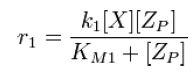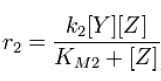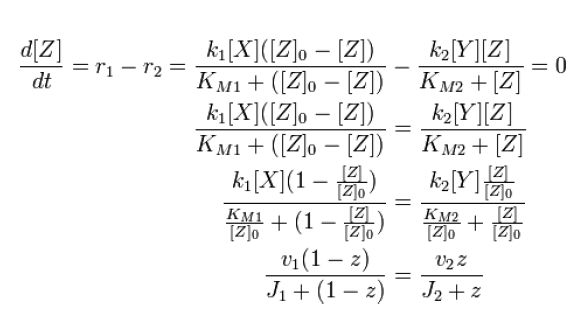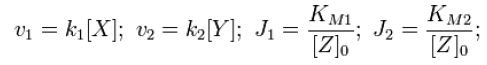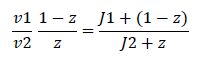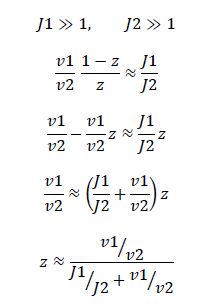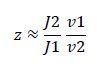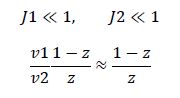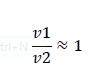Team:Stanford/Research/Modeling
From 2010.igem.org
Drummstikk (Talk | contribs) (→Kinase/Phosphatase System) |
|||
| (25 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
Our intuition for what makes a good ratio sensor could only take us so far. From the very first stages of design, we wanted to back up and test our ideas with mathematical tools. Luckily, we found that solving the equations of mass action kinetics at steady-state was enough to give us clear design criteria. We present the mathematical basis for sensors that are capable of sensing a single ratio digitally, or many ratios in an analog fashion. | Our intuition for what makes a good ratio sensor could only take us so far. From the very first stages of design, we wanted to back up and test our ideas with mathematical tools. Luckily, we found that solving the equations of mass action kinetics at steady-state was enough to give us clear design criteria. We present the mathematical basis for sensors that are capable of sensing a single ratio digitally, or many ratios in an analog fashion. | ||
| - | == | + | ==Kinase/Phosphatase System== |
| + | Throughout this derivation, we refer to Goldbeter and Koshland's seminal work on the kinetics of emzyme pairs [[Team:Stanford/Research/Modeling#References| [1]]]. | ||
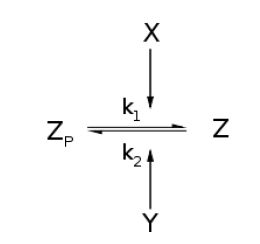
| + | For an opposing pair of enzymes ''X'' and ''Y'' modifying and unmodifying a substrate ''Z'' with modification ''p'', | ||
| + | [[Image:MainGBK.JPG|center]] | ||
| + | From Michaelis-Menten kinetics we know that the rate at which ''Zp'' is dephosphorylated is | ||
| + | [[Image:R1.JPG]][[Team:Stanford/Research/Modeling#References| [2]]] | ||
| - | + | and the rate at which ''Z'' is phosphorylated is | |
| - | + | [[Image:R2.JPG]][[Team:Stanford/Research/Modeling#References| [2]]] | |
| - | + | We also know that ''Z'' can be modified an unmodified, but its total is conserved: ''[Z]o = [Zp] + [Z]'' | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | At steady state, modification and unmodification rates are equal, leading us to | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:1stblock.JPG]][[Team:Stanford/Research/Modeling#References| [2]]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | where | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:2nd_block.JPG]][[Team:Stanford/Research/Modeling#References| [2]]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | Rearranging gives | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:3rdblock.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | Now, assume that the two opposing enzymes are nowhere near being saturated with substrate: | |
| - | + | ||
| - | + | ||
| - | + | [[Image:4thblock.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | which is the equation for a Michaelis-Menten-shaped curve as shown below: | |
| - | + | ||
| - | + | [[Image:Langmuir.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | So, the fraction of modified substrate is a saturating function of the ratio of inputs, the half-max value is the ratio of the strengths of the two enzymes, and the fraction of unmodified substrate is a linear function of the ratio of inputs, | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:6thblock.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | We now know that the requirements for this analog ratio sensor are | |
| - | + | <ol> | |
| - | < | + | <li>Consistent relationships between inputs and enzyme activities (non-trivial)</li> |
| - | + | <li>A non-saturating amount of substrate</li> | |
| - | + | </ol> | |
| - | + | ||
| - | < | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | But what happens if we disobey requirement 1? Let's increase the amount of substrate to saturation so that | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:7thblock.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:8thblock.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | When the enzymes are saturated, the only steady-state solution is'' v1'' = ''v2''. Any ratio of ''v1/v2'' below this, and all the substrate becomes modified. Any ratio above and all the substrate becomes unmodified. Simply by changing the concentration of substrate, we have converted our analog ratio sensor to a digital ratio sensor. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | The requirements for this digital ratio sensor are: | |
| - | + | <ol> | |
| - | + | <li>Consistent relationships between inputs and enzyme activities (non-trivial)</li> | |
| - | + | <li>An amount of substrate sufficient to saturate both opposing enzymes</li> | |
| + | </ol> | ||
| - | + | ==sRNA System== | |
| - | + | This sRNA system is bases its analysis on the work of Nicolas Buchler. [[Team:Stanford/Research/Modeling#References| [3]]] | |
| - | + | ||
| - | + | ||
| - | + | Consider an sRNA that can bind to an mRNA and inhibit its translation. The pair binds and dissociates with dissociation constant Kd. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | For a moment, imagine that this interaction is infinitely strong, such that Kd -> 0. In this case, all the mRNA available will bind to the sRNA, until every sRNA is occupied by one mRNA. Once this threshold, defined by the total sRNA concentration, is reached, additional mRNA added is free to be translated. Here we show the accumulation of translatable free mRNA, and hence Gene Expression vs. total mRNA, in the presence of varying concentrations of infinitely strong sRNA | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:sRNA1.JPG|center]] | |
| - | + | ||
| - | < | + | At the point that the sRNA becomes saturated, two interesting things occur. |
| - | + | <ol> | |
| - | < | + | <li>There is a sharp transition between no gene expression and a linearly-increasing amount of gene expression.</li> |
| - | < | + | <li>This transition occurs only when the concentration of mRNA is equal to the concentration of sRNA.</li> |
| - | + | </ol> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | We have designed a sensor that responds to a 1:1 mRNA:sRNA ratio with a sharp change in output. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | But will this system have the same desirable properties when the sRNA is only realistically strong? Here we assume a 1 nM interaction between sRNA and mRNA | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:sRNA2.JPG|center]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | We find that the transition is still quite sharp when the sRNA has a realistic dissociation constant. | |
| - | + | ||
| - | + | == References == | |
| - | + | [1] [http://www.ncbi.nlm.nih.gov/pubmed/6947258 Goldbeter A, Koshland DE Jr.: An amplified sensitivity arising from covalent modification in biological systems. PNAS. 1981 Nov; 78(11) 6840-4.] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [2] [http://en.wikipedia.org/wiki/Goldbeter-Koshland_kinetics Goldbeter-Koshland kinetics In <i>Wikipedia.</i> Retrieved July 13, 2010, from http://en.wikipedia.org/wiki/Goldbeter-Koshland_kinetics] | |
| - | + | </div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [3] [http://www.ncbi.nlm.nih.gov/pubmed/18938177 Buchler NE, Luis M. Molecular titration and ultrasensitivity in regulatory networks. J. Mol. Biol. 2008 Dec 31; 384(5):1106-19] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Latest revision as of 03:56, 28 October 2010

| Home | Project | Applications | Modeling | Parts | Team | Notebook |
Contents |
Goals
Our intuition for what makes a good ratio sensor could only take us so far. From the very first stages of design, we wanted to back up and test our ideas with mathematical tools. Luckily, we found that solving the equations of mass action kinetics at steady-state was enough to give us clear design criteria. We present the mathematical basis for sensors that are capable of sensing a single ratio digitally, or many ratios in an analog fashion.
Kinase/Phosphatase System
Throughout this derivation, we refer to Goldbeter and Koshland's seminal work on the kinetics of emzyme pairs [1]. For an opposing pair of enzymes X and Y modifying and unmodifying a substrate Z with modification p,
From Michaelis-Menten kinetics we know that the rate at which Zp is dephosphorylated is
and the rate at which Z is phosphorylated is
We also know that Z can be modified an unmodified, but its total is conserved: [Z]o = [Zp] + [Z]
At steady state, modification and unmodification rates are equal, leading us to
where
Rearranging gives
Now, assume that the two opposing enzymes are nowhere near being saturated with substrate:
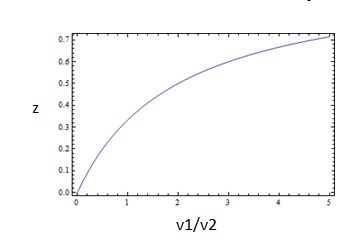
which is the equation for a Michaelis-Menten-shaped curve as shown below:
So, the fraction of modified substrate is a saturating function of the ratio of inputs, the half-max value is the ratio of the strengths of the two enzymes, and the fraction of unmodified substrate is a linear function of the ratio of inputs,
We now know that the requirements for this analog ratio sensor are
- Consistent relationships between inputs and enzyme activities (non-trivial)
- A non-saturating amount of substrate
But what happens if we disobey requirement 1? Let's increase the amount of substrate to saturation so that
When the enzymes are saturated, the only steady-state solution is v1 = v2. Any ratio of v1/v2 below this, and all the substrate becomes modified. Any ratio above and all the substrate becomes unmodified. Simply by changing the concentration of substrate, we have converted our analog ratio sensor to a digital ratio sensor.
The requirements for this digital ratio sensor are:
- Consistent relationships between inputs and enzyme activities (non-trivial)
- An amount of substrate sufficient to saturate both opposing enzymes
sRNA System
This sRNA system is bases its analysis on the work of Nicolas Buchler. [3]
Consider an sRNA that can bind to an mRNA and inhibit its translation. The pair binds and dissociates with dissociation constant Kd.
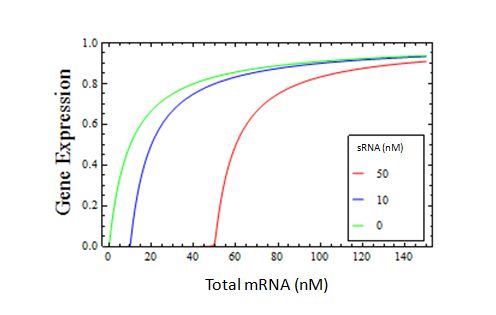
For a moment, imagine that this interaction is infinitely strong, such that Kd -> 0. In this case, all the mRNA available will bind to the sRNA, until every sRNA is occupied by one mRNA. Once this threshold, defined by the total sRNA concentration, is reached, additional mRNA added is free to be translated. Here we show the accumulation of translatable free mRNA, and hence Gene Expression vs. total mRNA, in the presence of varying concentrations of infinitely strong sRNA
At the point that the sRNA becomes saturated, two interesting things occur.
- There is a sharp transition between no gene expression and a linearly-increasing amount of gene expression.
- This transition occurs only when the concentration of mRNA is equal to the concentration of sRNA.
We have designed a sensor that responds to a 1:1 mRNA:sRNA ratio with a sharp change in output.
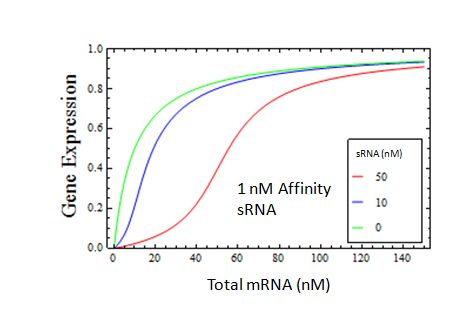
But will this system have the same desirable properties when the sRNA is only realistically strong? Here we assume a 1 nM interaction between sRNA and mRNA
We find that the transition is still quite sharp when the sRNA has a realistic dissociation constant.
References
[1] [http://www.ncbi.nlm.nih.gov/pubmed/6947258 Goldbeter A, Koshland DE Jr.: An amplified sensitivity arising from covalent modification in biological systems. PNAS. 1981 Nov; 78(11) 6840-4.]
[2] [http://en.wikipedia.org/wiki/Goldbeter-Koshland_kinetics Goldbeter-Koshland kinetics In Wikipedia. Retrieved July 13, 2010, from http://en.wikipedia.org/wiki/Goldbeter-Koshland_kinetics]
 "
"