Team:UCL London/Bioprocess Flowsheet Development
From 2010.igem.org
| Line 27: | Line 27: | ||
The intracellular accumulation of the fragment in the cytoplasm takes the form of inclusion bodies; in this environment refolding cannot be efficiently circumvented where the use mini-chaperones and oxidoreductases must be used for functional fragments (Humphreys et al 2004b). (Tsumoto et al., 1998) reported a highly efficient refolding process for immunoglobulin-folded proteins using stepwise dialysis (Figure 1). The method accentuates the critical factors for highly efficient recovery of proteins from a denatured and reduced state are prevention of aggregation by adding labilising agents and promotion of proper disulphide bond formation at the appropriate stage of refolding. Solubilized inclusion bodies could be refolded under these conditions with yields of up to 95%. | The intracellular accumulation of the fragment in the cytoplasm takes the form of inclusion bodies; in this environment refolding cannot be efficiently circumvented where the use mini-chaperones and oxidoreductases must be used for functional fragments (Humphreys et al 2004b). (Tsumoto et al., 1998) reported a highly efficient refolding process for immunoglobulin-folded proteins using stepwise dialysis (Figure 1). The method accentuates the critical factors for highly efficient recovery of proteins from a denatured and reduced state are prevention of aggregation by adding labilising agents and promotion of proper disulphide bond formation at the appropriate stage of refolding. Solubilized inclusion bodies could be refolded under these conditions with yields of up to 95%. | ||
| + | [[Image:Untitled.jpg|450px|right]] | ||
Another innovative method that could be exploited is the ability of inducing mutations that allow disulphide bonds to form in cytoplasm of ''E. coli'' (Derman et al, 1995). When the mutations are exhibited, export-defective versions of alkaline phosphatase and mouse urokinase were used to fold into their enzymatically active conformations in the cytoplasm because their disulphide bonds were formed. The mutations were mapped to the gene for thioredoxin reductase and diminish or eliminate the activity of this enzyme and thus can be used in keeping cysteines reduced in cytoplasmic proteins. Similarly, (Ohagea et al, 1999) have also resarched in overcoming the reducing enviroment of the cytoplasm, using sequence analysis into isolated immunoglobulin VL domains and ultimately a functional heterodimeric Fv fragment. | Another innovative method that could be exploited is the ability of inducing mutations that allow disulphide bonds to form in cytoplasm of ''E. coli'' (Derman et al, 1995). When the mutations are exhibited, export-defective versions of alkaline phosphatase and mouse urokinase were used to fold into their enzymatically active conformations in the cytoplasm because their disulphide bonds were formed. The mutations were mapped to the gene for thioredoxin reductase and diminish or eliminate the activity of this enzyme and thus can be used in keeping cysteines reduced in cytoplasmic proteins. Similarly, (Ohagea et al, 1999) have also resarched in overcoming the reducing enviroment of the cytoplasm, using sequence analysis into isolated immunoglobulin VL domains and ultimately a functional heterodimeric Fv fragment. | ||
Revision as of 16:15, 25 October 2010
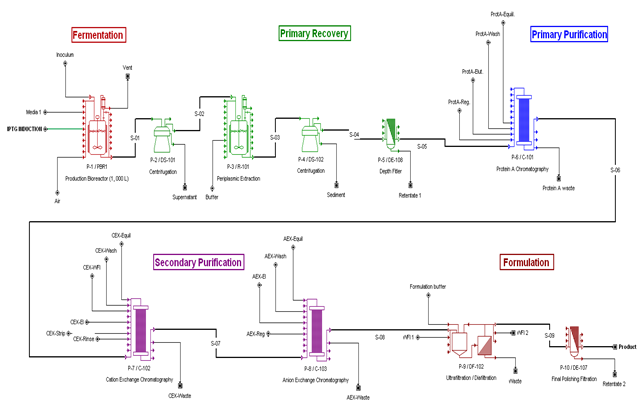
Bioprocess Flowsheet
E. coli Expression
All current monoclonal antibodies on the market and most in clinical trial have been procured through mammalian cell expression systems due to the bacterial cell’s lack of post-translational modifications such as glycosylation and secretion strategies (Farid, 2006). However, for the expression of non-glycosylated antibody fragments such as Fvs, scFvs, Fabs or F(ab’)2s, Escherichia coli is the most renowned host system of choice (A. Pluckthun, 1996).
For example, UCB product CIMZIA ( Crohn's disease), is a pegylated FabV fragment made using E. coli is currently in phase III trials (Reichert et al., 2005). Of the 15 antibodies identified in phase III trials, six were single chain or Fab fragments. Genentech, a global leader in antibody fragment production, manufactures Ranibizumab as Lucentis® at a large scale producing 0.5kg/year generating sales revenues of 1.17 Billion dollars (XOMA Ltd, 2009) can provide the basis for the current manipulation of E. coli and manufacture of the antibody fragment.
Utilising such a copious history of experience with E. coli-based protein expression and the ease of genetic manipulation in E. coli, makes it such an astounding attractive host for expressing antibodies, with research being pioneered to produce full monoclonal forms. The interest relays the necessity of time management where conventional and labour intensive mammalian production systems require approximately four to six months as opposed to a solitary month in E. coli (Peterson et al., 2005); with shorter processing times and increased scale of operation, cost-of-goods may be reduced (Bowering, 2004).
The chief drawback to E. coli expression is the distinct lack of post translational modifications such as glycosylation; thus PEGylation (polyethylene glycol) has found favour due to the ability of PEG to effectively increase the half-life of antibody fragments by altering the solubility, immunogenicity, pharmacokinetics, aggregation and proteolytic susceptibility of therapeutic proteins (Humphreys et al. 2007). The standard protocol for production of antibody fragments require the translocation to the periplasmic space using an N-terminal signal peptide, recognised by the common secretion pathway of E. coli, encoded by the sec genes, which are responsible for the targeted export of most cellular proteins to the extracytoplasmically (Tsumoto et al., 2003).
Periplasmic Expression
This secretion pathway into the periplasm becomes the ideal sub-cellular location for Fab expression due to their oxidising environments that enables formation of intra and inter-chain disulphide bonds (Humphreys., 2004a). Using such expression, the whole cell may not need to homogenised for lysis and rupture but instead ‘periplasmic extraction’ can be exploited maintaining the spheroplast.
The processing step involves the disruption of the outer membrane of the cell by the use of Tris-EDTA lyzozyme, permitting the extraction of the fragment. The product is translocated to the periplasm by an NH2 terminal PelB leader sequence where upon fermentation the addition of sucrose can provide a maximum 25 fold of increase in yield of soluble fragments. The major advantages of protein release into the medium include a higher degree of protection from cellular proteases, a lower degree of contamination with endotoxin and more importantly simplified downstream processing (Georgiou & Segatori, 2005a).
By the same set of researchers (Harvey et al, 2004), Anchored periplasmic expression (APEx) is a developed technology for the isolation of ligand-binding proteins from combinatorial libraries anchored on the periplasmic face of the inner membrane of Escherichia coli. Subsequent to disruption of the outer membrane by Tris-EDTA-lysozyme, the inner-membrane-anchored proteins readily bind fluorescently labelled ligands as large as 240 kDa.
Intracellular Expression
The intracellular accumulation of the fragment in the cytoplasm takes the form of inclusion bodies; in this environment refolding cannot be efficiently circumvented where the use mini-chaperones and oxidoreductases must be used for functional fragments (Humphreys et al 2004b). (Tsumoto et al., 1998) reported a highly efficient refolding process for immunoglobulin-folded proteins using stepwise dialysis (Figure 1). The method accentuates the critical factors for highly efficient recovery of proteins from a denatured and reduced state are prevention of aggregation by adding labilising agents and promotion of proper disulphide bond formation at the appropriate stage of refolding. Solubilized inclusion bodies could be refolded under these conditions with yields of up to 95%.
Another innovative method that could be exploited is the ability of inducing mutations that allow disulphide bonds to form in cytoplasm of E. coli (Derman et al, 1995). When the mutations are exhibited, export-defective versions of alkaline phosphatase and mouse urokinase were used to fold into their enzymatically active conformations in the cytoplasm because their disulphide bonds were formed. The mutations were mapped to the gene for thioredoxin reductase and diminish or eliminate the activity of this enzyme and thus can be used in keeping cysteines reduced in cytoplasmic proteins. Similarly, (Ohagea et al, 1999) have also resarched in overcoming the reducing enviroment of the cytoplasm, using sequence analysis into isolated immunoglobulin VL domains and ultimately a functional heterodimeric Fv fragment.
E.coli Advantages:
• Simple, well-understood genetics
• Established regulatory track record
• Rapid cell growth
• Inexpensive culture media
• Fermentation easy to scale up
• Inclusion bodies may be easy to purify
• No unintended glycosylation
• No viral or prion contamination risk
E.coli Disadvantages
• Intra-cellular expression in Gram-negative bacteria (although some systems have been designed for periplasmatic expression of the target protein)
• Expression of Met-protein at the N-terminus in Gram-negative bacteria
• High endotoxin and host cell protein levels in initial extracts when Gram-negative bacteria have been used
• No post-translational modifications possible (cannot express glycosylated, acetylated and amidated proteins)
• In vitro folding often necessary


 "
"



 Twitter
Twitter Facebook
Facebook UCL
UCL Flickr
Flickr YouTube
YouTube