Team:TU Delft/8 July 2010 content
From 2010.igem.org
(Difference between revisions)
(→Lab work) |
(→Lab work) |
||
| Line 7: | Line 7: | ||
[https://2010.igem.org/Team:TU_Delft#/blog?blog=6_July_2010 Tuesday's] ligation products of BioBricks K398500-K398504 yielded successful transformants. [[Team:TU_Delft/protocols/colony_PCR|Single colony PCRs]] were performed and loaded onto a gel. The bands on the [[Team:TU_Delft/protocols/agarose_gel |1% agarose gel]] indicated the presence of inserts with the proper lengths: | [https://2010.igem.org/Team:TU_Delft#/blog?blog=6_July_2010 Tuesday's] ligation products of BioBricks K398500-K398504 yielded successful transformants. [[Team:TU_Delft/protocols/colony_PCR|Single colony PCRs]] were performed and loaded onto a gel. The bands on the [[Team:TU_Delft/protocols/agarose_gel |1% agarose gel]] indicated the presence of inserts with the proper lengths: | ||
| - | [[Image:C-PCR_transformants_08-July.png| | + | [[Image:C-PCR_transformants_08-July.png|400px|thumb|left|1% Agarose gel of scPCR]] |
Lane descriptions: | Lane descriptions: | ||
Revision as of 19:35, 20 July 2010
Contents |
Lab work
Ordered DNA stocks
We harvested the 200 mL bacterial cells of the 16 DNA parts. We used 3 mL of the bacterial cells to make -80 °C stocks. With the rest we centrifuged at 4,000 rmp for 15 minutes and stored the pellets in the -20 °C.
Characterization of Anderson RBS sequences
Tuesday's ligation products of BioBricks K398500-K398504 yielded successful transformants. Single colony PCRs were performed and loaded onto a gel. The bands on the 1% agarose gel indicated the presence of inserts with the proper lengths:
Lane descriptions:
| # | Description | Expected Length (bp) | Remarks | ||||||||||||||||
| 1 | scPCR product of K398500 transformant #1 (5 μL + 1 μL loadingbuffer) | Unexpected band at 2700 | |||||||||||||||||
| 2 | scPCR product of K398500 transformant #2 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 3 | scPCR product of K398501 transformant #1 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 4 | scPCR product of K398501 transformant #2 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 5 | scPCR product of K398502 transformant #1 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 6 | scPCR product of K398502 transformant #2 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 7 | scPCR product of K398503 transformant #1 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 8 | scPCR product of K398503 transformant #2 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 9 | scPCR product of K398504 transformant #1 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 10 | scPCR product of K398504 transformant #2 (5 μL + 1 μL loadingbuffer) | Visible | |||||||||||||||||
| 11 | scPCR product of ligase control transformant #1 (5 μL + 1 μL loadingbuffer) | 12 | scPCR product of ligase control transformant #2 (5 μL + 1 μL loadingbuffer) | 13 | scPCR product of digestion control transformant #1 (5 μL + 1 μL loadingbuffer) | 14 | scPCR product of digestion control transformant #2 (5 μL + 1 μL loadingbuffer) | 15 | SmartLadder Marker | Varying | Visible |
The colonies belonging to lanes 2, 4, 6, 8 and 10 were plated out on AMP plates to yield reincultures for subsequent characterization experiments.
Alkane degradation
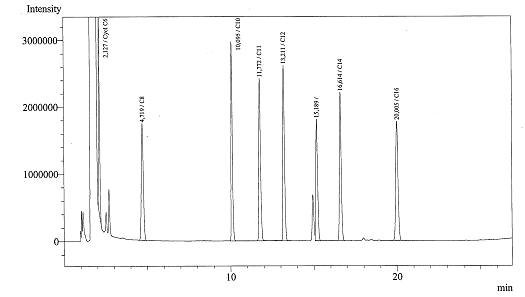
Today we got the Gas Chromatograph working, we could identify several peaks. Now we are ready for the real experiments!
 "
"