Team:UNIPV-Pavia/Calendar/June/settimana3
From 2010.igem.org
| (3 intermediate revisions not shown) | |||
| Line 27: | Line 27: | ||
<br> | <br> | ||
<html><p align="center"><font size="4"><b>JUNE: WEEK 3</b></font></p></html><hr><br> | <html><p align="center"><font size="4"><b>JUNE: WEEK 3</b></font></p></html><hr><br> | ||
| + | <html><a name="indice"/></html> | ||
==June, 14th== | ==June, 14th== | ||
| Line 40: | Line 41: | ||
Cultures were grown overnight at 37°C 220 rpm. | Cultures were grown overnight at 37°C 220 rpm. | ||
| + | |||
| + | <div align="right"><small>[[#indice|^top]]</small></div> | ||
==June, 15th== | ==June, 15th== | ||
| Line 112: | Line 115: | ||
Ligation of I4, I5 and I6 was performed at 16°C overnight. | Ligation of I4, I5 and I6 was performed at 16°C overnight. | ||
| + | |||
| + | <div align="right"><small>[[#indice|^top]]</small></div> | ||
==June, 16th== | ==June, 16th== | ||
| Line 121: | Line 126: | ||
1ul of ligation was transformed in 100ul competent cells. | 1ul of ligation was transformed in 100ul competent cells. | ||
| + | |||
| + | <div align="right"><small>[[#indice|^top]]</small></div> | ||
==June, 17th== | ==June, 17th== | ||
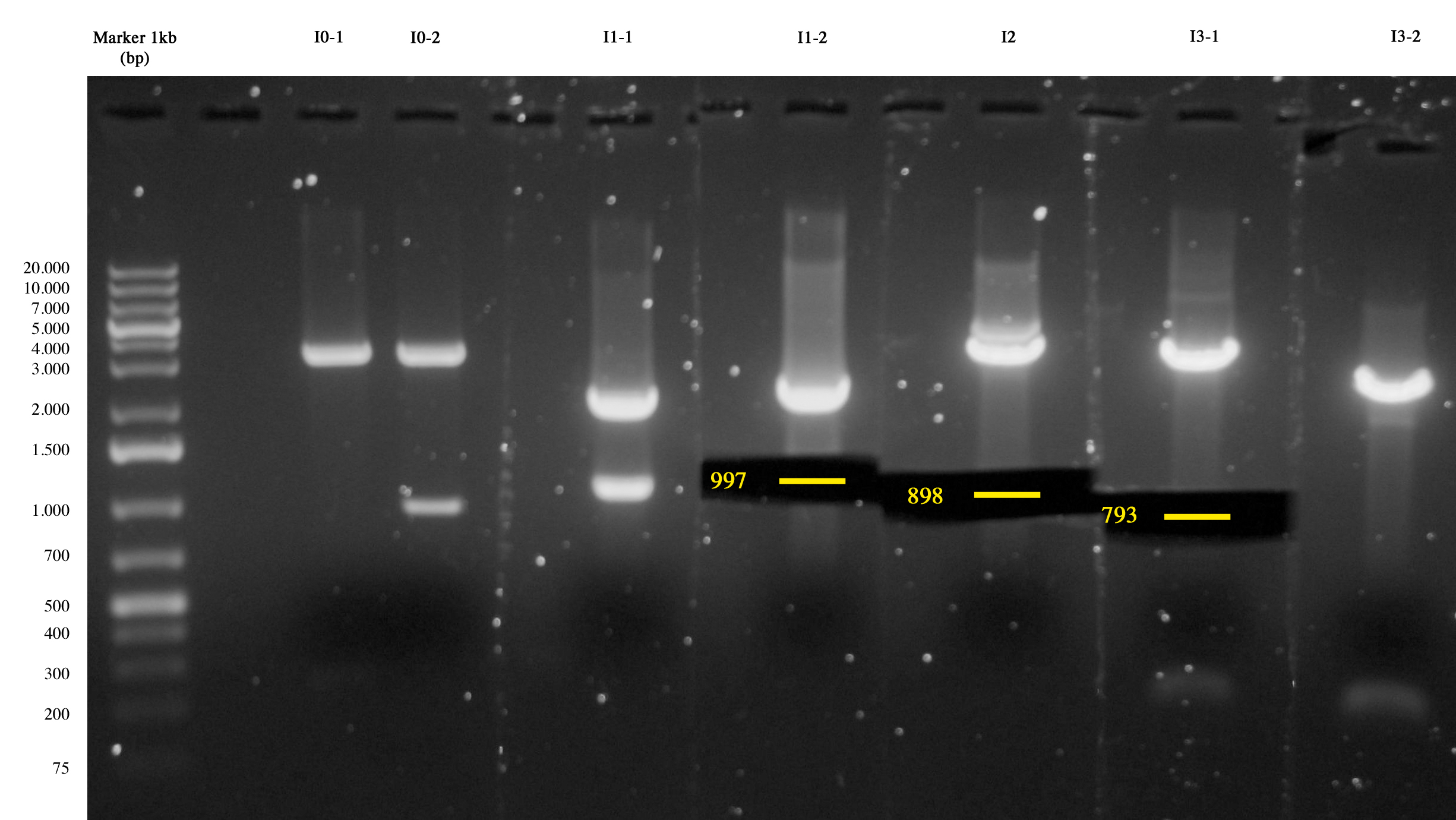
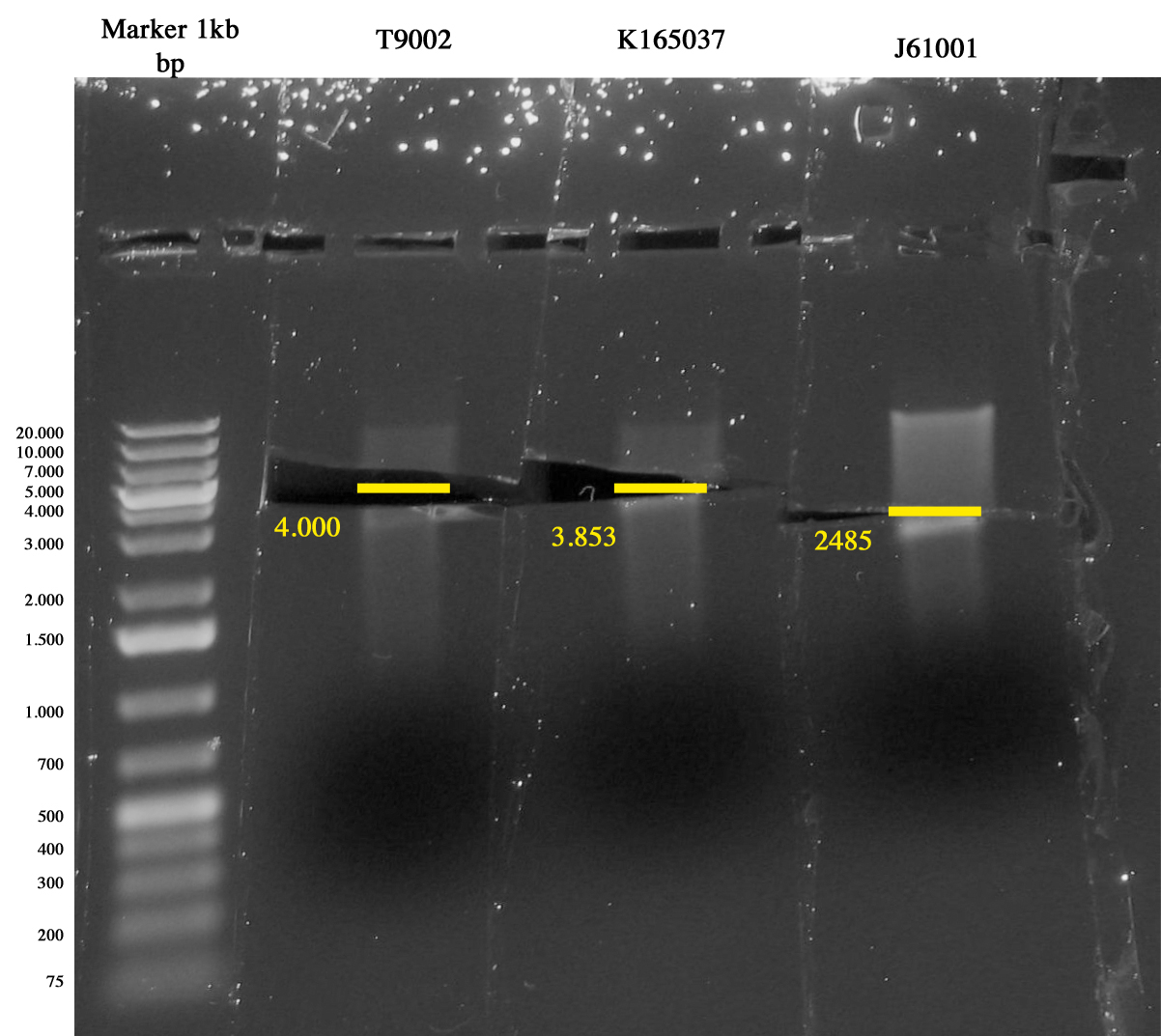
We checked the presence of colonies in plates of I4, I5 and I6 incubated overnight at 37°C. | We checked the presence of colonies in plates of I4, I5 and I6 incubated overnight at 37°C. | ||
| - | All plates showed colonies. I4 had big, round single colonies. I5 showed big colonies surrounded by small colonies. I6 showed few small colonies and for this reason it was further incubated for 3 hours. Colonies were | + | All plates showed colonies. I4 had big, round single colonies. I5 showed big colonies surrounded by small colonies. I6 showed few small colonies and for this reason it was further incubated for 3 hours. Colonies were picked and inoculated in 1ml LB+antibiotic. |
<center> | <center> | ||
{| | {| | ||
| Line 165: | Line 172: | ||
Remaining cultures were re-filled with 5ml LB+Amp and inoculated at 37°C 220rpm for tomorrow MiniPrep. | Remaining cultures were re-filled with 5ml LB+Amp and inoculated at 37°C 220rpm for tomorrow MiniPrep. | ||
| + | |||
| + | <div align="right"><small>[[#indice|^top]]</small></div> | ||
==June, 18th== | ==June, 18th== | ||
| Line 203: | Line 212: | ||
All the clones are OK :) | All the clones are OK :) | ||
| + | |||
| + | |||
| + | <div align="right"><small>[[#indice|^top]]</small></div> | ||
| + | |||
<!-- table previous next week --> | <!-- table previous next week --> | ||
<br><br> | <br><br> | ||
Latest revision as of 07:59, 31 August 2010
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"