| October
|
| M | T | W | T | F | S | S
|
|
|
|
|
| [http://2010.igem.org/Korea_U_Seoul/1_October_2010 1]
| [http://2010.igem.org/Korea_U_Seoul/2_October_2010 2]
| [http://2010.igem.org/Korea_U_Seoul/3_October_2010 3]
|
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/4_October_2010&action=edit 4]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/5_October_2010&action=edit 5]
| [http://2010.igem.org/Korea_U_Seoul/6_October_2010 6]
| [http://2010.igem.org/Korea_U_Seoul/7_October_2010 7]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/8_October_2010&action=edit 8]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/9_October_2010&action=edit 9]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/10_October_2010&action=edit 10]
|
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/11_October_2010&action=edit 11]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/12_October_2010&action=edit 12]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/13_October_2010&action=edit 13]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/14_October_2010&action=edit 14]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/15_October_2010&action=edit 15]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/16_October_2010&action=edit 16]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/17_October_2010&action=edit 17]
|
| [http://2010.igem.org/Korea_U_Seoul/18_October_2010 18]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/19_October_2010&action=edit 19]
| [http://2010.igem.org/Korea_U_Seoul/20_October_2010 20]
| [http://2010.igem.org/Korea_U_Seoul/21_October_2010 21]
| [http://2010.igem.org/Korea_U_Seoul/22_October_2010 22]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/23_October_2010&action=edit 23]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/24_October_2010&action=edit 24]
|
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/25_October_2010&action=edit 25]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/26_October_2010&action=edit 26]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/27_October_2010&action=edit 27]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/28_October_2010&action=edit 28]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/29_October_2010&action=edit 29]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/30_October_2010&action=edit 30]
| [http://2010.igem.org/wiki/index.php?title=Korea_U_Seoul/31_October_2010&action=edit 31]
|
Experimental notes
[Discussion] 2010-08-02 ~ 2010-08-29
1. Strategy and overview of iGEM 2010 experiment
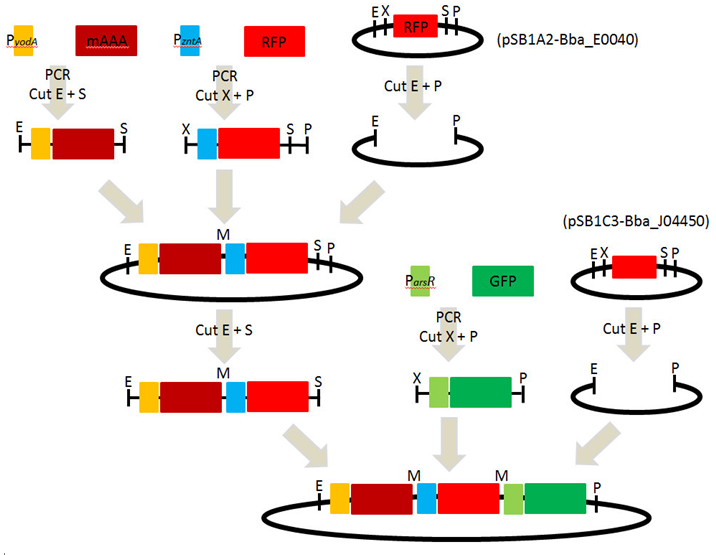
2. Design of primers
| Primer
| Sequence ( 5’ → 3’ )
|
| PyodA(EcoRI)_F
| zz
|
| mAAA(SpeI)_R
| zzzz
|
| PzntA(XbaI)_F
| xzzzz
|
| RFP(PstI)_R
| cxvcxv
|
| ParsR(XbaI)_F
| zzzz
|
| GFP(PstI)_R
| asasas
|
[ Preparation of competent cells ] 2010-09-01 ~ 2010-09-03
1. Inoculation of E. coli DH5α and E. coli BL21(DE3) to 3mL LB broth
2. Preparation of 200mL 2x LB broth, TSS solution and LB plates with ampicillin(100μg/mL) and chloramphenicol(25μg /mL), respectively
3. Inoculation of subcultured E. coli to 200mL 2x LB borth
4. Preparation of competent cells by CSBL laboratory protocol
5. Transformation of pUC19 plasmid(10ng/μL) to competent cells for transformation efficiency check
[ Transformation efficiency ] 2010-09-04
| Strain
| Number of colonies (colonies/μg DNA)
|
| E. coli DH5α
| Number of colonies (colonies/μg DNA)
|
| E. coli BL21(DE3)
| 1.5 x 105
|
[ Amplification of BioBrick parts : pSB1A2 and pSB1C3 ] 2010-09-05
1. Confirmed location : pSB1A2-BBa_E0040 (2010 Kit plate 1/ 14K) and pSB1C3-BBa_J04450 (2010 Kit plate 1/ 3A)
2. 20uL suspension by autoclaved distilled water
3. 3uL transformation to E. coli DH5α
4. Plating to LB(Amp100), LB(Cm25)
1. Inoculation for plasmid DNA purification
2. E. coli K12 genomic DNA extraction by AccuPrep® Genomic DNA Extraction Kit
3. Confirmation of genomic DNA by agarose gel electrophoresis (Figure 1)
4. Quantification of DNA concentration by NanoDrop : 137.5ng/μL
[ Plasmid DNA extraction : pSB1A3 and pSB1C3 ] 2010-09-07
1. Plasmid miniprep by LaboPass™ Plasmid Mini (Plasmid DNA purification kit)
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 2)
3. Quantification of DNA concentration by NanoDrop
[ PCR : promoters and reporter genes ] 2010-09-13 ~ 2010-09-16
1. PCR : PyodA-mAAA, PzntA-RFP(BBa_E1010) and ParsR-GFP(BBa_E0040)
| Reagent
| Volume (μL)
|
| 2.5mM dNTP
| 3
|
| 10x buffer
| 5
|
| Plasmid template (20ng/μL)
| 2
|
| Primers (10pmole/μL)
| 4
|
| α-Taq DNA polymerase (5U/μL)
| 0.5
|
| D.W.
| 35.5/total=50
|
| 95˚C(2’)-[95˚C(20”)-55˚C(20”)-72˚C(2’)]30-72˚C(5’)-4˚C
|
2. Confirmation of PCR products by agarose gel electrophoresis (Figure 3)
3. Purified PCR products
4. Quantification of DNA concentration by NanoDrop
[ Digestion] 2010-09-17
1. Digestion of PCR products and pSB1A2
- 1) PyodA-mAAA : EcoRI and SpeI
- 2) PzntA-RFP : XbaI and PstI
- 3) pSB1A3 : EcoRI and PstI
| Reagent
| Volume (μL)
|
| DNA (about 30ng/μL)
| 30
|
| 10x NEB buffer 2
| 5
|
| BSA (10mg/mL)
| 0.5
|
| Appropriate 1st and 2nd restriction enzymes
| 2 (each 1)
|
| D.W.
| 12.5 / total = 50
|
| Completely digestion at 37˚C for 2 hours (at least)
and stop at 80˚C for 20min
|
2. Confirmation of digested products by agarose gel electrophoresis (Figure 4)
3. Quantification of DNA concentration by NanoDrop
[Chuseok, Korean thanksgiving day] 2010-09-20 ~ 2010-09
[ Ligation & Transformation ] 2010-09-27
1. Ligation of each parts : PyodA-mAAA, PzntA-RFP and pSB1A2
| Reagent
| Volume (μL)
|
| 10x T4 DNA ligase reaction buffer
| 2
|
| T4 DNA ligase
| 2
|
| Each of the digests
| 2 + 2 + 2 = 8
|
| D.W.
| 8 / total = 20
|
| Incubation at room temperature for 30min
and stop at 80˚C for 20min
|
2. Transformation to E. coli DH5α
[ Confirmation of 1st cloning ] 2010-09-28
1. Check : the color of colonies (pSB1A2 : green, recombinant plasmid : white)
2. Inoculation of white colonies to 3mL LB(Amp100)
1. Plasmid DNA purification by LaboPass™ Plasmid Mini
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 5)
3. Recombinant plasmid sequencing by COSMO GeneTech
[ Digestion ] 2010-10-01 ~ 2010-10-03
1. Check : recombinant plasmid sequence
2. Selection of correct clones
3. Digestion of PCR products(ParsR-GFP) and pSB1C3
- 1) ParsR-GFP : EcoRI and SpeI
- 2) pSB1C3 : EcoRI and PstI
| Reagent
| Volume (μL)
|
| DNA (about 30ng/μL)
| 30
|
| 10x NEB buffer 2
| 5
|
| BSA (10mg/mL)
| 0.5
|
| Appropriate 1st and 2nd restriction enzymes
| 2 (each 1)
|
| D.W.
| 12.5 / total = 50
|
| Completely digestion at 37˚C for 2 hours (at least)
and stop at 80˚C for 20min
|
4. Confirmation of digested products by agarose gel electrophoresis (Figure 6)
5. Quantification of DNA concentration by NanoDrop
6. Ligation of each parts : PyodA-mAAA-PzntA-RFP, ParsR-GFP and pSB1C3
| Reagent
| Volume (μL)
|
| 10x T4 DNA ligase reaction buffer
| 2
|
| T4 DNA ligase
| 2
|
| Each of the digests
| 2 + 2 + 2 = 8
|
| D.W.
| 8 / total = 20
|
| Incubation at room temperature for 30min
and stop at 80˚C for 20min
|
7. Transformation to E. coli DH5α
[ Confirmation of 2nd cloning ] 2010-10-06
1. Check : the color of colonies (pSB1C3 : red, recombinant plasmid : white)
2. Inoculation of white colonies to 3mL LB(Amp100)
1. Plasmid DNA purification by LaboPass™ Plasmid Mini
2. Confirmation of extracted plasmids by agarose gel electrophoresis (Figure 7)
3. Recombinant plasmid full-sequencing by COSMO GeneTech
[ Completion : Heavy-metal detector ] 2010-10-18
1. Check : recombinant plasmid sequence
2. Selection of correct clones
3. Transformation to E. coli BL21(DE3) for expression test
|

 "
"
