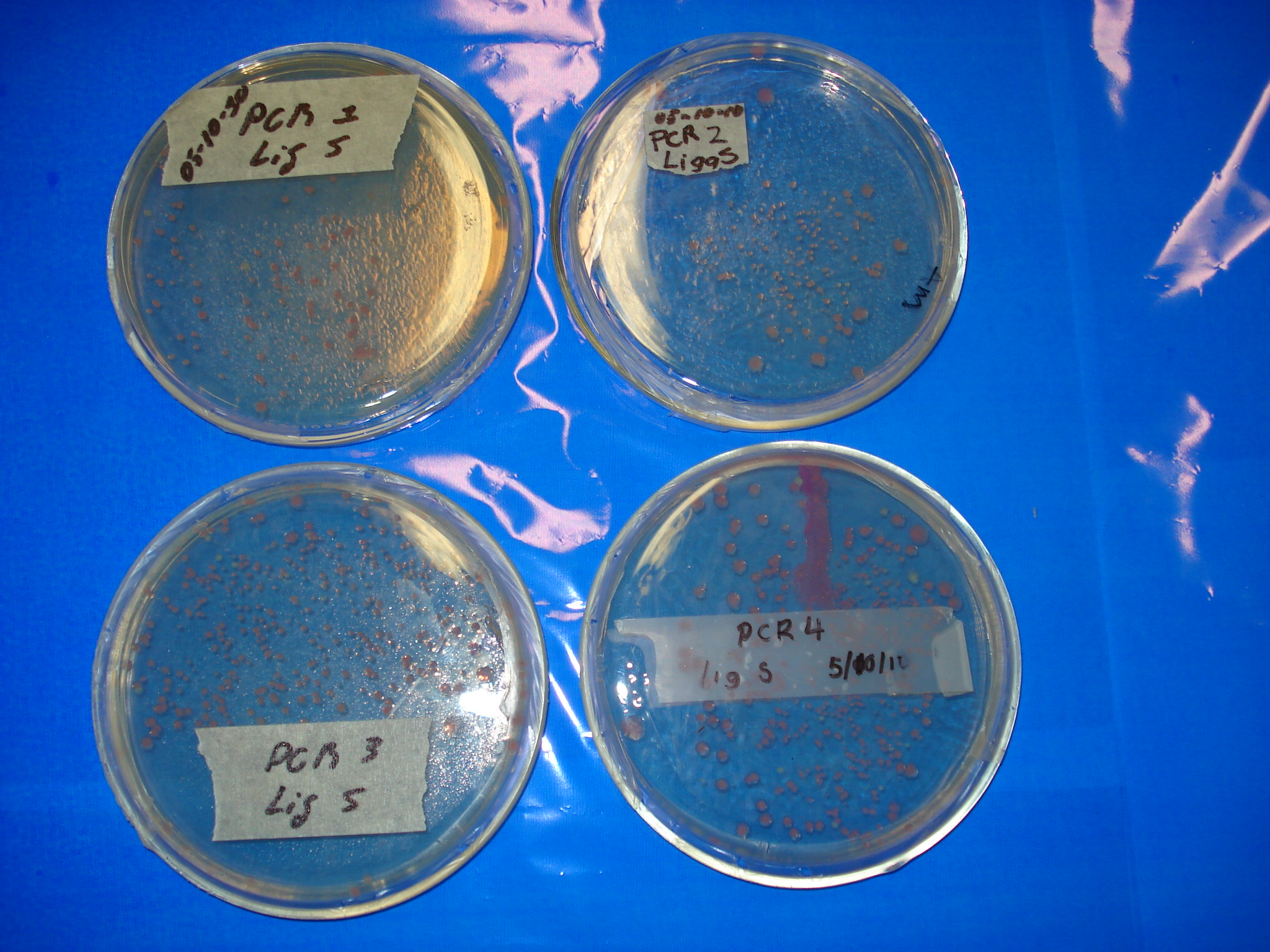
Meanwhile our results of previous day ligations.
- PCR1 Lig S Positive
- PCR2 Lig S Positive
- PCR3 Lig S Positive
- PCR4 Lig Pu Positive
- PCR4 Lig S Positive
 "
"